The FDA on June 13, 2018 approved AVASTIN® for patients with epithelial ovarian, fallopian tube, or primary peritoneal cancer, in combination with Carboplatin and Paclitaxel, followed by single-agent AVASTIN®, for stage III or IV disease, after initial surgical resection. AVASTIN® is a product of Genentech, Inc.
Tag: Ovarian Cancer
RUBRACA® (Rucaparib)
The FDA on April 6, 2018 approved RUBRACA®, a Poly ADP-Ribose Polymerase (PARP) inhibitor, for the maintenance treatment of recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a Complete or Partial Response to platinum-based chemotherapy. RUBRACA® is a product of Clovis Oncology Inc.
FDA Approves RUBRACA® for Maintenance Treatment of Recurrent Ovarian Cancer
SUMMARY: The FDA on April 6, 2018, approved RUBRACA® (Rucaparib), a Poly ADP-Ribose Polymerase (PARP) inhibitor, for the maintenance treatment of recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a Complete or Partial Response to platinum-based chemotherapy. RUBRACA® was initially approved in December 2016 as monotherapy for the treatment of patients with deleterious BRCA mutation (germline and/or somatic) associated advanced ovarian cancer, who have been treated with two or more chemotherapies.
RUBRACA® is an oral, small molecule inhibitor of Poly-Adenosine diphosphate [ADP] Ribose Polymerase (PARP), developed for treatment of ovarian cancer, associated with Homologous Recombination DNA repair deficiency (HRD). Previously published clinical data had suggested that ovarian cancer patients with and without evidence of a germline BRCA mutation, benefit from treatment with a PARP inhibitor, and that maintenance treatment with a PARP inhibitor following a response to platinum-based treatment increases Progression Free Survival (PFS), in patients with ovarian cancer. Even though patients with or without BRCA mutation benefited, the most benefit was derived in those with BRCA mutation.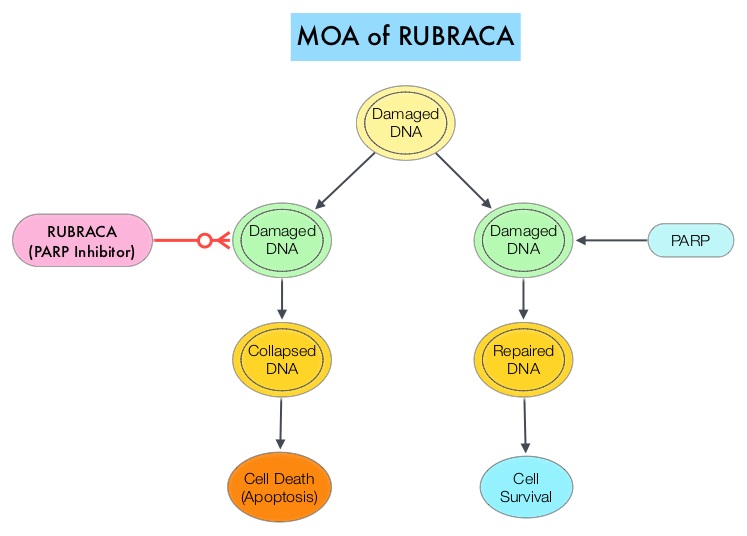
The approval of RUBRACA® was based on ARIEL3, a randomized, double-blind, placebo-controlled, phase III trial, which evaluated the benefit of RUBRACA® versus placebo, after response to second-line or later platinum-based chemotherapy, in patients with high-grade, recurrent, platinum-sensitive ovarian carcinoma. In this trial, 561 patients were randomly assigned in a 2:1 ratio to receive RUBRACA® 600 mg orally twice daily (N=372) or placebo (N=189). Treatment was continued until disease progression or unacceptable toxicity. Eligible patients had recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, and had been treated with at least two prior treatments of platinum-based chemotherapy, and were in Complete or Partial Response to the most recent platinum-based chemotherapy. Patients had CA-125 level of less than the upper limit of normal. Using Next-Generation Sequencing assay, tumor tissue was examined to determine whether DNA contained a deleterious somatic or germline BRCA mutation (tBRCA), in addition to determining the percentage of genomic Loss of Heterozygosity (LOH). Positive Homologous Recombination Deficiency (HRD) status was defined as tBRCA-positive and/or LOH high. The Primary end point was Progression Free Survival in three patient cohorts – all patients, HRD subgroup, and tumor BRCA subgroup.
