The FDA on April 19, 2019, approved KEYTRUDA® (Pembrolizumab) plus INLYTA® (Axitinib) for the first-line treatment of patients with advanced Renal Cell Carcinoma (RCC). KEYTRUDA® is a product of Merck & Co. Inc. and INLYTA® is a product of Pfizer Inc.
Tag: Renal Cell Carcinoma
Late Breaking Abstract – ESMO 2018 First Line BAVENCIO® plus INLYTA® Highly Effective in Advanced Renal Cell Carcinoma
SUMMARY: The American Cancer Society estimates that 73,820 new cases of kidney and renal pelvis cancers will be diagnosed in the United States in 2019 and about 14,770 people will die from the disease. Renal Cell Carcinoma (RCC) is by far the most common type of kidney cancer and is about twice as common in men as in women. Modifiable risk factors include smoking, obesity, workplace exposure to certain substances and high blood pressure. The five year survival of patients with advanced RCC is less than 10% and there is significant unmet need for improved therapies for this disease. SUTENT® (Sunitinib) is a MultiKinase Inhibitor (MKI) which simultaneously targets the tumor cell wall, vascular endothelial cell wall as well as the pericyte/fibroblast/vascular/ smooth vessel cell wall and is capable of specifically binding to tyrosine kinases, inhibiting the earlier signaling events and thereby inhibits phosphorylation of VEGF receptor, PDGF receptor, FLT-3 and c-KIT. SUTENT® is the standard first-line intervention for treatment naïve patients with advanced RCC. In a large, multi-center, randomized, phase III study, the median Progression Free Survival (PFS) with SUTENT® was 9.5 months, the Objective Response Rate (ORR) was 25%, and the median Overall Survival was 29.3 months, when compared with Interferon Alfa, in patients with treatment-naïve Renal Cell Carcinoma. This was however associated with a high rate of hematological toxicities.
BAVENCIO® (Avelumab) is a human, immunoglobulin G1 lambda, PD-L1 targeted monoclonal antibody that binds to PD-L1 and blocks the interaction between PD-L1 and its receptors PD-1. This in turn negates the inhibitory effects of PD-L1 on the immune response by unleashing the immune system and restoring antitumor immune responses. In addition, BAVENCIO® induces Antibody Dependent Cell-mediated Cytotoxicity (ADCC). INLYTA® (Axitinib) is a kinase inhibitor and inhibits receptor tyrosine kinases including Vascular Endothelial Growth Factor Receptors (VEGFR)-1, VEGFR-2, and VEGFR-3. These receptors have been implicated in pathologic angiogenesis, tumor growth, and cancer progression. INLYTA® is approved by the FDA for the treatment of advanced Renal Cell Carcinoma (RCC) after failure of one prior systemic therapy. The rationale behind combining these two agents was that BAVENCIO® stimulates the immune system while INLYTA® inhibits tumor neoangiogenesis by preventing VEGF activity. Preclinical data suggested that combining these two agents is effective, as their mechanisms of action complement each other. A combination of BAVENCIO® and INLYTA® also showed encouraging antitumor activity among patients with advanced RCC in a phase 1b trial.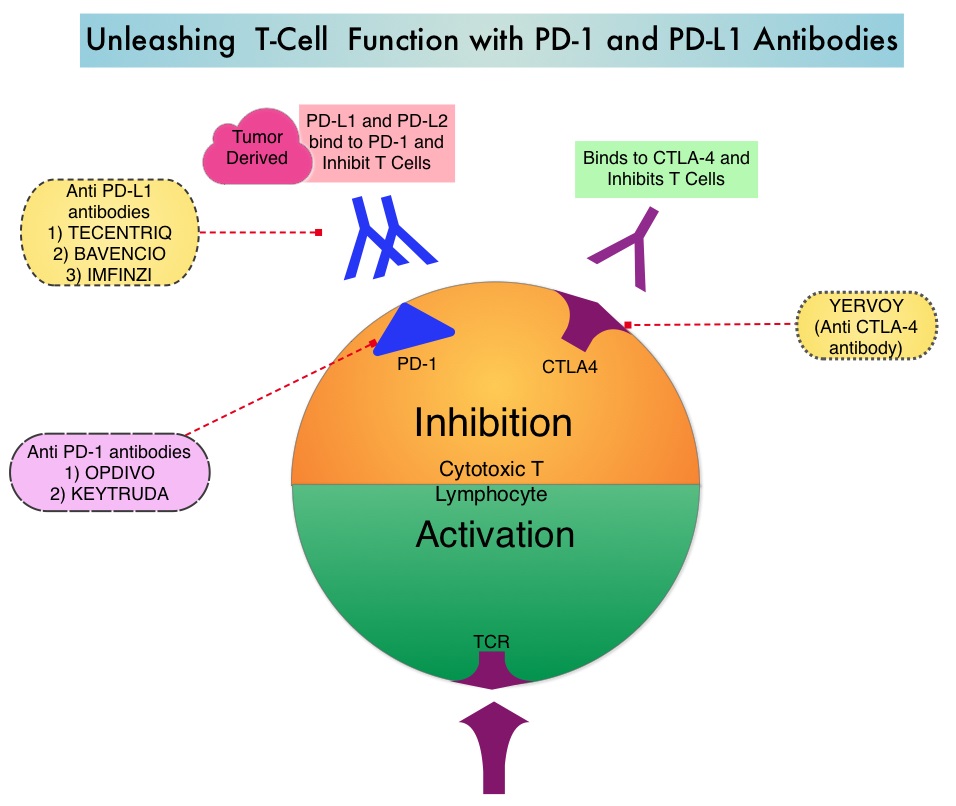
