SUMMARY: The American Cancer Society estimates that about 63,990 new cases of kidney cancer will be diagnosed in the United States in 2017 and about 14,400 patients will die from this disease. The understanding of the Immune checkpoints has lead to the development of novel immunotherapies. Immune checkpoints or gate keepers are cell surface inhibitory proteins/receptors that are expressed on activated T cells. They harness the immune system and prevent uncontrolled immune reactions. Survival of cancer cells may be related to their ability to escape immune surveillance, by inhibiting T lymphocyte activation. With the recognition of Immune checkpoint proteins and their role in suppressing antitumor immunity, antibodies have been developed that target the membrane bound inhibitory Immune checkpoint proteins/receptors such as PD-1(Programmed cell Death-1), etc. Following inhibition of PD-1 by specific antibodies, T cells are unleashed, resulting in T cell proliferation and activation with subsequent therapeutic responses.
Preclinical studies have suggested that immune-based therapies for cancer may have a very complex interplay with the host’s microbiome and there may be a relationship between gut bacteria and immune response to cancer. Broad-spectrum antibiotics can alter the bacterial composition and bacterial diversity of our gut, by killing the good bacteria. It has been postulated that this may negate the benefits of immunotherapy and influence treatment outcomes. Previously published studies have shown that intestinal microbiota modulates the anticancer immune effects of YERVOY® (Ipilimumab), an Immune checkpoint inhibitor.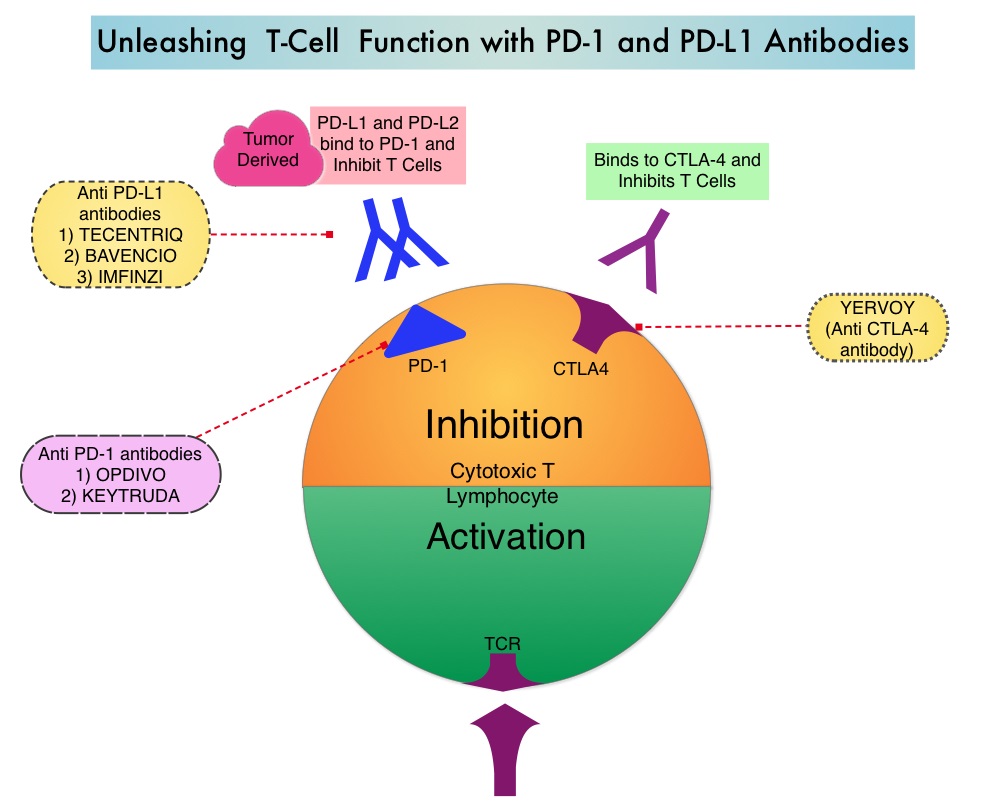
The authors in this publication evaluated the effect of broad-spectrum antibiotic use, in patients with metastatic Renal Cell Carcinoma (mRCC), treated with Immune checkpoint inhibitors. In this study, 80 mRCC patients treated in prospective trials with checkpoint inhibitors were enrolled. These patients had received anti-PD-1/PD-L1 monotherapy, primarily Nivolumab (N=67), a combination of a PD-1 inhibitor and a CTLA-4 inhibitor such as Ipilimumab (N=10), and a combination of anti-PD-L1 therapy and Bevacizumab (N=3). A majority of the patients (65%) were male, 88% had mRCC with clear cell histology, and 80% of the patients had prior nephrectomy. Using IMDC (International Metastatic Renal Cell Carcinoma Database Consortium) criteria, 21% had favorable risk disease, 57% had intermediate risk and 22% had poor risk disease. Sixteen patients (20%) had been treated with broad-spectrum antibiotics, mostly Beta-lactamases or Fluoroquinolones, for up to 1 month prior to starting treatment with immunotherapy. A retrospective analysis was conducted and the Progression Free Survival (PFS), Objective Response Rate (ORR), and Overall Survival (OS), in the group that received broad-spectrum antibiotics, were compared with the group that did not receive broad-spectrum antibiotics.
It was noted that the PFS was four-fold higher in patients who did not receive antibiotics compared to those who received antibiotics (8.1 months vs 2.3 months, P<0.001) and this was statistically significant. This benefit was maintained after adjusting for age, gender, IMDC risk groups, tumor burden and proton pump inhibitors. Additionally, the ORR was higher in those who did not receive antibiotics compared to those who were treated with antibiotics (P<0.002). At a median follow up of less than 6 months, there was already a negative trend in Overall Survival, driven by broad-spectrum antibiotic usage.
It was concluded that in this first analysis, broad-spectrum antibiotics had a negative impact on outcomes, among patients with metastatic Renal Cell Carcinoma, treated with Immune checkpoint inhibitors. Caution should therefore be exercised when prescribing antibiotics to this patient group, so that the efficacy of immunotherapy is not compromised. These concerns may be true across different tumor types and additional follow up is underway. Impact of antibiotics on outcome in patients with metastatic renal cell carcinoma treated with immune checkpoint inhibitors. Derosa L, Routy B, Enot D, et al. J Clin Oncol 35, 2017 (suppl 6S; abstract 462)

