The FDA on November 9, 2017, granted regular approval to SPRYCEL®, for the treatment of pediatric patients with Philadelphia chromosome-positive (Ph+) Chronic Myeloid Leukemia (CML) in the Chronic Phase. SPRYCEL® is a product of Bristol-Myers Squibb Co.
Tag: Chronic Myeloid Leukemia
ASH – 2016 Discontinuing Tyrosine Kinase Inhibitors is Feasible in Some Patients with CML
SUMMARY: Chronic Myeloid Leukemia (CML) constitutes a little over 10% of all new cases of leukemia. The American Cancer Society estimates that about 8,220 new CML cases will be diagnosed in the United States in 2016 and about 1,070 patients will die of the disease. The hallmark of CML, the Philadelphia Chromosome (Chromosome 22), is a result of a reciprocal translocation between chromosomes 9 and 22, wherein the ABL gene from chromosome 9 fuses with the BCR gene on chromosome 22. As a result, the auto inhibitory function of the ABL gene is lost and the BCR-ABL fusion gene is activated resulting in cell proliferation and leukemic transformation of hematopoietic stem cells. The presently available Tyrosine Kinase Inhibitors (TKI’s) approved in the United States including GLEEVEC® (Imatinib), share the same therapeutic target, which is BCR-ABL kinase. Resistance to TKI’s can occur as a result of mutations in the BCR-ABL kinase domain or amplification of the BCR-ABL gene. With the availability of newer therapies for CML, monitoring response to treatment is important. This is best accomplished by measuring the amount of residual disease using Reverse Transcription-Polymerase Chain Reaction (RT-PCR). Molecular response in CML is expressed using the International Scale (IS) as BCR-ABL%, which is the ratio between BCR-ABL and a control gene. BCR-ABL kinase domain point mutations are detected, using the mutational analysis by Sanger sequencing. Majority of the patients receiving a TKI following diagnosis of CML achieve a Complete Cytogenetic Response (CCyR) within 12 months following commencement of therapy and these patients have a life expectancy similar to that of their healthy counterparts. Previously published studies have shown that Deep Molecular Response (BCR-ABL <0.01% on the International Scale – MR4) is a new molecular predictor of long term survival in CML patients and this was achieved in a majority of patients treated with optimized dose of GLEEVEC®. It has been hypothesized based on previous observations, that a subgroup of CML patients experiencing deeper responses (MR3, MR4, and MR4.5), may stay in unmaintained remission even after treatment discontinuation. Despite this observation, stopping CML therapy is currently not a clinical recommendation and should only be considered in the context of a clinical trial.
The European Stop TKI (EURO-SKI) trial was conducted to assess the safety of stopping Tyrosine Kinase Inhibitor therapy in patients with CML, whose leukemia was in Deep Molecular Response (DMR). This trial enrolled 821 patients with chronic phase CML without prior TKI failure, in DMR (BCR-ABL <0.01% on the International Scale – MR4) for at least one year, following treatment with either Imatinib, Nilotinib or Dasatinib. Following cessation of treatment with TKIs, patients were followed up testing by RQ-PCR (Real-time Quantitative Polymerase Chain Reaction) every 4 weeks for the first 6 months followed by every 6 weeks, the first year and every 3 months thereafter. Molecular recurrence was defined by the loss of the Major Molecular Response (BCR-ABL <0.1% IS – MR3) at any one point.
It was noted that after stopping TKI therapy, 62% showed no evidence of molecular recurrence at 6 months, and 52% showed no recurrence at 24 months. Patients who had taken a TKI for more than 5.8 years before stopping, were significantly less likely to experience relapse within the first 6 months and had a Molecular Relapse Free Survival at 6 months of 65.5% compared with 42.6% for those on treatment for 5.8 years or less. Further, each additional year of TKI therapy increased a patient’s chances of maintaining Major Molecular Response successfully at 6 months by 16%, after TKI therapy was discontinued. Most of the patients who experienced molecular recurrence were able to regain their previous remission level, after resuming TKI therapy and none of the patients in the study had progression to advanced stage.