JAVELIN Renal 101 is a global, randomized phase III trial in which 886 patients with clear cell advanced Renal Cell Carcinoma who had no prior systemic therapy, were randomly assigned in a 1:1 to receive BAVENCIO® 10 mg/kg IV every 2 weeks along with INLYTA® 5 mg orally twice daily, in 6 week cycles (N=442) or SUTENT® 50 mg orally daily, 4 weeks on followed by 2 weeks off (N=444). This study included all MSKCC (Memorial Sloan Kettering Cancer Center) prognostic subgroups (good, intermediate, and poor risk). According to the IMDC (International Metastatic RCC Database Consortium), 21% were in the favorable risk group, 62% were in the Intermediate risk group and 16% were in the poor risk group. Among the enrolled patients, 63.2% (N=560) patients were PD-L1positive (1% or more positive immune cells) of whom 270 patients received the BAVENCIO® and INLYTA® combination whereas 290 patients received SUTENT®. The Primary endpoints were Progression Free Survival (PFS) and Overall Survival (OS) in the PD-L1 positive group and Secondary endpoints included PFS and OS irrespective of PD-L1 expression, Objective Response Rate (ORR) and Safety.
It was noted that in the patient group with PD-L1 positive tumors, the median PFS was 13.8 months in the combination group compared to 7.2 months in the single agent SUTENT® group (HR=0.61; P<0.0001). The median PFS in patients irrespective of PD-L1 expression was 13.8 months with the combination treatment compared to 8.4 months with SUTENT® (HR=0.69; P=0.0001). The confirmed Objective Response Rate (ORR) among those with PD-L1 positive tumors was 55.2% in the combination group and 25.5% in the SUTENT® group. The benefit with combination treatment was noted in all prognostic risk groups. The OS data were immature at the time of data cutoff. Grade 3 or more treatment related adverse events were similar in both treatment groups and led to discontinuation of drug in 22.8% of patients in the combination group versus 13.4% in the SUTENT® group.
It was concluded that a combination of BAVENCIO® given along with INLYTA® significantly improved Progression Free Survival as well as Objective Response Rate, irrespective of PD-L1 expression, and across all prognostic risk groups. The authors added that these results support this combination as a potential new first line standard of care for patients with advanced Renal Cell Carcinoma. JAVELIN Renal 101: a randomized, phase III study of avelumab + axitinib vs sunitinib as first-line treatment of advanced renal cell carcinoma (aRCC). Motzer RJ, Penkov K, Hannen JBAG, et al. Proceedings from the 2018 ESMO Congress; October 19-23, 2018; Munich, Germany. Abstract LBA6_PR.