It was noted that there was a statistically significant improvement in median Progression Free Survival (PFS) for all patients assigned to RUBRACA®, compared with placebo (median PFS 10.8 versus 5.4 months, HR=0.36; P<0.0001). In the HRD subgroup, the median PFS was 13.6 months for those assigned to RUBRACA®, versus 5.4 months for the placebo group (HR=0.32; P<0.0001), and in the tumor BRCA subgroup, the median PFS was 16.6 versus 5.4 months (HR=0.23; P <0.0001), respectively. The most common adverse reactions were fatigue, rash, nausea, vomiting, diarrhea, abdominal discomfort, cytopenias and abnormal liver function studies. Discontinuation due to adverse reactions occurred in 15% of patients receiving RUBRACA®.
It was concluded that RUBRACA® significantly improved Progression Free Survival in patients with platinum-sensitive ovarian cancer who had achieved a response to platinum-based chemotherapy, and could be considered a new standard of care for women with platinum-sensitive ovarian cancer, following a complete or partial response to second-line or later lines of platinum-based chemotherapy. The FDA also concurrently approved the complementary diagnostic test, FoundationFocusTM CDx BRCA LOH for tumor samples, to determine HRD status.
Rucaparib maintenance treatment for recurrent ovarian carcinoma after response to platinum therapy (ARIEL3): a randomised, double-blind, placebo-controlled, phase 3 trial. Coleman RL, Oza AM, Lorusso D, et al. The Lancet 2017;390:1949-1961
LYNPARZA® (Olaparib)
The FDA on August 17, 2017 granted regular approval to LYNPARZA® tablets for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. LYNPARZA® is a product of AstraZeneca.
ZEJULA® (Niraparib)
The FDA on March 27, 2017 approved ZEJULA®, a poly ADP-ribose polymerase (PARP) inhibitor, for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in complete or partial response to platinum-based chemotherapy. ZEJULA® is marketed by Tesaro, Inc.
Niraparib Maintenance Therapy Significantly Improves Progression Free Survival in Platinum-Sensitive Ovarian Cancer
SUMMARY: It is estimated that in the United States, approximately 22,440 women will be diagnosed with ovarian cancer in 2017 and a little over 14,000 women will die of the disease. Ovarian cancer ranks fifth in cancer deaths among women, and accounts for more deaths than any other cancer of the female reproductive system. Approximately 75% of the ovarian cancer patients are diagnosed with advanced disease and the 5 year Overall Survival rate is about 20-30%. These patients are often treated with platinum based chemotherapy following primary surgical cytoreduction.
BRCA1 and BRCA2 are tumor suppressor genes and functional BRCA proteins that repair damaged DNA and play an important role in maintaining cellular genetic integrity. They regulate cell growth and prevent abnormal cell division and development of malignancy. Mutations in BRCA1 and BRCA2 account for about 20 to 25 percent of hereditary breast cancers and about 5 to 10 percent of all breast cancers. They also account for 15 percent of ovarian cancers in addition to other cancers such as colon and prostate.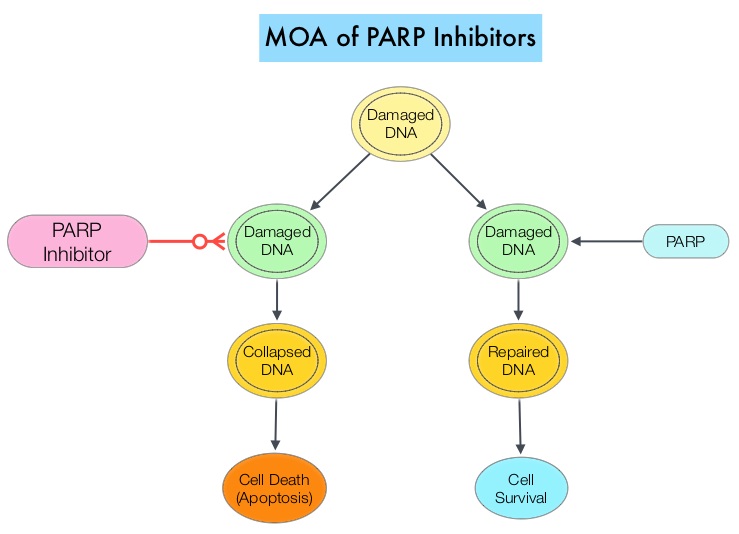
Homologous Recombination (HR) is an important pathway that allows repair of double-stranded DNA breaks and operates during the S and G2 phases of the cell cycle, relying on several proteins including BRCA1 and BRCA2. Deficiency of BRCA1 and BRCA2 results in non-functioning HR pathway (HR Deficiency), and other pathways then come in to play, which are less precise and error prone, resulting in the accumulation of additional mutations and chromosomal instability in the cell, with subsequent malignant transformation. Hereditary Epithelial Ovarian Cancer was thought to be caused almost exclusively by mutations in BRCA1 and BRCA2. It however now appears that about 50% of the high grade serous ovarian cancers have aberrations in HR repair pathway. BRCA mutations can either be inherited (germline) and present in all individual cells or can be acquired and occur exclusively in the tumor cells (somatic). Somatic mutations account for a significant portion of overall BRCA1 and BRCA2 aberrations, and loss of BRCA function due to frequent somatic aberrations in ovarian cancers likely deregulates HR pathway and increases sensitivity to platinum drugs. Majority of the women with germline BRCA mutations (gBRCA) are positive for HR deficiency.