The authors concluded that stopping TKI therapy of CML patients appeared safe and feasible in over 50% of the patients and longer duration of therapy with TKIs (5.8 years or more) prior to stopping therapy with TKIs, was associated with a higher probability of Molecular Recurrence Free Survival. Cessation of Tyrosine Kinase Inhibitors Treatment in Chronic Myeloid Leukemia Patients with Deep Molecular Response: Results of the Euro-Ski Trial. Mahon F-X, Richter J, Guilhot J, et al. 58th ASH Annual Meeting and Exposition; San Diego, California; December 2-6, 2016. Abstract 787.
TASIGNA® Maintains Durable Deep Molecular Responses Even after Treatment Discontinuation in Chronic Phase CML
SUMMARY: Chronic Myeloid Leukemia (CML) constitutes a little over 10% of all new cases of leukemia. The American Cancer Society estimates that about 8,220 new CML cases will be diagnosed in the United States in 2016 and about 1,070 patients will die of the disease. The hallmark of CML, the Philadelphia Chromosome (Chromosome 22), is a result of a reciprocal translocation between chromosomes 9 and 22, wherein the ABL gene from chromosome 9 fuses with the BCR gene on chromosome 22. As a result, the auto inhibitory function of the ABL gene is lost and the BCR-ABL fusion gene is activated resulting in cell proliferation and leukemic transformation of hematopoietic stem cells.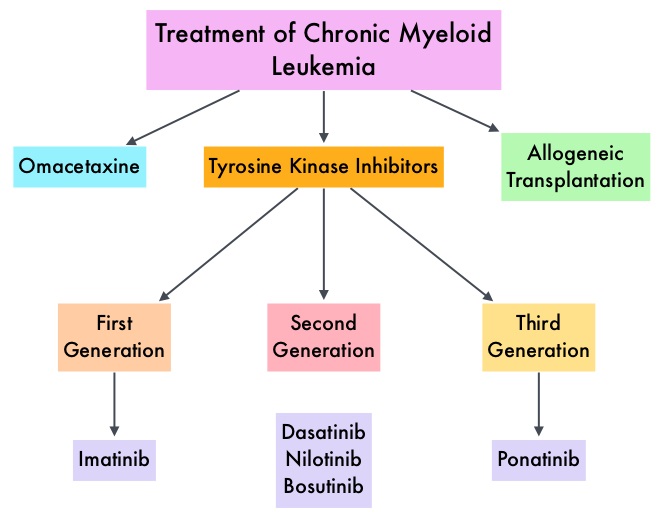 The presently available Tyrosine Kinase Inhibitors (TKI’s) approved in the United States including GLEEVEC®, share the same therapeutic target, which is BCR-ABL kinase. Resistance to TKI’s can occur as a result of mutations in the BCR-ABL kinase domain or amplification of the BCR-ABL gene. With the availability of newer therapies for CML, monitoring response to treatment is important. This is best accomplished by measuring the amount of residual disease using Reverse Transcription-Polymerase Chain Reaction (RT-PCR). Molecular response in CML is expressed using the International Scale (IS) as BCR-ABL%, which is the ratio between BCR-ABL and a control gene. BCR-ABL kinase domain point mutations are detected, using the mutational analysis by Sanger sequencing. Majority of the patients receiving a TKI following diagnosis of CML achieve a Complete Cytogenetic Response (CCyR) within 12 months following commencement of therapy and these patients have a life expectancy similar to that of their healthy counterparts. Previously published studies have shown that deep molecular response (MR4.5) is a new molecular predictor of long term survival in CML patients and was achieved in a majority of patients treated with optimized dose of GLEEVEC®. It has been hypothesized based on previous observations, that a subgroup of CML patients experiencing deeper responses (MR3, MR4, and MR4.5), may stay in unmaintained remission even after treatment discontinuation. Despite this observation, stopping CML therapy is currently not a clinical recommendation and should only be considered in the context of a clinical trial.