First line treatment with KEYTRUDA® and INLYTA® Combination Significantly Improves Survival in advanced Renal Cell Carcinoma
SUMMARY: The American Cancer Society estimates that 63,340 new cases of kidney cancer will be diagnosed in the United States in 2018 and about 14,970 people will die from the disease. Renal Cell Carcinoma (RCC) is by far the most common type of kidney cancer and is about twice as common in men as in women. Modifiable risk factors include smoking, obesity, workplace exposure to certain substances and high blood pressure. The five year survival of patients with advanced RCC is less than 10% and there is significant unmet need for improved therapies for this disease. SUTENT® (Sunitinib) is a MultiKinase Inhibitor (MKI) which simultaneously targets the tumor cell wall, vascular endothelial cell wall as well as the pericyte/fibroblast/vascular/ smooth vessel cell wall and is capable of specifically binding to tyrosine kinases, inhibiting the earlier signaling events and thereby inhibits phosphorylation of VEGF receptor, PDGF receptor, FLT-3 and c-KIT. SUTENT® is the standard first-line intervention for treatment naïve patients with advanced RCC. In a large, multi-center, randomized, phase III study, the median Progression Free Survival (PFS) with SUTENT® was 9.5 months, the Objective Response Rate (ORR) was 25%, and the median Overall Survival was 29.3 months, when compared with Interferon Alfa, in patients with treatment-naïve Renal Cell Carcinoma. This was however associated with a high rate of hematological toxicities.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response and unleashes the tumor-specific effector T cells. INLYTA® (Axitinib) is a kinase inhibitor and inhibits Receptor Tyrosine Kinases including Vascular Endothelial Growth Factor Receptors (VEGFR)-1, VEGFR-2, and VEGFR-3 at therapeutic plasma concentrations. These receptors have been implicated in pathologic angiogenesis, tumor growth, and cancer progression. Previously published data from a Phase I clinical trial showed that antitumor activity with a combination of KEYTRUDA® and INLYTA® was superior to monotherapy with either PD-1/PD-L1 inhibitor or INLYTA®, in treatment-naïve patients with advanced RCC. Further unlike excess toxicities associated with a combination of VEGF and PD-1 checkpoint inhibitors, a combination of INLYTA® and PD-1 inhibitor was associated with fewer liver function test abnormalities and less fatigue. The excess toxicities have been attributed to off-target effects of multitargeted Tyrosine Kinase Inhibitors.
KEYNOTE-426 is a pivotal, open label, randomized, double-arm, Phase III trial in which the safety and efficacy of KEYTRUDA® in combination with INLYTA® as first-line treatment for advanced or metastatic, clear cell RCC, was compared to SUTENT®. In this study, 861 patients treatment naïve patients who had prior nephrectomy were randomly assigned to receive KEYTRUDA 200 mg IV every three weeks along with INLYTA® 5 mg orally twice daily for up to 24 months, or SUTENT® 50 mg orally once daily for four weeks followed by no treatment for two weeks, given continuously. The dual Primary endpoints were Overall Survival (OS) and Progression Free Survival (PFS). The Secondary endpoints included Objective Response Rate (ORR), Disease Control Rate (DCR), and Duration of Response (DOR). PFS and OS were assessed at 12, 18 and 24 months.
Merck, the manufacturer of KEYTRUDA® in a press release on October 18, 2018 announced that based on the first interim analysis by the independent Data Monitoring Committee (DMC), the study had met the coprimary endpoints and the combination of KEYTRUDA® and INLYTA® resulted in statistically significant and clinically meaningful improvements in Overall Survival and Progression Free Survival, compared to SUTENT® monotherapy. The study also met the key Secondary endpoint of Objective Response Rate, with significant improvements for the KEYTRUDA® and INLYTA® combination, compared with SUTENT® monotherapy. Results for OS, PFS and ORR were consistent regardless of PD-L1 expression and across all risk groups. It was noted that the safety profile of KEYTRUDA® and INLYTA® in this trial was generally consistent with that observed in previously reported studies for each therapy.
According to the manufacturer, this is the first time that combination treatment with an anti PD-1 inhibitor achieved the dual Primary endpoints of Overall Survival and Progression Free Survival, as first line therapy in advanced RCC. Merck’s KEYTRUDA® (pembrolizumab) in Combination with Pfizer’s Inlyta® (axitinib) Significantly Improved Overall Survival (OS) and Progression-free Survival (PFS) as First-Line Therapy for Advanced or Metastatic Renal Cell Carcinoma. Merck. Published October 18, 2018.
OPDIVO® (Nivolumab) and YERVOY® (Ipilimumab)
The FDA on April 16, 2018 granted approvals to OPDIVO® and YERVOY® in combination, for the treatment of intermediate or poor risk, previously untreated advanced Renal Cell Carcinoma. OPDIVO® and YERVOY® are products of Bristol-Myers Squibb Co.
FDA Approves OPDIVO® plus YERVOY® Combination Immunotherapy for intermediate or Poor-risk Advanced Renal Cell Carcinoma
SUMMARY: The FDA on April 16, 2018, granted approvals to OPDIVO® (Nivolumab) and YERVOY® (Ipilimumab) in combination, for the treatment of intermediate or poor-risk, previously untreated advanced Renal Cell Carcinoma (RCC). SUTENT® (Sunitinib) is a MultiKinase Inhibitor (MKI) which simultaneously targets the tumor cell wall, vascular endothelial cell wall as well as the pericyte/fibroblast/vascular/ smooth vessel cell wall and is capable of specifically binding to tyrosine kinases, inhibiting the earlier signaling events and thereby inhibits phosphorylation of VEGF receptor, PDGF receptor, FLT-3 and c-KIT. SUTENT® is the standard first-line intervention for treatment naïve patients with advanced Renal Cell Carcinoma. In a large, multi-center, randomized, phase III study, the median Progression Free Survival (PFS) with SUTENT® was 9.5 months, the Objective Response Rate (ORR) was 25%, and the median Overall Survival was 29.3 months, when compared with Interferon Alfa, in patients with treatment-naïve Renal Cell Carcinoma. This was however associated with a high rate of hematological toxicities.