The PARP (Poly ADP Ribose Polymerase) family of enzymes which include PARP1 and PARP2, repair damaged DNA. PARP inhibitors kill tumors defective in the BRCA1 or BRCA2 genes through the concept of synthetic lethality. Epithelial Ovarian Cancers with Homologous Recombination Deficiency (HRD) have demonstrated sensitivity to PARP inhibitors. The two currently FDA approved PARP inhibitors include LYNPARZA® (Olaparib) for the treatment of ovarian cancer with gBRCA mutations after three lines of therapy and RUBRACA® (Rucaparib) for the treatment of ovarian cancer with gBRCA mutations and /or somatic mutations after two lines of therapy.
Niraparib is a highly selective PARP 1/2 inhibitor, that detects DNA damage and promotes its repair. Previously published studies demonstrated the antitumor activity of Niraparib in patients with ovarian cancer, at a maximum dose of 300 mg per day, with a low frequency of high grade adverse events. Based on this preliminary data, the authors conducted a randomized, placebo-controlled, phase III trial (ENGOT-OV16/NOVA) to evaluate the efficacy and safety of Niraparib versus placebo, as maintenance treatment in patients with platinum-sensitive, recurrent ovarian cancer.
This study enrolled two independent cohort of patients based on the presence or absence of a germline BRCA mutation (gBRCA cohort and non-gBRCA cohort), as determined on BRACAnalysis® testing (Myriad Genetics). All enrolled patients had tumors sensitive to platinum-based therapy and had received at least 2 lines of therapy. Enrolled patients (N=553) were randomly assigned in a 2:1 ratio to receive Niraparib 300 mg or placebo once daily. The gBRCA cohort included 203 patients (138 assigned to Niraparib and 65 to placebo) and the non-gBRCA cohort included 350 patients (234 assigned to Niraparib and 116 to placebo). The Primary end point was Progression Free Survival (PFS) and Secondary end points included chemotherapy-free interval, time to first subsequent therapy, Overall Survival and safety.
It was noted that patients in the Niraparib group had a significantly longer Progression Free Survival compared to those in the placebo group. In the gBRCA cohort, the PFS with Niraparib compared to placebo was 21.0 vs. 5.5 months (HR=0.27), in the non-gBRCA cohort for patients who had tumors with Homologous Recombination Deficiency (HRD), the PFS was 12.9 months vs. 3.8 months (HR=0.38) and for the overall non-gBRCA cohort, the PFS was 9.3 months vs. 3.9 months (HR=0.45). The P value was significant for all three comparisons (P<0.001). The most common grade 3 or 4 toxicities in the Niraparib group were thrombocytopenia (34%), anemia (25%), and neutropenia (20%), and this was managed with dose modifications.
