The FDA on May 1, 2018 approved KYMRIAH®, a CD19-directed genetically modified autologous T-cell immunotherapy, for adult patients with relapsed or refractory large B-cell lymphoma, after two or more lines of systemic therapy including Diffuse Large B-Cell Lymphoma (DLBCL) not otherwise specified, high grade B-cell lymphoma and DLBCL arising from Follicular Lymphoma. KYMRIAH® is a product of Novartis Pharmaceuticals Corp.
Author: RR
KYPROLIS® along with REVLIMID® and Dexamethasone Improves Overall Survival in Relapsed or Refractory Multiple Myeloma
SUMMARY: Multiple Myeloma is a clonal disorder of plasma cells in the bone marrow and the American Cancer Society estimates that in the United States, 30,770 new cases will be diagnosed in 2018 and 12,770 patients are expected to die of the disease. Multiple Myeloma in 2018 remains an incurable disease. The therapeutic goal therefore is to improve Overall Survival (OS) in the relapsed setting.
KYPROLIS® (Carfilzomib) is a second generation selective, epoxyketone proteasome inhibitor and unlike VELCADE® (Bortezomib), proteasome inhibition with KYPROLIS® is irreversible. Most of the recent phase III trials in Relapsed or Refractory Myeloma have used Progression Free Survival (PFS) as the Primary end point, with the exception of the phase III trial ENDEAVOR trial in which patients treated with KYPROLIS® and Dexamethasone achieved a statistically significant 7.6-month improvement in median Overall Survival (OS) compared to those patients treated with VELCADE® and Dexamethasone (HR=0.79; P=0.01). REVLIMID® (Lenalidomide) given along with weekly Dexamethasone, was associated with significantly improved Progression Free Survival (PFS) when administered until disease progression, in patients with newly diagnosed Multiple Myeloma. The combination of REVLIMID® and weekly Dexamethasone is considered a reference regimen for both newly diagnosed and relapsed Multiple Myeloma. VELCADE® in combination with REVLIMID® and Dexamethasone showed an overall response rate of 64% and a median PFS of 9.5 months in patients with Relapsed or Refractory Multiple Myeloma.
Based on this background, a randomized, open label, multicenter, phase III study (ASPIRE) was conducted, in which the safety and efficacy of a combination of KYPROLIS® (Carfilzomib), REVLIMID® and weekly Dexamethasone (KRd) was compared with a combination of REVLIMID® and weekly Dexamethasone (Rd), in patients with Relapsed or Refractory Multiple Myeloma. Seven hundred and ninety two (N=792) patients were randomly assigned in a 1:1 ratio to KRd (N=396) and Rd (N=396). Eligible patients included those with Multiple Myeloma who had received one to three prior treatments which included VELCADE® or REVLIMID® and Dexamethasone combination, provided that they did not have disease progression during treatment with these agents. The 28 day treatment cycle consisted of KYPROLIS® IV given on days 1, 2, 8, 9, 15, and 16 (starting dose, 20 mg/m2 on days 1 and 2 of cycle 1 with a target dose of 27 mg/m2 thereafter) during cycles 1 through 12 and on days 1, 2, 15, and 16 during cycles 13 through 18, following which KYPROLIS® was discontinued. REVLIMID® 25 mg PO was given on days 1 through 21 and Dexamethasone 40 mg PO was administered on days 1, 8, 15, and 22. Patients in both treatment groups received only REVLIMID® and Dexamethasone after cycle 18 until disease progression. Antiviral and antithrombotic prophylaxis was administered to patients in both treatment groups. The Primary end point was Progression Free Survival (PFS) and secondary end points included Overall Survival (OS), the rate of overall response (partial response or better), response duration, health-related quality of life, and safety.
The study met its Primary endpoint at the time of the pre-specified interim analysis with a significant improvement in the median PFS for those patients in the KRd group compared to the Rd (26.3 months versus 17.6 months; HR=0.69; P=0.0001). This benefit in the PFS was demonstrated across all predefined subgroups. The overall response rates (partial response or better) were 87.1% and 66.7% in the KYPROLIS® and control groups, respectively (P<0.001). Further, patients in the KYPROLIS® group reported superior health-related quality of life.