OPDIVO® is a fully human, immunoglobulin G4 monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, whereas YERVOY® is a fully human immunoglobulin G1 monoclonal antibody that blocks Immune checkpoint protein/receptor CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4, also known as CD152). Blocking the Immune checkpoint proteins unleashes the T cells, resulting in T cell proliferation, activation and a therapeutic response. OPDIVO® was approved by the FDA in November 2015, for the treatment of advanced RCC in patients who had received prior anti-angiogenic therapy, based on an Overall Survival benefit. YERVOY® is approved for the treatment of metastatic melanoma. Combining OPDIVO® with YERVOY® (Combination immunotherapy) has shown promising efficacy in multiple tumor types, including advanced RCC, with higher Objective Response Rate than either agent alone, and is presently approved for the treatment of advanced malignant melanoma.
This FDA approval was based on CheckMate 214, a randomized open-label phase III trial in which a combination of OPDIVO® plus YERVOY® (N=550) was compared with SUTENT® (N=546), among treatment naïve, clear-cell, advanced Renal Cell Carcinoma (RCC) patients. The authors randomly assigned 1096 patients in a 1:1 ratio to receive OPDIVO® 3 mg/kg IV plus YERVOY® 1 mg/kg IV every 3 weeks for four doses (induction phase) followed by OPDIVO® monotherapy at 3 mg/kg every 2 weeks (maintenance phase) or SUTENT® 50 mg orally once daily for 4 weeks, of each 6-week cycle. Four hundred and twenty five (425) patients in the combination group and 422 patients in the SUTENT® group had intermediate or poor-risk patients. It is estimated that approximately 75% of patients with advanced RCC have intermediate or poor-risk disease and have worse outcomes than those with favorable-risk disease. The coprimary end points were Overall Survival, Objective Response Rate and Progression Free Survival among patients with intermediate or poor prognostic risk disease.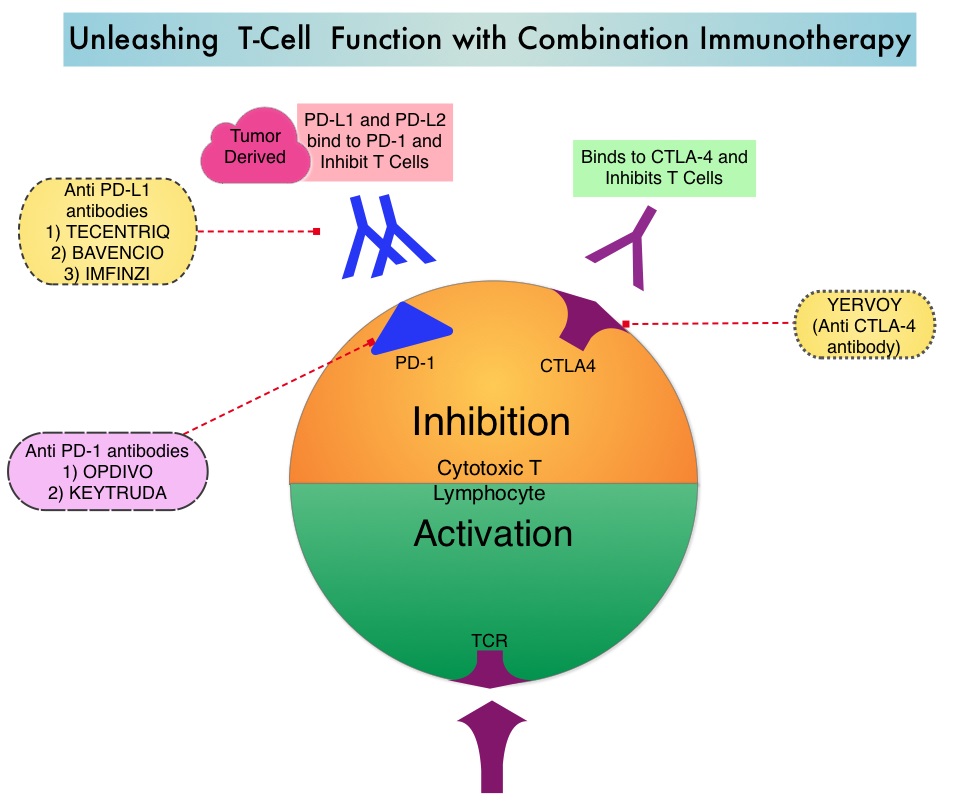
At a median follow-up of 25.2 months, the combination of OPDIVO® and YERVOY® had a significant Overall Survival benefit over SUTENT®. The 18-month Overall Survival rate was 75% with combination immunotherapy and 60% with SUTENT®. The median Overall Survival was not reached with combination immunotherapy versus 26.0 months with SUTENT® (HR=0.63; P<0.001). The Objective Response Rate was 42% with combination immunotherapy versus 27% with SUTENT® (P<0.001), and the Complete Response rate was 9% versus 1% respectively. The median Progression Free Survival was 11.6 months and 8.4 months, respectively but this was not statistically significant per the prespecified threshold. The benefit with combination immunotherapy was not noted in patients with favorable-risk disease. The superior outcomes with combination immunotherapy in patients with intermediate and poor-risk RCC may very well be related to a higher tumor mutational load in this group of patients, compared to those with favorable-risk disease.