The authors concluded that among patients with platinum-sensitive, recurrent ovarian cancer, Niraparib significantly prolonged Progression Free Survival compared to placebo and this benefit was achieved regardless of the presence or absence of gBRCA mutations or HRD status, with acceptable toxicities. Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. Mirza MR, Monk BJ, Herrstedt J, et al. N Engl J Med 375:2154-2164, 2016
The FDA recommends against using screening tests for ovarian cancer screening FDA Safety Communication
SUMMARY: The American Cancer Society estimates that about 22,440 women will be diagnosed with ovarian cancer in the United States for 2017 and about 14,080 will die of the disease. Ovarian cancer ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. Women who have reached menopause, women who have a family history of ovarian cancer, and women with the BRCA1 or BRCA2 genetic mutations have the highest risk for developing ovarian cancer. Over 75% of patients with ovarian cancer have advanced disease at the time of diagnosis. The FDA Safety Communication noted that despite extensive research and published studies there are currently no screening tests for ovarian cancer that are sensitive enough to reliably screen for ovarian cancer without a high number of inaccurate results.
The 2 tests used most often to screen for ovarian cancer are TransVaginal UltraSound (TVUS) and serum marker CA-125. TVUS is unable to differentiate benign from malignant ovarian mass. Serum marker CA-125 is usually associated with high-grade serous ovarian cancer, but is also expressed in normal tissues of the body such as the lungs and other reproductive organs. An increase in the serum marker CA-125 can be seen in non-malignant conditions such as endometriosis, peritonitis and in women with uterine fibroids. Even though serum marker CA-125 when elevated in patients, with an established diagnosis of ovarian cancer, is often used to follow the course of the disease, it has never been proven as an effective screening test for the early detection of ovarian cancer.
Nonetheless, numerous companies continue to claim that their commercially available diagnostic tests can effectively screen and detect ovarian cancer, with no data to support their claims. The FDA is concerned that women and their health care providers may rely on these inaccurate test results to make treatment decisions. Women with a false-positive result may undergo additional medical tests and/or unnecessary surgery, and may experience complications related to both. Conversely, women with a false-negative test may delay or not seek surgery or other treatment interventions for ovarian cancer. The later is particularly relevant for patients with BRCA mutations. Approximately 40% of BRCA1-mutation carriers and 18% of BRCA2-mutation carriers will develop ovarian cancer by age 70. It is recommended that patients who have BRCA1 mutations consider risk-reduction surgery (hysterectomy and bilateral bilateral salpingo-oophorectomy) by age 40 and those with BRCA2-mutations consider risk-reduction surgery no later than age 50.
Even though screenings for breast, colon and cervical cancers are successfully used for early detection and prevention of cancer-related deaths, a screening test for ovarian cancer with valid scientific data presently does not exist, and the FDA recommends against using currently offered tests to screen for ovarian cancer.
Recommendations:
For women, including those at increased risk of developing ovarian cancer
• Be aware that there is currently no safe and effective ovarian cancer screening test
• Do not rely on ovarian cancer screening test results to make health or treatment decisions
• Talk to your doctor about ways to reduce your risk of developing ovarian cancer, especially if you have a family history of ovarian cancer, or have the BRCA1 or BRCA2 genetic mutations
For physicians
• Do not recommend or use tests that claim to screen for ovarian cancer in the general population of women
• Be aware that testing higher risk asymptomatic patients for ovarian cancer has no proven benefit and is not a substitute for preventive actions that may reduce their risk
• Consider referring women at high risk of developing ovarian cancer, including those with BRCA mutations, to a genetic counselor or gynecologic oncologist, or other appropriate health care provider for more specialized care
U.S. Food and Drug Administration: The FDA recommends against using screening tests for ovarian cancer screening: FDA Safety Communication. Issued September 7, 2016. Available at http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm519413.htm
RUBRACA® (Rucaparib)
The FDA on November 19, 2016 granted accelerated approval to RUBRACA® for treatment of patients with deleterious BRCA mutation (germline and/or somatic) associated advanced ovarian cancer, who have been treated with two or more chemotherapies. RUBRACA® is a product of Clovis Oncology, Inc.
FDA Approves AVASTIN® for Platinum-Sensitive Ovarian Cancer
SUMMARY: The FDA on December 7, 2016 approved AVASTIN® (Bevacizumab), either in combination with Carboplatin and Paclitaxel or in combination with Carboplatin and Gemcitabine chemotherapy, followed by AVASTIN® alone, for the treatment of patients with platinum-sensitive recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer. Platinum-sensitive disease is defined as relapse occurring six months or longer following the last treatment with a platinum-based chemotherapy. The American Cancer Society estimates that over 22,280 women will be diagnosed with ovarian cancer in the United States for 2016 and over 14,240 will die of the disease. Ovarian cancer ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. The FDA had approved AVASTIN® in combination with Paclitaxel, Pegylated Liposomal Doxorubicin, or Topotecan in 2014, for the treatment of patients with Platinum-resistant, recurrent epithelial Ovarian, Fallopian tube, or Primary Peritoneal cancer.
