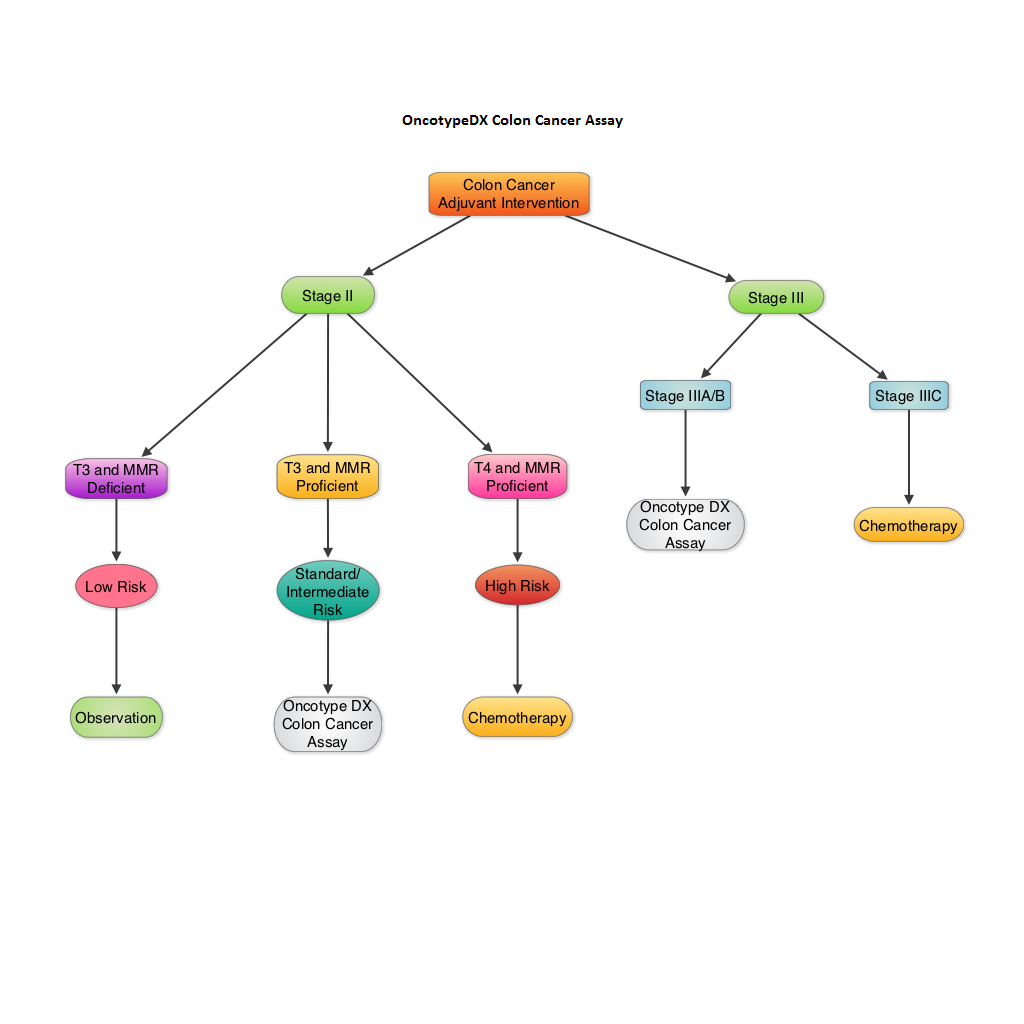
SUMMARY: The beneficial role of adjuvant chemotherapy is established in Stage III colon Cancer. This however is less convincing in patients with stage II colon cancer. The conventional clinical and pathological features such as Tumor stage, Grade, Lymphovascular invasion and number of lymph nodes evaluated, may be of some prognostic significance in Stage II colon cancer, but have not been validated. Nonetheless, a majority of these patients receive ELOXATIN® (Oxaliplatin) based adjuvant chemotherapy, with very little benefit and significant toxicity. The 12-gene Colon Cancer Recurrence Score assay was developed after it was validated in multiple studies in over 3000 patients. This assay is not recommended for Stage II MMR (MisMatchRepair) Deficient patients, as these patients have a better survival and their tumors are resistant to 5-Fluorouracil and may actually do worse with adjuvant chemotherapy following surgery. The recurrent score corresponds to the likelihood of colon cancer recurrence 3 years after surgery, with a higher score suggesting a higher risk of recurrence. The authors in this analysis conducted a prospectively designed, clinical validation study of Recurrence Score, in 892 fixed Paraffin embedded tumor specimens from patients with stage II and III colon cancer, randomly assigned to Fluorouracil (FU) or FU plus ELOXATIN® in the National Surgical Adjuvant Breast and Bowel Project C-07. The primary objective of this study was to retrospectively determine the relationship between the recurrent score and the recurrent risk in stage II/III patients treated with 5-FU or 5-FU/ELOXATIN®. Secondary objectives were to determine if recurrence score provided significant information beyond T stage, tumor grade, MMR status and number of lymph nodes examined. From this analysis, the authors reported that recurrent score performs similarly in both Stage II and Stage III colon cancer patients and predicts recurrence risk beyond T and N stage, Grade, MMR status, number of lymph nodes examined and treatment. Amongst Stage II patients, T4 tumors that are MMR proficient fall in the high risk category and chemotherapy should be considered regardless of the recurrent score whereas T3 tumors that are MMR proficient fall in the standard/intermediate risk category and recurrent score assay may assist in the decision making process. In this group, a recurrence score of 41 or more puts these patients at a recurrence risk that is similar to those who have T4 tumors that is MMR proficient and will therefore benefit from adjuvant chemotherapy. In certain stage IIIA/B patients, a low recurrence score of less than 30 is associated with low absolute benefit with ELOXATIN® and therefore is not recommended. The authors concluded that the 12- Gene Colon Cancer Recurrent Score results can help guide adjuvant therapy decisions for patients with Stage II colon cancer and certain patients with Stage III colon cancer. Yothers G, O’Connell MJ, Lee M, et al. J Clin Oncol 2013;31:4512-4519.