The authors in this prespecified analysis reported the final Overall Survival (OS) data and updated safety results. The median follow up was 67.1 months. The median OS was 48.3 months in the KRd group and 40.4 months in the Rd group (HR=0.79; P=0.0045). This represented a 7.9 month prolongation of OS and 21% decrease in the risk of death with KRd. Among patients who had received one prior line of therapy, KRd improved median OS by 11.4 months and among those who had received 2 or more prior lines of therapy, KRd improved median OS by 6.5 months, compared to Rd. Among patients who had received one prior line of VELCADE® based therapy, the median OS was improved by 12 months with KRd versus Rd, with a 18% reduction in the risk of death, and among patients with prior transplantation at first relapse, the median OS was improved by 18.6 months with KRd versus Rd, with a 29% reduction in the risk of death. The OS benefit with KRd was noted across all age, Creatinine Clearance (CrCL) and ECOG PS subgroups, including those 75 years or older, patients with impaired renal function (CrCL 30 to less than 60 mL/min), and patients with decreased Performance Status (ECOG PS, 2). The median time to next treatment from time of randomization was 39.0 months for patients who received KRd and 24.4 months for those who received Rd (HR=0.65; P<0.001).
An updated median PFS with longer follow up (median , 48.4 months) was 26.1 months in the KRd group versus 16.6 months in the Rd group (HR=0.66; P<0.001). Grade 3 or higher adverse events were reported in 87% and 83.3% of patients in the KRd and Rd groups, respectively.
It was concluded that treatment with KRd resulted in a statistically significant and clinically meaningful reduction in the risk of death, compared to Rd, among patients with Relapsed or Refractory Myeloma. This analysis supports the early use of KYPROLIS® at first relapse, regardless of prior treatment with VELCADE® or transplantation. Because each subsequent line of therapy can result in shorter response duration and increased treatment resistance, the authors suggested that early treatment with an effective regimen is important to maximize Overall Survival and KRd regimen should be considered a preferred treatment option in Relapsed or Refractory Multiple Myeloma. Improvement in Overall Survival With Carfilzomib, Lenalidomide, and Dexamethasone in Patients With Relapsed or Refractory Multiple Myeloma. Siegel DS, Dimopoulos MA, Ludwig H, et al. J Clin Oncol. 2018;36:728-734
Dual HER2 Blockade Along with an Aromatase Inhibitor is an Effective Chemotherapy-Sparing Alternative Treatment in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1 (EGFR), HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Approximately 50% of HER2-positive breast cancers are Hormone Receptor positive.
Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine).
Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival. The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Not all HER2-positive, Hormone Receptor positive metastatic breast cancer patients, are candidates for chemotherapy. These patients however may benefit from anti-HER2 targeted therapy given along with endocrine therapy.
Preclinical evidence suggested that endocrine resistance may be related to cross talk between HER2- and Hormone Receptor-signaling pathways. HER2 inhibition may in turn enable the Estrogen Receptor (ER) to become the primary driver of cell proliferation, resulting in relative resistance to anti-HER2 therapy. Therefore, targeting both HER2 and ER simultaneously may be essential to derive optimal benefit among patients with HER2-positive Hormone Receptor positive metastatic breast cancer. Previously published studies demonstrated improved median PFS when single HER2 blockade combined with endocrine therapy was compared with endocrine therapy alone, among treatment naïve patients with HER2-positive and Hormone Receptor positive metastatic breast cancer.
Based on the improved outcomes with dual anti-HER2 blockade compared with single HER2 blockade in both neoadjuvant as well as metastatic settings, this present study was designed to evaluate the superiority of dual HER2 blockade with TYKERB® and HERCEPTIN® given along with an Aromatase Inhibitor (AI), over single HER2 blockade with HERCEPTIN® given along with an AI, in patients with HER2-positive, Hormone Receptor positive metastatic breast cancer, who experienced disease progression after prior neo(adjuvant)/first-line HERCEPTIN® based chemotherapy. This study also included a third treatment arm of TYKERB® plus an AI, which was compared with the other two treatment groups.