The present approval was based on results from two randomized, controlled Phase III studies, GOG-0213 and OCEANS trial. GOG-0213 is phase III study and was conducted by the Gynecologic Oncology Group (GOG) that enrolled 673 women with platinum-sensitive recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer. This study enrolled 673 patients with predominantly serous adenocarcinoma histology. The patients were randomly assigned to receive combination chemotherapy with Paclitaxel and Carboplatin (N=336) or the same chemotherapy along with Avastin 15 mg/kg IV every 3 weeks (N=337), followed by AVASTIN® maintenance. The median age was 60 years. The Primary endpoint of this study was Overall Survival (OS) and Secondary endpoints included Progression Free Survival (PFS) and Objective Response Rate (ORR).
There was a 5 month improvement in the median Overall Survival with the addition of AVASTIN® to chemotherapy compared with chemotherapy alone (42.6 months vs 37.3 months, respectively; HR=0.84). There was a 3.4 improvement in the median PFS in the AVASTIN® group compared to chemotherapy alone (13.8 months vs 10.4 months, respectively; HR=0.61). The Objective Response Rate (ORR) was 78% with the addition of AVASTIN® to chemotherapy versus 56% with chemotherapy alone.
OCEANS trial is a placebo-controlled, randomized, multicentre Phase III study that evaluated the safety and efficacy of AVASTIN® in combination with Carboplatin and Gemcitabine chemotherapy. This study included 484 patients with platinum-sensitive recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who were randomly assigned to receive combination chemotherapy with Carboplatin and Gemcitabine along with placebo (N=242) or combination chemotherapy along with Avastin (N=242). The Primary endpoint of the study was Progression Free Survival and Secondary endpoints included Objective Response Rate, Overall Survival and safety.
The addition of AVASTIN® to chemotherapy significantly improved PFS compared to chemotherapy alone (12.4 months vs. 8.4 months; HR=0.46, P<0.0001). The ORR was 78% in the AVASTIN® group compared with 57% in the chemotherapy alone group.
These studies supports the use of AVASTIN® either in combination with Carboplatin and Paclitaxel or in combination with Carboplatin and Gemcitabine chemotherapy, followed by AVASTIN® alone, for patients with platinum-sensitive recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer. A phase III randomized controlled clinical trial of carboplatin and paclitaxel alone or in combination with bevacizumab followed by bevacizumab and secondary cytoreductive surgery in platinum-sensitive, recurrent ovarian, peritoneal primary and fallopian tube cancer (Gynecologic Oncology Group 0213). Coleman RL , Bradya MF, Herzog TJ, et al. Scientific Plenary (Late-Breaking Abstract). SGO 2015. Abstract 3. Presented March 28, 2015
Neoadjuvant Chemotherapy in Advanced Ovarian Cancer – ASCO Clinical Practice Guideline
SUMMARY: The American Cancer Society estimates that over 22,280 women will be diagnosed with ovarian cancer in the United States for 2016 and over 14,240 will die of the disease. Ovarian cancer ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. For the past 40 years, primary cytoreductive surgery followed by chemotherapy has been the standard approach, for women with advanced epithelial ovarian cancer. The benefit of neoadjuvant chemotherapy followed by interval debulking surgery was recognized in the early 1990’s, especially when treating those with advanced age, stage and associated comorbidities. An Expert Panel from the Society of Gynecologic Oncology and the American Society of Clinical Oncology conducted a systematic review of the literature and the primary evidence for these recommendations is based on four phase III clinical trials. The following recommendations are meant to provide guidance to Health Care Providers regarding the use of neoadjuvant chemotherapy and interval cytoreduction among women with stage IIIC or IV epithelial ovarian cancer. The following clinical questions were addressed:
What clinical evaluations should be performed in all women with suspected or newly diagnosed stage IIIC or IV epithelial ovarian cancer?
Recommendation 1.1. All women with suspected stage IIIC or IV invasive epithelial ovarian cancer should be evaluated by a gynecologic oncologist prior to initiation of therapy to determine whether they are candidates for primary cytoreductive surgery.