The presently available Tyrosine Kinase Inhibitors (TKI’s) approved in the United States including GLEEVEC®, share the same therapeutic target, which is BCR-ABL kinase. Resistance to TKI’s can occur as a result of mutations in the BCR-ABL kinase domain or amplification of the BCR-ABL gene. With the availability of newer therapies for CML, monitoring response to treatment is important. This is best accomplished by measuring the amount of residual disease using Reverse Transcription-Polymerase Chain Reaction (RT-PCR). Molecular response in CML is expressed using the International Scale (IS) as BCR-ABL%, which is the ratio between BCR-ABL and a control gene. BCR-ABL kinase domain point mutations are detected, using the mutational analysis by Sanger sequencing. Majority of the patients receiving a TKI following diagnosis of CML achieve a Complete Cytogenetic Response (CCyR) within 12 months following commencement of therapy and these patients have a life expectancy similar to that of their healthy counterparts. Previously published studies have shown that deep molecular response (MR4.5) is a new molecular predictor of long term survival in CML patients and was achieved in a majority of patients treated with optimized dose of GLEEVEC®. It has been hypothesized based on previous observations, that a subgroup of CML patients experiencing deeper responses (MR3, MR4, and MR4.5), may stay in unmaintained remission even after treatment discontinuation. Despite this observation, stopping CML therapy is currently not a clinical recommendation and should only be considered in the context of a clinical trial.
ENESTfreedom (Evaluating Nilotinib Efficacy and Safety in Clinical Trial – Following REsponsE in De nOvo CML-CP Patients) is a single arm, open-label, international, multicenter phase II study, which evaluated Treatment-Free Remission (TFR) of patients with CML-Chronic Phase, after stopping TASIGNA® (Nilotinib), following frontline treatment with this agent. This analysis included 215 patients Philadelphia Chromosome positive (Ph+) CML patients in the Chronic Phase, who received at least 2 years of frontline treatment with TASIGNA® and had achieved a Molecular Response 4.5 (BCR-ABL International Scale 0.0032%). Patients then continued TASIGNA® for an additional year (Consolidation Phase) with close monitoring for sustained deep molecular response using real-time quantitative PCR assessments at 12-week intervals. Patients who had a molecular response no worse than MR4 (0.01% or less) in the last assessment of the Consolidation phase (N=190), were then eligible to stop treatment (TFR phase). The median age of these patients was 55 years. The median time from study entry to first MR4.5 was 18 months. The median duration of treatment on TASIGNA® prior to TFR was 43 months.
After 48 weeks without treatment, 51.6% of the 190 patients in the TFR phase remained in MMR and did not require re-initiation of treatment. Of the 86 patients who restarted treatment with TASIGNA® due to loss of MMR, 99% were able to regain MMR and 88% were able to regain MR4.5. The median time to MMR for these patients was 7.9 weeks and for MR4.5 was 13.1 weeks. About 25% of the patients experienced musculoskeletal pain during the first year of the TFR phase, which is an expected adverse event from TKI withdrawal.