The ALTERNATIVE study is an open-label, phase III trial, in which 355 patients were randomly assigned in a 1:1:1 ratio to receive either TYKERB® along with HERCEPTIN® plus an AI (N=120), HERCEPTIN® plus an AI (N=117) or TYKERB® plus an AI. TYKERB® was administered at 1000 mg orally daily in the dual HER2 blockade group and at 1500 mg orally daily in the TYKERB® plus AI group. Patients receiving TYKERB® were urged to initiate treatment with Loperamide at the onset of diarrhea. HERCEPTIN® was administered IV at a loading dose of 8 mg/kg, followed by the maintenance dose of 6 mg/kg IV every 3 weeks. Physician’s choice of AIs included either Letrozole 2.5 mg, Anastrozole 1 mg or Exemestane 25 mg, orally daily. Enrolled patients were postmenopausal women, with histologically or cytologically confirmed ER-positive and/or Progesterone Receptor-positive, HER2-positive metastatic breast Cancer, as determined in a local laboratory. Prior treatment with endocrine therapy and disease progression during or after a prior HERCEPTIN® based chemotherapy regimen in the neo(adjuvant) setting and/or in the first-line metastatic setting, was a requirement for enrollment in this study. Only one prior regimen in the metastatic setting was allowed. Patients for whom chemotherapy was felt appropriate per treating physician’s judgement, were excluded from the study. Two thirds of the patients had received HERCEPTIN® based regimens in adjuvant setting and approximately one third in metastatic setting. The Primary end point was Progression Free Survival (PFS) with dual HER2 blockade plus AI compared with HERCEPTIN® plus AI. Secondary end points included PFS comparison of other treatment groups, Overall Survival, Overall Response Rate (ORR), Clinical Benefit Rate and Safety.
The study met its Primary end point and the median PFS utilizing dual HER2 blockade with a combination of TYKERB® along with HERCEPTIN® plus an AI was 11 months, compared with 5.7 months for HERCEPTIN® plus an AI (HR=0.62, P=0.0064). This represented a 38% reduction in the risk of disease progression. The PFS benefit was consistently observed in various predefined subgroups of patients. Further, the ORR and Clinical Benefit Rate were superior in the TYKERB® along with HERCEPTIN® plus AI group compared to HERCEPTIN® plus AI group ((31.7% versus 13.7% and 41% versus 31%, respectively). Although survival data were immature, there was also a trend favoring treatment with dual HER2 (median 46 months versus 40 months). When other treatment groups were compared, the median PFS with TYKERB® plus an AI was 8.3 months compared to 5.7 months with HERCEPTIN® plus an AI (HR=0.71, P=0.036), suggesting that among HER2-positive, Hormone Receptor positive metastatic breast cancer patients, who had progressed after prior treatment with HERCEPTIN®, anti-HER2 treatment with TYKERB® along with an AI may be a reasonable alternative, although this hypothesis will need to be confirmed. Common adverse events included higher incidence of diarrhea and rash in the groups treated with TYKERB® and serious adverse events were similar across the three treatment groups. Treatment discontinuation due to adverse events was lower in the dual HER2 blockade group.
The authors concluded that dual HER2 blockade with a combination of TYKERB®, HERCEPTIN® and an Aromatase Inhibitor (AI) resulted in improved PFS compared with HERCEPTIN® plus an AI, among HER2-positive and Hormone Receptor positive metastatic breast cancer patients, who had prior HERCEPTIN® based chemotherapy and endocrine therapy in the neo(adjuvant) and/or first line metastatic setting. This dual HER2 blockade combination offers an effective and safe chemotherapy-sparing alternative treatment regimen for this patient population. Phase III, Randomized Study of Dual Human Epidermal Growth Factor Receptor 2 (HER2) Blockade With Lapatinib Plus Trastuzumab in Combination With an Aromatase Inhibitor in Postmenopausal Women With HER2-Positive, Hormone Receptor–Positive Metastatic Breast Cancer: ALTERNATIVE. Johnston SR, Hegg R, Im S, et al. J Clin Oncol 2017;36:741-748
TAFINLAR® and MEKINIST®
The FDA on April 30, 2018 granted regular approval to TAFINLAR® (Dabrafenib) and MEKINIST® (Trametinib), in combination, for the adjuvant treatment of patients with Melanoma with BRAF V600E or V600K mutations, as detected by an FDA-approved test, and involvement of lymph node(s), following complete resection. TAFINLAR® and MEKINIST® are products of Novartis Pharmaceuticals Corp.
