SUMMARY: The FDA on August 20, 2020, approved KYPROLIS® (Carfilzomib) and DARZALEX® (Daratumumab), in combination with Dexamethasone, for adult patients with Relapsed or Refractory multiple myeloma, who have received one to three lines of therapy. Multiple Myeloma is a clonal disorder of plasma cells in the bone marrow and the American Cancer Society estimates that in the United States, 32, 270 new cases will be diagnosed in 2020 and 12,830 patients are expected to die of the disease. Multiple Myeloma (MM) in 2020 remains an incurable disease. The therapeutic goal therefore is to improve Progression Free Survival (PFS) and Overall Survival (OS). Multiple Myeloma is a disease of the elderly, with a median age at diagnosis of 69 years and characterized by intrinsic clonal heterogeneity. Almost all patients eventually will relapse, and patients with a high-risk cytogenetic profile or refractory disease have the worst outcomes. The median survival for patients with Myeloma is over 10 years.
REVLIMID® (Lenalidomide) in combination with VELCADE® (Bortezomib) and Dexamethasone is the preferred regimen according to the NCCN guidelines, for both transplant and non-transplant candidates with newly diagnosed Multiple Myeloma, and when given continuously or with maintenance therapy, has improved survival outcomes. Nonetheless, a significant number of patients progress while on these agents or discontinue therapy due to toxicities. There is therefore a need for effective and tolerable regimens for patients who are exposed or refractory to REVLIMID® or VELCADE®.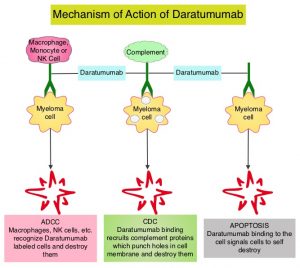
KYPROLIS® (Carfilzomib) is a second generation selective, epoxyketone Proteasome Inhibitor and unlike VELCADE®, proteasome inhibition with KYPROLIS® is irreversible. DARZALEX® (Daratumumab) is a human IgG1 antibody that targets CD38, a transmembrane glycoprotein abundantly expressed on malignant plasma cells and with low levels of expression on normal lymphoid and myeloid cells. DARZALEX® exerts its cytotoxic effect on myeloma cells by multiple mechanisms, including Antibody Dependent Cellular Cytotoxicity (ADCC), Complement Dependent Cytotoxicity (CDC) and direct Apoptosis. Additionally, DARZALEX® may play a role in immunomodulation, by depleting CD38-positive regulator immune suppressor cells, and thereby expanding T cells, in patients responding to therapy. Both KYPROLIS® and DARZALEX® are approved as single agents, as well as in combination with other drugs, for the treatment of patients with Relapsed/Refractory Multiple Myeloma. In a Phase I study, KYPROLIS® in combination with Dexamethasone and DARZALEX® demonstrated safety and efficacy in patients Relapsed/Refractory Multiple Myeloma.
The efficacy of KYPROLIS® and DARZALEX® along with Dexamethasone was evaluated in two clinical trials, CANDOR and EQUULEUS. CANDOR is a multicenter, open-label, Phase III trial, which included Relapsed/Refractory Multiple Myeloma patients with measurable disease who had received 1-3 prior lines of therapy, with Partial Response or better to one or more lines of therapy. A total of 466 patients were randomly assigned 2:1 to receive triplet of KYPROLIS®, Dexamethasone, and DARZALEX® (KdD)- N=312 or KYPROLIS® and Dexamethasone (Kd) alone- N=154. All patients received KYPROLIS® as a 30 minute IV infusion on days 1, 2, 8, 9, 15, and 16 of each 28-day cycle (20 mg/m2 on days 1 and 2 during cycle 1 and 56 mg/m2 thereafter). DARZALEX® 8 mg/kg was administered IV on days 1 and 2 of cycle 1 and at 16 mg/kg once weekly for the remaining doses of the first 2 cycles, then every 2 weeks for 4 cycles (cycles 3-6), and every 4 weeks thereafter. All patients received Dexamethasone 40 mg oral or IV weekly (20 mg for patients over 75 years of age). The median age was 64 years, 42% and 90% received prior REVLIMID® and VELCADE® (Bortezomib) containing regimens respectively, and a third of patients were refractory to REVLIMID®. The Primary endpoint was Progression Free Survival (PFS) and Secondary endpoints included Overall Response Rate (ORR), Minimal Residual Disease (MRD)-negative status, Complete Response (CR) rate at 12 months, Overall Survival (OS), Duration of Response, and Safety.
After a median follow up of 17 months, the study met its Primary endpoint and the median PFS was not reached for the KdD arm and was 15.8 months for the Kd arm (HR=0.63; P=0.0027). This represented a 37% reduction in the risk of progression or death in the KdD group. The PFS benefit of KdD was maintained across prespecified subgroups, particularly among REVLIMID®-exposed and REVLIMID®-refractory patients. The ORR was 84.3% in the KdD group versus 74.7% in the Kd group (P=0.004), with a CR rate or better of 28.5% versus 10.4% respectively. The median time to first response was one month in both treatment groups. Patients treated with KdD achieved deeper responses which was nearly 10 times higher, with a MRD-negative Complete Response rate at 12 months of 12.5% for KdD versus 1.3% for Kd (P<0.0001). The median treatment duration was longer in the KdD group compared to the Kd group (70.1 versus 40.3 wks). The median OS was not reached in either groups, at a median follow up time of 17 months. Toxicities were generally manageable and the incidence of Adverse Events leading to treatment discontinuation was similar in both treatment groups.
EQUULEUS is an open label, multicohort trial which evaluated the combination of KYPROLIS® administered weekly on days 1, 8, and 15 of each 28-day cycle (20 mg/m2 IV on cycle 1, day 1, and if tolerated, increased to 70 mg/m2 on Cycle 1 Day 8 and thereafter) along with DARZALEX® IV and Dexamethasone (KdD). Efficacy was based on Overall Response Rate (ORR). Of the 85 patients with Relapsed or Refractory multiple myeloma who had received 1 to 3 prior lines of therapy enrolled in the KdD cohort, the ORR was 81%, with response duration of 27.5 months.
It was concluded that a combination of KYPROLIS® along with Dexamethasone and DARZALEX® resulted in a significant PFS benefit over KYPROLIS® and Dexamethasone alone, with deeper responses, and the PFS benefit of KdD was maintained across prespecified, clinically important subgroups, particularly REVLIMID®-exposed and REVLIMID®-refractory patients. The authors added that KdD regimen should be considered as a novel, efficacious, and tolerable immunomodulatory-free treatment option for Relapsed/Refractory Multiple Myeloma patients.
Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Dimopoulos M, Quach H, Mateos M-V, et al. The Lancet 2020;396:186-197.