In exploratory analyses among 776 intermediate and poor-risk patients, who had quantifiable PD-L1 expression in this study, Overall Survival was longer with Immunotherapy combination compared with SUTENT®, across PD-L1 expression levels. In patients with PD-L1 expression of 1% or greater, the 18-month Overall Survival rate was 81% with combination immunotherapy and 53% with SUTENT®, and the median Overall Survival was not reached versus 19.6 months respectively (HR=0.45). Among patients with PD-L1 expression of 1% or greater, the Objective Response Rate was 58% versus 22% for SUTENT® (P<0.001), the median PFS was 22.8 and 5.9 months, respectively (HR=0.46). A similar trend was noted in patients with PD-L1 expression 5% or greater, as compared with patients with less than 5% PD-L1 expression. Treatment discontinuation, related to adverse events occurred in 22% of the patients in the combination immunotherapy group and 12% in the SUTENT® group.
It was concluded that treatment with a combination of OPDIVO® and YERVOY® resulted in a significantly higher Overall Survival and Objective Response Rates, compared with SUTENT®, among intermediate and poor-risk, previously untreated patients, with advanced Renal Cell Carcinoma. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. Motzer RJ, Tannir NM, McDermott DF, et al. N Engl J Med 2018; 378:1277-1290
CABOMETYX® (Cabozantinib)
The FDA on December 19, 2017 granted regular approval to CABOMETYX® for treatment of patients with advanced Renal Cell Carcinoma (RCC). CABOMETYX® is a product of Exelixis, Inc.
FDA Approves Adjuvant Therapy with SUTENT® in High-Risk Renal Cell Carcinoma after Nephrectomy
SUMMARY: The FDA on November 16, 2017 approved SUTENT® (Sunitinib malate) for the adjuvant treatment of adult patients at high risk of recurrent Renal Cell Carcinoma following nephrectomy. The American Cancer Society estimates that about 62,700 new cases of kidney cancer will be diagnosed in the United States in 2016 and over 14,000 patients will die from this disease. The prognosis for patients with Renal Cell Carcinoma (RCC) is dependent on the stage of disease and risk factors. Two validated models, the University of California Los Angeles Integrated Staging System (UISS) and the Stage, Size, Grade, and Necrosis (SSIGN) score were developed, to assess the risk for relapse. UISS is based on ECOG Performance Status, Fuhrman nuclear grading and TNM pathological stage, whereas the SSIGN score takes Stage, Size, Grade and Necrosis into consideration. Approximately 16% of patients with RCC present with Locoregional disease, and up to 40% of these patients relapse with metastatic disease, following nephrectomy. The 5-year survival for locoregional (stage III) disease is 53%, and 8% for metastatic disease. The standard management of high risk patients following nephrectomy has been surveillance, as there has been limited data demonstrating the benefit of adjuvant therapy in reducing the risk of relapse.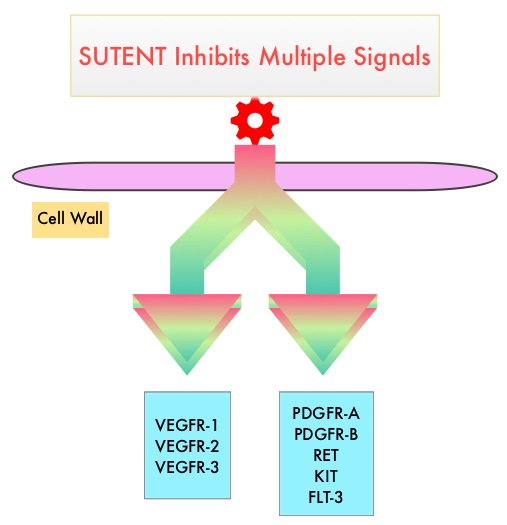
SUTENT® (Sunitinib) is a MultiKinase Inhibitor (MKI) which simultaneously targets the tumor cell wall, vascular endothelial cell wall as well as the pericyte/fibroblast/vascular/ smooth vessel cell wall and is capable of specifically binding to tyrosine kinases, inhibiting the earlier signaling events and thereby inhibits phosphorylation of VEGF receptor, PDGF receptor, FLT-3 and c-KIT. SUTENT® is indicated for the treatment of advanced Renal Cell Carcinoma and in a multi-center, randomized study, demonstrated superior Progression Free Survival and Objective Response Rate, when compared with Interferon Alfa, in patients with treatment-naïve Renal Cell Carcinoma. The authors in this study examined the efficacy and safety of SUTENT® in patients with locally advanced RCC, at high risk for tumor recurrence, following nephrectomy.
