SUMMARY: The FDA in 2017 granted accelerated approval to KEYTRUDA® (Pembrolizumab), a PD-1 blocking antibody, for adult and pediatric patients with unresectable or metastatic, MicroSatellite Instability-High (MSI-H) or MisMatch Repair Deficient (dMMR) solid tumors that have progressed following prior treatment, and who have no satisfactory alternative treatment options. This has led to routine MSI-H/dMMR testing in advanced solid tumors. The DNA MisMatchRepair (MMR) system is responsible for molecular surveillance and works as an editing tool that identifies errors within the microsatellite regions of DNA and removes them. Defective MMR system leads to MSI (Micro Satellite Instability) and hypermutation, triggering an enhanced antitumor immune response. MSI is therefore a hallmark of defective/deficient DNA MisMatchRepair (dMMR) system and occurs in 15% of all colorectal cancers.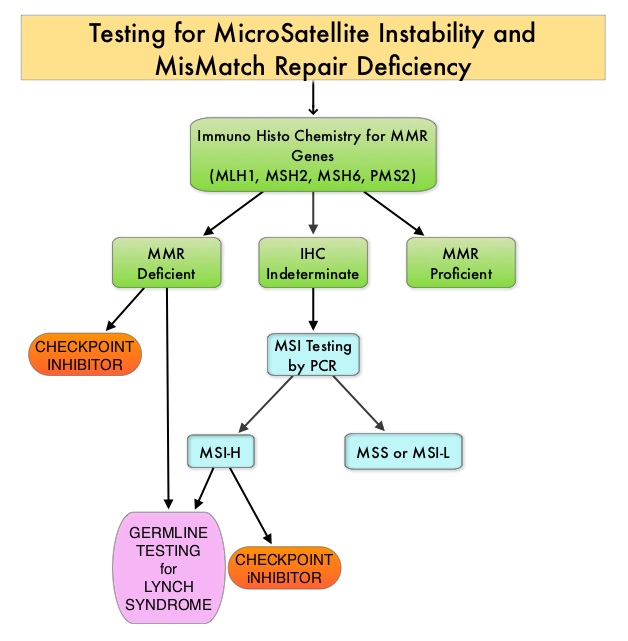
Defective MisMatchRepair can be a sporadic or heritable event. Defective MisMatchRepair can manifest as a germline mutation occurring in MisMatchRepair genes including MLH1, MSH2, MSH6, PMS2 and EPCAM. This produces Lynch Syndrome often called Hereditary Nonpolyposis Colorectal Carcinoma – HNPCC, an Autosomal Dominant disorder, that is often associated with a high risk for Colorectal and Endometrial carcinoma, as well as several other malignancies including Ovary, Stomach, Small bowel, Hepatobiliary tract, Brain and Skin. MSI is a hallmark of Lynch Syndrome-associated cancers. MSI tumors tend to have better outcomes and this has been attributed to the abundance of tumor infiltrating lymphocytes in these tumors from increase immunogenicity. These tumors therefore are susceptible to blockade with immune checkpoint inhibitors. MSI testing is performed using a PCR based assay and MSI-High refers to instability at 2 or more of the 5 mononucleotide repeat markers and MSI-Low refers to instability at 1 of the 5 markers. Patients are considered Micro Satellite Stable (MSS) if no instability occurs. MSI-L and MSS are grouped together because MSI-L tumors are uncommon and behave similar to MSS tumors. Tumors considered MSI-H have deficiency of one or more of the DNA MisMatchRepair genes. MMR gene deficiency can be detected by ImmunoHistoChemistry (IHC). NCCN Guidelines recommend MMR or MSI testing for all patients with a history of Colon or Rectal cancer. Unlike Colorectal and Endometrial cancer, where MSI-H/dMMR testing is routinely undertaken, the characterization of Lynch Syndrome across heterogeneous MSI-H/dMMR tumors is unknown.
The aim of the study was to determine the prevalence of germline mutations in the DNA mismatch repair genes diagnostic of Lynch Syndrome, across MSI-H tumors. The researchers in this study analyzed 15,045 tumor samples collected from patients with more than 50 different types of cancer using a comprehensive genomic test called MSK-IMPACT (Integrated Mutation Profiling of Actionable Cancer Targets), a next-generation sequencing platform. The MSK-IMPACT assay is a qualitative in-vitro diagnostic test that uses targeted next-generation sequencing of Formalin Fixed Paraffin-Embedded (FFPE) tumor tissue matched with normal specimens, from patients with solid tumors, to detect tumor gene alterations in a broad multigene panel. It is the first multiplex tumor profiling test to receive FDA authorization. The MSK-IMPACT test can look for gene mutations and other errors in all solid tumors, regardless of their origin.
Scores of less than 3, 3-9 and 10 or more were designated MSS, MSI-Indeterminate (MSI-I) or MSI-H status, respectively. Germline mutations were assessed in MLH1, MSH2, MSH6, PMS2, EPCAM. ImmunoHistoChemical staining (IHC) for dMMR, and tumor signatures in Lynch Syndrome patients were assessed.
Of the tumor samples analyzed, 93.2% were MSS, 4.6% were MSI-I, and 2.2% were MSI-H. Germline mutations indicative of Lynch Syndrome were identified in 0.3% of microsatellite-stable tumors, 1.9% of MSI-I tumors, and 16.3% of MSI-H tumors (P<0.001). The authors noted that nearly 50% of patients with MSI-H/MSI-I tumors identified as having Lynch Syndrome, had cancers other than colorectal or endometrial carcinoma – the two malignancies that are typically seen with Lynch Syndrome. The cancer types identified that were previously not linked to or rarely, linked to the Lynch Syndrome, included Mesothelioma, Sarcoma, Adrenocortical cancer, Melanoma, Prostate and Ovarian germ cell cancer. Nearly 40% of these patients did not meet the genetic testing criteria for Lynch Syndrome. MMR-deficiency was found in 98.3% of MSI-I/MSI-H tumor samples.
It was concluded that MSI-H/dMMR tumors, regardless of cancer type and irrespective of the family history, should prompt germline testing for the evaluation of Lynch Syndrome. This will increase the ability to recognize Lynch Syndrome, not only in the patients tested, but also in at-risk family members, thus enabling the implementation of enhanced surveillance and risk reduction measures. Pan-cancer microsatellite instability to predict for presence of Lynch syndrome. Schwark AL, Srinivasan P, Kemel Y, et al. J Clin Oncol 36, 2018 (suppl; abstr LBA1509)