The authors concluded that TFR is clinically meaningful and a high rate of sustained MR4.5 leads to stable disease. TASIGNA® is the first Tyrosine Kinase Inhibitor to demonstrate successful TFR in a large proportion of eligible patients after relatively short duration of treatment of 3.6 years. Over 50% of patients who stopped therapy were able to remain treatment free at 48 weeks. The authors recommended that frequent patient monitoring during Treatment-Free Remission (TFR) period, allows timely determination of loss of MR4.0 and MMR and the need for treatment re-initiation. Treatment-free remission (TFR) in patients (pts) with chronic myeloid leukemia in chronic phase (CML-CP) treated with frontline nilotinib: Results from the ENESTFreedom study. Hochhaus A, Masszi T, Giles FJ, et al. J Clin Oncol 34, 2016 (suppl; abstr 7001)
BCR-ABL Transcript Type May Predict Outcomes in Patients with Chronic Phase CML Treated with Tyrosine Kinase Inhibitors
SUMMARY: Chronic Myeloid Leukemia (CML) constitutes a little over 10% of all new cases of leukemia. The American Cancer Society estimates that about 8,220 new CML cases will be diagnosed in the United States in 2016 and about 1,070 patients will die of the disease. The hallmark of CML, the Philadelphia Chromosome (Chromosome 22), is a result of a reciprocal translocation-t(9;22), between chromosomes 9 and 22, wherein the ABL gene from chromosome 9, fuses with the BCR gene on chromosome 22. As a result, the auto inhibitory function of the ABL gene is lost and the BCR-ABL fusion gene is activated resulting in cell proliferation and leukemic transformation of hematopoietic stem cells. Even though the reciprocal translocation resulting in the formation of Philadelphia chromosome involves a fairly constant breakpoint in the ABL gene on chromosome 9, the breakpoint in the BCR gene on chromosome 22 can vary, resulting in different BCR-ABL transcript types. There has been ongoing debate whether the type of transcript, has prognostic significance, for patients with newly diagnosed chronic phase CML. Over 95% of patients with CML have expression of e13a2 (b2a2), e14a2 (b3a2), or both transcripts, coding for p210 BCR-ABL tyrosine kinase, whereas a small minority of patients express rare variants such as e1a2 transcripts, which code for p190 BCR-ABL, which is associated with aggressive disease. Previously published studies have shown that expression of certain type of transcripts may predict response to therapy, as well as outcomes.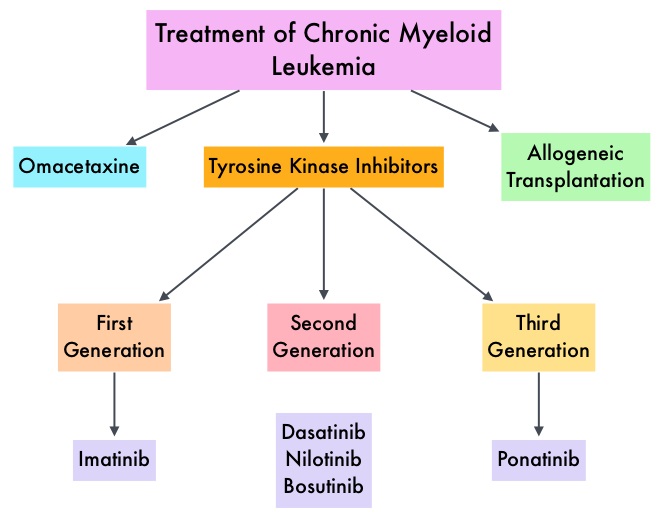
The purpose of this study was to evaluate the prognostic relevance of the commonly expressed BCR-ABL transcripts in patients with chronic phase CML and the influence of the transcript type, on molecular and cytogenetic responses, across chronic phase CML patients, treated with different Tyrosine Kinase Inhibitors (TKI). This analysis included 481 treatment naïve patients with chronic phase CML treated with different TKI modalities, expressing e13a2 (42%), e14a2 (41%), or coexpression of both e13a2 with e14a2 (18%) transcripts. These patients were treated with 4 different frontline TKIs which included, GLEEVEC® (Imatinib) 400 mg daily (N=69), GLEEVEC® 800 mg daily (N=199), SPRYCEL® (Dasatinib) 50 mg twice daily or 100 mg daily (N=105) and TASIGNA® (Nilotinib) 400 mg twice daily (N=108).
It was noted that patients with e13a2 transcripts who received GLEEVEC® 400 mg had an inferior Complete Cytogenetic Response (77%) compared with other TKI modalities (90-95%). Unlike these patients, patients with e14a2 transcripts or those expressing both e13a2 and e14a2 transcripts treated with GLEEVEC® 400 mg, had a Complete Cytogenetic Response rate of 93%, which was similar to treatment with other TKI modalities (93-96%). Even though the time to Complete Cytogenetic Response was 3 months and similar in all treatment groups, the trend for lower rates of Complete Cytogenetic Response and Major Cytogenetic Response for the e13a2 transcript cohort compared with the e14a2 cohort, persisted even at 60 months. Patients with e13a2 treated with GLEEVEC® 400 mg similarly had an inferior Major Molecular Response (MMR) at all time points than individuals with e14a2 and inferior MR4.5, compared with those treated with other TKI modalities. In patients with e14a2 transcripts, the MMR and MR4.5 rates were generally similar with all TKI modalities. In a multivariate analysis, patients with e14a2 alone or those with coexpressed e13a2, achieved an earlier and deeper response, compared to those with e13a2 transcripts, and predicted for longer event-free and transformation-free survival.