Recommendation 1.2. A primary clinical evaluation should include a CT scan of the abdomen and pelvis with Oral and IV contrast and chest imaging (CT preferred), to evaluate the extent of disease and feasibility of surgical resection. The use of other tools to refine this assessment may include laparoscopic evaluation or additional radiographic imaging such as PET scan or MRI.
Which patient and disease factors should be used as criteria for identifying patients who are not suitable for primary cytoreductive surgery?
Recommendation 2.1. Women who have a high perioperative risk profile, or a low likelihood of achieving cytoreduction to less than 1 cm, ideally to no visible disease, should receive neoadjuvant chemotherapy.
Recommendation 2.2. Decisions that women are not eligible for medical or surgical cancer treatment, should be made after consultation with a gynecologic oncologist and/or a medical oncologist with gynecologic expertise.
How do neoadjuvant chemotherapy and primary cytoreductive surgery compare with respect to progression-free survival, overall survival, and perioperative morbidity and mortality in women who are fit for primary cytoreduction and have potentially resectable disease, and how should this information be used to select initial treatment?
Recommendation 3.1. For women who are fit for primary cytoreductive surgery, with potentially resectable disease, either neoadjuvant chemotherapy or primary cytoreductive surgery may be offered, based on data from phase III randomized, controlled trials that demonstrate neoadjuvant chemotherapy is noninferior to primary cytoreductive surgery with respect to Progression Free and Overall Survival. Neoadjuvant chemotherapy is associated with less peri- and postoperative morbidity and mortality and shorter hospitalizations, but primary cytoreductive surgery may offer superior survival in selected patients.
Recommendation 3.2. For women with a high likelihood of achieving cytoreduction to less than 1 cm (ideally to no visible disease) and with acceptable morbidity, primary cytoreductive surgery is recommended over neoadjuvant chemotherapy.
Recommendation 3.3. For women who are fit for primary cytoreductive surgery but are deemed unlikely to have cytoreduction to less than 1 cm (ideally to no visible disease) by a gynecologic oncologist, neoadjuvant chemotherapy is recommended over primary cytoreductive surgery. Neoadjuvant chemotherapy is associated with less peri and postoperative morbidity and mortality and shorter hospitalizations.
What additional clinical evaluations should be performed in women with suspected or newly diagnosed stage IIIC or IV epithelial ovarian cancer before neoadjuvant chemotherapy is delivered?
Recommendation 4. Before neoadjuvant chemotherapy is delivered, all patients should have histologic confirmation (core biopsy preferred) of an invasive ovarian, fallopian tube, or peritoneal cancer. In exceptional cases, when a biopsy cannot be performed, cytologic evaluation combined with a serum CA-125 to carcinoembryonic antigen (CEA) ratio more than 25 is acceptable, to confirm the primary diagnosis and exclude a non-gynecologic cancer.
What is the preferred chemotherapy regimen for women with stage IIIC or IV epithelial ovarian cancer who will receive neoadjuvant chemotherapy?
Recommendation 5. For neoadjuvant chemotherapy, a Platinum-Taxane doublet is recommended. However, alternative regimens, containing a Platinum agent, may be selected based on individual patient factors.
Among women treated with neoadjuvant chemotherapy, does the timing of interval cytoreduction or the number of chemotherapy cycles after interval cytoreduction affect the safety or efficacy of treatment?
Recommendation 6. Randomized, controlled trials tested surgery following three or four cycles of chemotherapy in women who had a response to neoadjuvant chemotherapy or stable disease. Interval cytoreductive surgery should be performed after up to four cycles of neoadjuvant chemotherapy for women with a response to chemotherapy or stable disease. Alternative timing of surgery has not been prospectively evaluated but may be considered based on patient-centered factors.
What are the treatment options for patients with progressive disease on neoadjuvant chemotherapy?
Recommendation 7. Patients with progressive disease on neoadjuvant chemotherapy have a poor prognosis. Options include alternative chemotherapy regimens, clinical trials, and/or discontinuation of active cancer therapy and initiation of end-of-life care. In general, there is little role for surgery, and it is not typically advised, unless for palliation such as relief of bowel obstruction. Neoadjuvant Chemotherapy for Newly Diagnosed, Advanced Ovarian Cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. Wright AA, Bohlke K, Armstrong DK, et al. Journal of Clinical Oncology 2016;34:3460-3473