TAGRISSO® (Osimertinib)
The FDA on April 19, 2018 approved TAGRISSO® for the first-line treatment of patients with metastatic Non Small Cell Lung Cancer (NSCLC), whose tumors have Epidermal Growth Factor Receptor (EGFR) exon 19 deletions or exon 21 L858R mutations, as detected by an FDA-approved test. TAGRISSO® is a product of AstraZeneca Pharmaceuticals LP.
TAVALISSE® (Fostamatinib disodium hexahydrate)
The FDA on April 17, 2018 approved TAVALISSE® for the treatment of thrombocytopenia in adult patients with chronic Immune Thrombocytopenia (ITP) who have had an insufficient response to a previous treatment. TAVALISSE® is a product of Rigel Pharmaceuticals, Inc.
OPDIVO® (Nivolumab) and YERVOY® (Ipilimumab)
The FDA on April 16, 2018 granted approvals to OPDIVO® and YERVOY® in combination, for the treatment of intermediate or poor risk, previously untreated advanced Renal Cell Carcinoma. OPDIVO® and YERVOY® are products of Bristol-Myers Squibb Co.
FDA Approves TAVALISSE® for Chronic ITP
SUMMARY: The FDA on April 17, 2018, approved TAVALISSE® (Fostamatinib tablets) for the treatment of thrombocytopenia in adult patients with chronic Immune Thrombocytopenic Purpura (ITP), who have had an insufficient response to a previous treatment. ITP can manifest as an acute self limited disease often seen in children (Acute ITP) or in a chronic form (Chronic ITP) seen in adults with the thrombocytopenia lasting for 6 months or longer.
TAVALISSE is an oral Tyrosine Kinase Inhibitor that targets SYK kinase (Spleen Tyrosine Kinase). SYK associates with Fcγ receptors (FcγR) on the cell surface of various inflammatory cells, including macrophages, which are in turn responsible for platelet clearance in ITP. Inhibition of SYK by the active metabolite of TAVALISSE® reduces the destruction of platelets by macrophages, that are activated in the immune system. TAVALISSE® is the first and only SYK inhibitor indicated for adults with Chronic ITP and the present study validated the therapeutic effect of SYK inhibition in an autoimmune disease.
The approval of TAVALISSE® was supported by data from two randomized placebo-controlled phase III trials and an open label extension study, as well as an initial proof-of-concept study. FIT-1 and FIT-2 are two identical, multicenter, randomized, double-blind, placebo-controlled, phase III trials that included 150 patients with persistent or Chronic ITP, who had an insufficient response to previous treatment, which included Corticosteroids, Immunoglobulins, Splenectomy, and/or a Thrombopoietin Receptor Agonist (TPO-RA). Patients were randomized 2:1 to receive either TAVALISSE® 100 mg orally twice daily or placebo for 24 weeks. Dose could be escalated to 150 mg orally twice daily after one month. The median age was 54 years and enrolled patients had three documented platelet counts of 30,000/µL or less. Patients had ITP for a median of 8.5 years prior to enrollment and the median baseline platelet count was 16,000/μL. The most common prior treatments for ITP were Steroids (94%), TPO-RAs (47%), Splenectomy (35%), and Rituximab (32%). The Primary endpoint was Stable platelet response of 50,000/ µL or more on at least 4 of the 6 biweekly visits between Weeks 14 and 24 of the study, without rescue treatment.