Sunitinib as Adjuvant Treatment for Patients at High Risk of Recurrence of Renal Cell Carcinoma Following Nephrectomy (S-TRAC) is a randomized, double blind, phase III trial in which 615 patients with locoregional, high risk, clear cell Renal Cell Carcinoma were randomly assigned to receive SUTENT® (N=309) or placebo (N=306). Treatment consisted of either SUTENT® 50 mg PO daily or placebo, on a 4-weeks-on, 2-weeks-off schedule, for 1 year or until disease recurrence or unacceptable toxicity. Eligible patients had tumor Stage III or higher, regional lymph node metastasis, or both and were required to have absence of macroscopic residual or metastatic disease after nephrectomy, as confirmed by a CT scan. The primary end point was Disease Free Survival and secondary end points included Overall Survival, and Safety.
It was noted that the median duration of Disease Free Survival was 6.8 years in the SUTENT® group and 5.6 years in the placebo group (HR=0.76; P=0.03). Overall Survival data were not mature at the time of this analysis. Grade 3 or 4 adverse events were more frequent in the SUTENT® group compared to the placebo group and dose reductions, dose interruptions and discontinuations were more frequent in the SUTENT® group as well. The most commonly reported adverse events were skin toxicity (palmar-plantar erythrodysesthesia), hypertension, and fatigue, with declines in quality of life while on active therapy. In a previously published adjuvant trial (ASSURE trial), there was no improvement in Disease Free Survival in patients receiving Sunitinib or Sorafenib as compared with placebo. This has been attributed to the ASSURE trial including many patients with early (Stage 1) tumors as well as those with non-clear cell histology. Additionally, the dosing schedule in the ASSURE trial was lower than this present study.
It was concluded that adjuvant treatment with SUTENT® following nephrectomy in patients with high risk disease, results in significantly improved Disease Free Survival but this benefit may be associated with higher rate of toxicities during treatment. Adjuvant Sunitinib in High-Risk Renal-Cell Carcinoma after Nephrectomy. Ravaud A, Motzer RJ, Pandha HS, et al. for the S-TRAC Investigators. N Engl J Med 2016; 375:2246-2254
SUTENT® (Sunitinib malate)
The FDA on November 16, 2017, approved SUTENT® for the adjuvant treatment of adult patients at high risk of recurrent Renal Cell Carcinoma, following nephrectomy. SUTENT® is a product of Pfizer Inc.
Antibiotics May Impair the Efficacy of Immune Checkpoint Inhibitors
SUMMARY: The American Cancer Society estimates that about 63,990 new cases of kidney cancer will be diagnosed in the United States in 2017 and about 14,400 patients will die from this disease. The understanding of the Immune checkpoints has lead to the development of novel immunotherapies. Immune checkpoints or gate keepers are cell surface inhibitory proteins/receptors that are expressed on activated T cells. They harness the immune system and prevent uncontrolled immune reactions. Survival of cancer cells may be related to their ability to escape immune surveillance, by inhibiting T lymphocyte activation. With the recognition of Immune checkpoint proteins and their role in suppressing antitumor immunity, antibodies have been developed that target the membrane bound inhibitory Immune checkpoint proteins/receptors such as PD-1(Programmed cell Death-1), etc. Following inhibition of PD-1 by specific antibodies, T cells are unleashed, resulting in T cell proliferation and activation with subsequent therapeutic responses.