The Overall Response Rate with TAVALISSE® in the two trials, FIT-1 and FIT-2 was 29% versus 2% in the placebo group (P<0.001). Among the responders on TAVALISSE®, Stable Response (Primary end point) was noted in 18% versus 1% in the placebo group (P=0.007) and Intermediate Response (defined as at least 2 consecutive biweekly platelet counts of 50,000/μL or more, without rescue treatment) was noted in 11% versus 0% in the placebo group (P<0.001).
Among patients treated with TAVALISSE®, the median platelet counts at 24 weeks of follow up was 95,000/μL for those who had a Stable Response, 49,000/μL for those who had Intermediate Response, and 20,500/μL in non-responders. For those in the placebo group, however, median platelet counts only reached 17,500/μL. More than half of patients treated with TAVALISSE® (54%) had increment in platelet count (20,000/μL or more), compared with 29% of patients receiving placebo (P=0.005). The median time to response (platelet count of 50,000/μL or more) in those treated with TAVALISSE®, was two weeks. Rescue medications (including Platelet transfusions and intravenous Immunoglobulin) were required in 26% of responding patients in the TAVALISSE® group and for 45% of patients in the placebo group. Response rates were not influenced by Age, Sex, baseline platelet count, Splenectomy or prior treatment with a TPO-RA. Among patients who had received and later failed to respond to a TPO-RA before enrollment, 17% had a Stable Response to TAVALISSE®. Serious bleeding was not noted in the 29% of patients who achieved a response, whereas it occurred in 5.6% of non-responders and 10.2% of those receiving placebo.
Patients from FIT-1 and FIT-2 trials were also included in an open label expansion cohort (FIT-3). In this study, 23% of those who received placebo in FIT-1 or FIT-2 had a Stable platelet response to TAVALISSE®. The most common toxicities were rash, fatigue, nausea, diarrhea, abdominal pain, hypertension, liver function abnormalities and neutropenia.
It was concluded that TAVALISSE® is the first and only Spleen Tyrosine Kinase (SYK) inhibitor, and by its unique mechanism of action, is an important alternative for patients with difficult to treat chronic ITP. Bussel J, Mayer J, Cervinek L, et al. Treatment of primary adult chronic immune thrombocytopenia (CITP) with fostamatinib, an oral SYK inhibitor: results of two randomized, placebo-controlled phase 3 studies. Abstract #S435. Presented at the 22nd Congress of the European Hematology Association, June 24, 2017; Madrid, Spain.
FDA Approves TAGRISSO® for First-Line Treatment of Metastatic NSCLC
SUMMARY: The FDA on April 19, 2018, approved TAGRISSO® (Osimertinib) for the first-line treatment of patients with metastatic Non Small Cell Lung Cancer (NSCLC), whose tumors have Epidermal Growth Factor Receptor (EGFR) exon 19 deletions or exon 21 L858R mutations, as detected by an FDA-approved test.
Lung cancer is the second most common cancer in both men and women and accounts for about 14% of all new cancers and 27% of all cancer deaths. The American Cancer Society estimates that for 2018 about 234,030 new cases of lung cancer will be diagnosed and over 154,050 patients will die of the disease. Lung cancer is the leading cause of cancer-related mortality in the United States. Non Small Cell Lung Cancer (NSCLC) accounts for approximately 85% of all lung cancers. Of the three main subtypes of Non Small Cell Lung Cancer (NSCLC), 30% are Squamous Cell Carcinomas (SCC), 40% are Adenocarcinomas and 10% are Large cell carcinomas. With changes in the cigarette composition and decline in tobacco consumption over the past several decades, Adenocarcinoma now is the most frequent histologic subtype of lung cancer. Approximately 10-15% of Caucasian patients and 35-50% of Asian patients with Adenocarcinomas, harbor activating EGFR (Epidermal Growth Factor Receptor) mutations and 90% of these mutations are either exon 19 deletions or L858R point mutations in exon 21.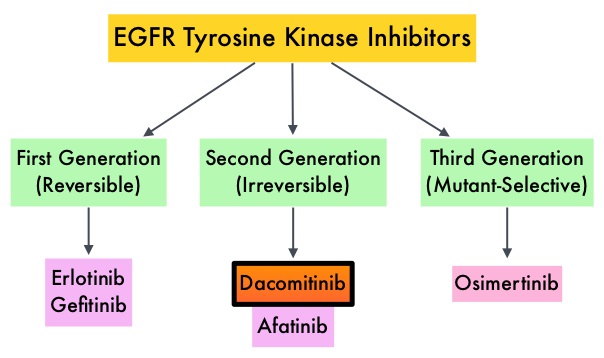 EGFR-Tyrosine Kinase Inhibitors (TKIs) such as TARCEVA® (Erlotinib), IRESSA® (Gefitinib) and GILOTRIF® (Afatinib), have demonstrated a 60-70% response rate as monotherapy when administered as first line treatment, in patients with metastatic NSCLC, who harbor the sensitizing EGFR mutations. However, majority of these patients experience disease progression within 9-14 months. This resistance to frontline EGFR TKI therapy has been attributed to acquired T790M “gatekeeper” point mutation in EGFR, identified in 50-60% of patients. Put another way, T790M is not relevant in about 40% of patients whose disease progression may be related to other mechanisms.
EGFR-Tyrosine Kinase Inhibitors (TKIs) such as TARCEVA® (Erlotinib), IRESSA® (Gefitinib) and GILOTRIF® (Afatinib), have demonstrated a 60-70% response rate as monotherapy when administered as first line treatment, in patients with metastatic NSCLC, who harbor the sensitizing EGFR mutations. However, majority of these patients experience disease progression within 9-14 months. This resistance to frontline EGFR TKI therapy has been attributed to acquired T790M “gatekeeper” point mutation in EGFR, identified in 50-60% of patients. Put another way, T790M is not relevant in about 40% of patients whose disease progression may be related to other mechanisms.
TAGRISSO® is a third-generation Epidermal Growth Factor Receptor (EGFR) TKI presently approved by the FDA, for the treatment of patients with metastatic EGFR T790M mutation-positive NSCLC, who had progressed on prior systemic therapy, including an EGFR-TKI. Previously published studies had suggested that TAGRISSO® may also be effective as initial therapy for EGFR mutation-positive advanced NSCLC.
The recent new indication for TAGRISSO® was based on FLAURA, which is a randomized, double blind, phase III clinical trial, conducted to compare the efficacy and safety of first line TAGRISSO® to TARCEVA® or IRESSA® (which are considered standard first line therapies), in NSCLC patients with activating mutations EGFR exon 19 deletions or L858R substitution mutation on exon 21. This study randomized 556 advanced NSCLC treatment naïve patients, with EGFR exon 19 or 21 mutations in a 1:1 ratio, to TAGRISSO® 80 mg orally once daily (N=279) or Standard of Care EGFR-TKI, IRESSA® 250 mg or TARCEVA® 150 mg, orally once daily (N=277). Patients were stratified by mutation status (exon 19 vs 21 mutations) and race (Asian vs non-Asian). Patients with CNS metastases who were neurologically stable, were allowed in this study. The Primary endpoint was Progression Free Survival (PFS).
The median PFS was 18.9 months with TAGRISSO® compared to 10.2 months for the standard therapy (HR=0.46; P<0.001), suggesting a 54% reduction in the risk of disease progression, compared with Standard of Care. TAGRISSO® extended the median Time To Progression by about 9 months. This PFS benefit was consistent across all subgroups of patients, including those with and without CNS metastases at study entry. The Objective Response Rate (ORR) with TAGRISSO® was 80% compared with 76% for TARCEVA® and IRESSA®. The median Duration of Response with TAGRISSO® was 17.2 months versus 8.5 months in the comparator arm. The median Overall Survival was not reached. Grade 3 and 4 toxicities were lower for TAGRISSO® (34%) compared with 45% for TARCEVA® and IRESSA®. Toxicities led to treatment discontinuation for 13% and 18% of patients in the TAGRISSO® and comparator groups, respectively.
It was concluded that TAGRISSO® demonstrated superior efficacy, with a near doubling in median Progression Free Survival, and better tolerability, compared to the Standard of Care, when given as first-line therapy, for patients with advanced EGFR mutation positive NSCLC. Studies are underway, assessing treatments, following resistance to TAGRISSO®.