Preclinical studies have suggested that immune-based therapies for cancer may have a very complex interplay with the host’s microbiome and there may be a relationship between gut bacteria and immune response to cancer. Broad-spectrum antibiotics can alter the bacterial composition and bacterial diversity of our gut, by killing the good bacteria. It has been postulated that this may negate the benefits of immunotherapy and influence treatment outcomes. Previously published studies have shown that intestinal microbiota modulates the anticancer immune effects of YERVOY® (Ipilimumab), an Immune checkpoint inhibitor.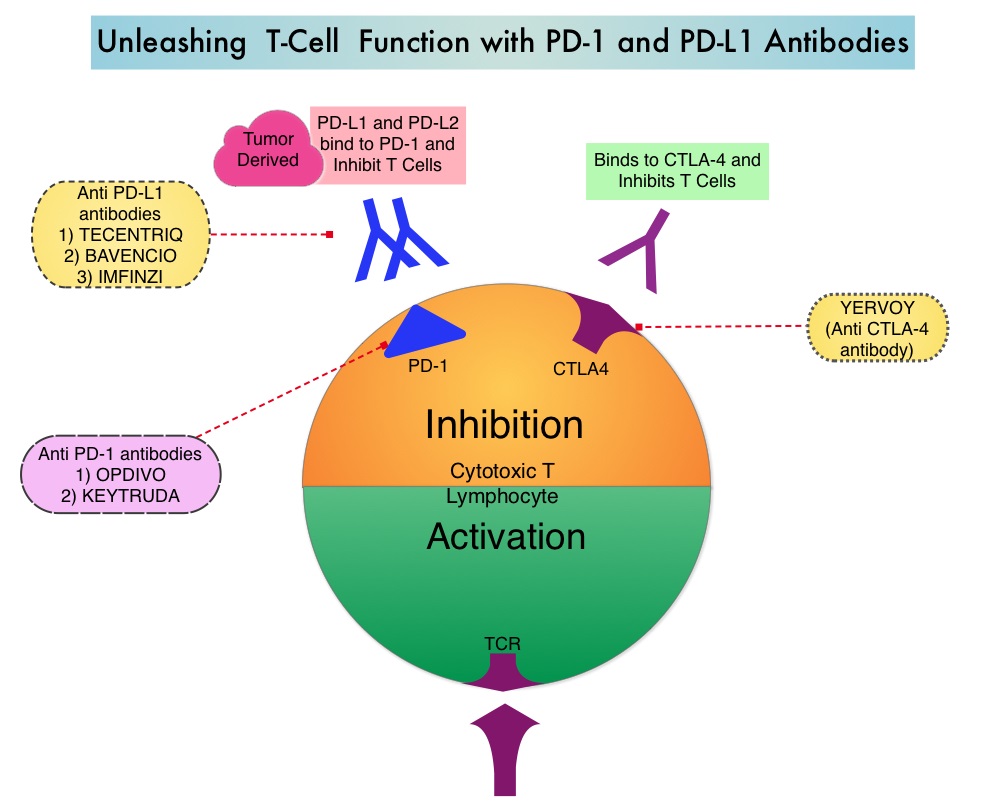
The authors in this publication evaluated the effect of broad-spectrum antibiotic use, in patients with metastatic Renal Cell Carcinoma (mRCC), treated with Immune checkpoint inhibitors. In this study, 80 mRCC patients treated in prospective trials with checkpoint inhibitors were enrolled. These patients had received anti-PD-1/PD-L1 monotherapy, primarily Nivolumab (N=67), a combination of a PD-1 inhibitor and a CTLA-4 inhibitor such as Ipilimumab (N=10), and a combination of anti-PD-L1 therapy and Bevacizumab (N=3). A majority of the patients (65%) were male, 88% had mRCC with clear cell histology, and 80% of the patients had prior nephrectomy. Using IMDC (International Metastatic Renal Cell Carcinoma Database Consortium) criteria, 21% had favorable risk disease, 57% had intermediate risk and 22% had poor risk disease. Sixteen patients (20%) had been treated with broad-spectrum antibiotics, mostly Beta-lactamases or Fluoroquinolones, for up to 1 month prior to starting treatment with immunotherapy. A retrospective analysis was conducted and the Progression Free Survival (PFS), Objective Response Rate (ORR), and Overall Survival (OS), in the group that received broad-spectrum antibiotics, were compared with the group that did not receive broad-spectrum antibiotics.
It was noted that the PFS was four-fold higher in patients who did not receive antibiotics compared to those who received antibiotics (8.1 months vs 2.3 months, P<0.001) and this was statistically significant. This benefit was maintained after adjusting for age, gender, IMDC risk groups, tumor burden and proton pump inhibitors. Additionally, the ORR was higher in those who did not receive antibiotics compared to those who were treated with antibiotics (P<0.002). At a median follow up of less than 6 months, there was already a negative trend in Overall Survival, driven by broad-spectrum antibiotic usage.