Osimertinib in Untreated EGFR-Mutated Advanced Non–Small-Cell Lung Cancer. Soria J-C, Ohe Y, Vansteenkiste J, et al. for the FLAURA Investigators. N Engl J Med 2018; 378:113-125
FDA Approves OPDIVO® plus YERVOY® Combination Immunotherapy for intermediate or Poor-risk Advanced Renal Cell Carcinoma
SUMMARY: The FDA on April 16, 2018, granted approvals to OPDIVO® (Nivolumab) and YERVOY® (Ipilimumab) in combination, for the treatment of intermediate or poor-risk, previously untreated advanced Renal Cell Carcinoma (RCC). SUTENT® (Sunitinib) is a MultiKinase Inhibitor (MKI) which simultaneously targets the tumor cell wall, vascular endothelial cell wall as well as the pericyte/fibroblast/vascular/ smooth vessel cell wall and is capable of specifically binding to tyrosine kinases, inhibiting the earlier signaling events and thereby inhibits phosphorylation of VEGF receptor, PDGF receptor, FLT-3 and c-KIT. SUTENT® is the standard first-line intervention for treatment naïve patients with advanced Renal Cell Carcinoma. In a large, multi-center, randomized, phase III study, the median Progression Free Survival (PFS) with SUTENT® was 9.5 months, the Objective Response Rate (ORR) was 25%, and the median Overall Survival was 29.3 months, when compared with Interferon Alfa, in patients with treatment-naïve Renal Cell Carcinoma. This was however associated with a high rate of hematological toxicities.
OPDIVO® is a fully human, immunoglobulin G4 monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, whereas YERVOY® is a fully human immunoglobulin G1 monoclonal antibody that blocks Immune checkpoint protein/receptor CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4, also known as CD152). Blocking the Immune checkpoint proteins unleashes the T cells, resulting in T cell proliferation, activation and a therapeutic response. OPDIVO® was approved by the FDA in November 2015, for the treatment of advanced RCC in patients who had received prior anti-angiogenic therapy, based on an Overall Survival benefit. YERVOY® is approved for the treatment of metastatic melanoma. Combining OPDIVO® with YERVOY® (Combination immunotherapy) has shown promising efficacy in multiple tumor types, including advanced RCC, with higher Objective Response Rate than either agent alone, and is presently approved for the treatment of advanced malignant melanoma.
This FDA approval was based on CheckMate 214, a randomized open-label phase III trial in which a combination of OPDIVO® plus YERVOY® (N=550) was compared with SUTENT® (N=546), among treatment naïve, clear-cell, advanced Renal Cell Carcinoma (RCC) patients. The authors randomly assigned 1096 patients in a 1:1 ratio to receive OPDIVO® 3 mg/kg IV plus YERVOY® 1 mg/kg IV every 3 weeks for four doses (induction phase) followed by OPDIVO® monotherapy at 3 mg/kg every 2 weeks (maintenance phase) or SUTENT® 50 mg orally once daily for 4 weeks, of each 