It was concluded that in this first analysis, broad-spectrum antibiotics had a negative impact on outcomes, among patients with metastatic Renal Cell Carcinoma, treated with Immune checkpoint inhibitors. Caution should therefore be exercised when prescribing antibiotics to this patient group, so that the efficacy of immunotherapy is not compromised. These concerns may be true across different tumor types and additional follow up is underway. Impact of antibiotics on outcome in patients with metastatic renal cell carcinoma treated with immune checkpoint inhibitors. Derosa L, Routy B, Enot D, et al. J Clin Oncol 35, 2017 (suppl 6S; abstract 462)
CABOMETYX® Superior to SUTENT® in Metastatic Renal Cell Carcinoma
SUMMARY: The American Cancer Society estimates that about 63,990 new cases of kidney cancer will be diagnosed in the United States in 2017 and about 14,400 patients will die from this disease. The VHL (Von Hippel-Lindau) protein is a tumor suppressor gene which is frequently mutated and inactivated in approximately 90% of clear cell Renal Cell Carcinomas (RCC). The VHL gene under normal conditions binds to Hypoxia-Inducible Factor (HIF-1 alpha) and facilitates degradation of this factor. Under hypoxic conditions and in patients having biallelic loss of function and mutation of VHL genes, HIF-1alpha is not degraded. Build up of HIF-1 alpha results in increased angiogenesis, increased tumor cell proliferation and survival, as well as metastasis.
SUTENT® (Sunitinib) is the standard first-line intervention for treatment naïve patients with advanced Renal Cell Carcinoma. VOTRIENT® (Pazopanib) another VEGFR-targeted therapy, is an alternative choice, as it was found to be non-inferior to SUTENT® in the COMPARZ trial. CABOMETYX® (Cabozantinib) is an oral, small-molecule Tyrosine Kinase Inhibitor (TKI) but, unlike SUTENT® which targets the Vascular Endothelial Growth Factor Receptors (VEGFR), CABOMETYX® additionally inhibits the action of tyrosine kinases MET and AXL. Both MET and AXL are up-regulated in Renal Cell Carcinoma as a consequence of VHL inactivation and increased expression of MET and AXL is associated with tumor progression and development of resistance to VEGFR inhibitors. CABOMETYX® was approved by the FDA in 2016 for the treatment of advanced Renal Cell Carcinoma (RCC), in patients who have received prior anti-angiogenic therapy.
The Alliance for Clinical Trials in Oncology reported the results of a randomized, multicenter, open-label phase II CABOSUN trial, which compared CABOMETYX® with standard-of-care SUTENT®, in IMDC intermediate and poor-risk untreated patients with locally advanced or metastatic clear-cell RCC. The study population had a high rate of bone metastases, a known negative prognostic factor in RCC. This study enrolled 157 patients who were randomized in a 1:1 ratio to receive CABOMETYX® 60 mg orally daily (N=79) or SUTENT® 50 mg orally 4 weeks on, 2 weeks off (N=78). A treatment cycle was defined as 6 weeks in both study groups and treatment was continued until disease progression or intolerance to therapy. Patients were stratified by IMDC risk category (intermediate or poor) and presence of bone metastases. Crossover between treatment groups was not allowed. The median age was 63 years and 81% of the enrolled patients were classified as IMDC intermediate risk and 19% as poor risk, 36% of patients had bone metastases and 75% of the patients had prior nephrectomy. The Primary end point was Progression Free Survival (PFS) and Secondary end points included Objective Response Rate (ORR), Overall Survival (OS) and safety.
It was noted that the treatment with CABOMETYX® significantly increased median PFS compared with SUTENT® (8.2 versus 5.6 months) and was associated with a 34% reduction in rate of disease progression or death (HR=0.66; P=0.012). The ORR was 46% in the CABOMETYXreg; group compared with 18% in the SUTENT® group. The median OS with CABOMETYX® was 30.3 months versus 21.8 months with SUTENT® (HR=0.80), with this preliminary data showing a 20% decrease in the risk of death with CABOMETYX®. Grade 3 or 4 adverse events were similar in both treatment groups with more hypertension and Palmar-Plantar Erythrodysesthesia in the CABOMETYX® group whereas the SUTENT® group experienced more fatigue and hematologic toxicities. Discontinuation rates related to adverse events were similar in both treatment groups.
The authors concluded that CABOMETYX® significantly improved PFS and ORR compared to SUTENT®, in the initial treatment of patients with intermediate or poor-risk clear cell metastatic Renal Cell Carcinoma. CABOMETYX® is the first agent to demonstrate clinical superiority over SUTENT®, which has been the established standard of care for more than 10 years. The authors attributed the superiority of CABOMETYX® over SUTENT® due to its mechanism of action, which targets MET and AXL, in addition to VEGFR. Cabozantinib Versus Sunitinib As Initial Targeted Therapy for Patients With Metastatic Renal Cell Carcinoma of Poor or Intermediate Risk: The Alliance A031203 CABOSUN Trial. Choueiri TK, Halabi S, Sanford BL, et al. DOI: 10.1200/JCO.2016.70.7398 Journal of Clinical Oncology 35, no. 6 (February 2017) 591-597.