6-week cycle. Four hundred and twenty five (425) patients in the combination group and 422 patients in the SUTENT® group had intermediate or poor-risk patients. It is estimated that approximately 75% of patients with advanced RCC have intermediate or poor-risk disease and have worse outcomes than those with favorable-risk disease. The coprimary end points were Overall Survival, Objective Response Rate and Progression Free Survival among patients with intermediate or poor prognostic risk disease.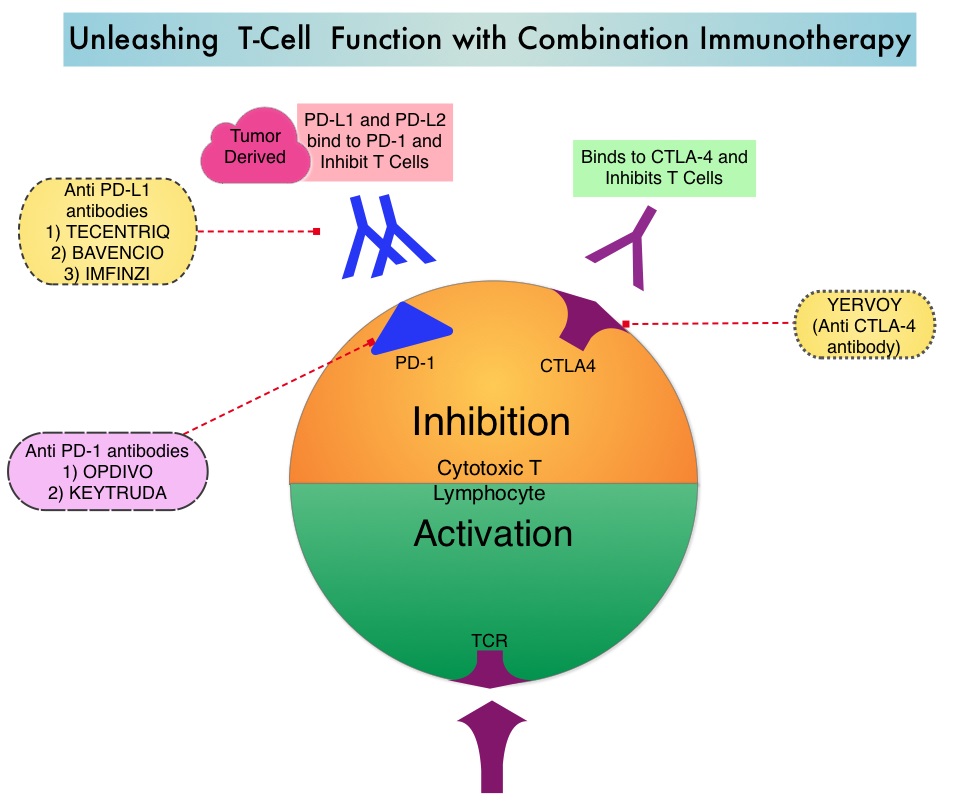
At a median follow-up of 25.2 months, the combination of OPDIVO® and YERVOY® had a significant Overall Survival benefit over SUTENT®. The 18-month Overall Survival rate was 75% with combination immunotherapy and 60% with SUTENT®. The median Overall Survival was not reached with combination immunotherapy versus 26.0 months with SUTENT® (HR=0.63; P<0.001). The Objective Response Rate was 42% with combination immunotherapy versus 27% with SUTENT® (P<0.001), and the Complete Response rate was 9% versus 1% respectively. The median Progression Free Survival was 11.6 months and 8.4 months, respectively but this was not statistically significant per the prespecified threshold. The benefit with combination immunotherapy was not noted in patients with favorable-risk disease. The superior outcomes with combination immunotherapy in patients with intermediate and poor-risk RCC may very well be related to a higher tumor mutational load in this group of patients, compared to those with favorable-risk disease.
In exploratory analyses among 776 intermediate and poor-risk patients, who had quantifiable PD-L1 expression in this study, Overall Survival was longer with Immunotherapy combination compared with SUTENT®, across PD-L1 expression levels. In patients with PD-L1 expression of 1% or greater, the 18-month Overall Survival rate was 81% with combination immunotherapy and 53% with SUTENT®, and the median Overall Survival was not reached versus 19.6 months respectively (HR=0.45). Among patients with PD-L1 expression of 1% or greater, the Objective Response Rate was 58% versus 22% for SUTENT® (P<0.001), the median PFS was 22.8 and 5.9 months, respectively (HR=0.46). A similar trend was noted in patients with PD-L1 expression 5% or greater, as compared with patients with less than 5% PD-L1 expression. Treatment discontinuation, related to adverse events occurred in 22% of the patients in the combination immunotherapy group and 12% in the SUTENT® group.
It was concluded that treatment with a combination of OPDIVO® and YERVOY® resulted in a significantly higher Overall Survival and Objective Response Rates, compared with SUTENT®, among intermediate and poor-risk, previously untreated patients, with advanced Renal Cell Carcinoma. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. Motzer RJ, Tannir NM, McDermott DF, et al. N Engl J Med 2018; 378:1277-1290