The authors concluded that the type of BCR-ABL transcript may determine outcomes in patients with chronic phase CML. Patients with e13a2 transcripts have lower platelet count and inferior outcomes with GLEEVEC® 400 mg, whereas patients with e14a2 have favorable outcomes regardless of TKI treatment modality. Further, expression of e14a2 or both e14a2 and e13a2 predicts optimal responses and longer Event Free Survival and Transformation Free Survival. Thus BCR-ABL transcript type may help in selecting the appropriate treatment and may predict outcomes in patients with chronic phase CML. Impact of BCR-ABL transcript type on outcome in patients with chronic-phase CML treated with tyrosine kinase inhibitors. Jain P, Kantarjian H, Patel KP, et al. Blood 2016;127:1269-1275
Choosing Appropriate Therapy in Chronic Myeloid Leukemia
SUMMARY: Chronic Myeloid Leukemia (CML) constitutes approximately 10% of all new cases of leukemia. The American Cancer Society estimates that 6,660 new CML cases will be diagnosed in the United States in 2015 and about 1,140 people will die of the disease. The hallmark of CML, the Philadelphia Chromosome (Chromosome 22), is a result of a reciprocal translocation between chromosomes 9 and 22, wherein the ABL gene from chromosome 9, fuses with the BCR gene on chromosome 22. As a result, the auto inhibitory function of the ABL gene is lost and the BCR-ABL fusion gene is activated resulting in cell proliferation and leukemic transformation of hematopoietic stem cells.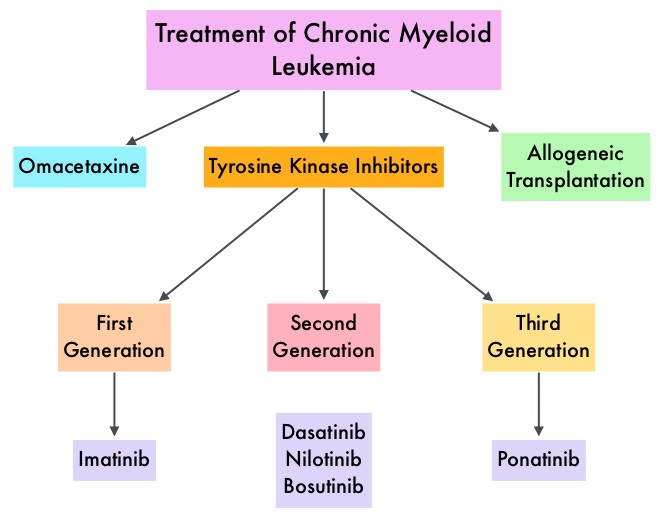 Gleevec® (Imatinib) inhibits the BCR-ABL tyrosine kinase and is the standard first line treatment, of Ph chromosome positive (Ph+) leukemias. Lack of response due to resistance to GLEEVEC® and in some instances drug intolerance, has led to the development of newer agents including Second and Third generation Tyrosine Kinase Inhibitors (TKIs). Resistance to Gleevec® and other TKIs sharing the same therapeutic target (BCR-ABL kinase), has been attributed to point mutations in the ABL kinase domain, amplification of the BCR-ABL gene as well as other BCR- ABL independent mechanisms such as upregulation of SRC kinases. Mutation analysis at the time of TKI failure, utilizing high sensitivity sequencing techniques such as Next Generation Sequencing, can give clinically relevant information related to low level mutations and compound mutations and this information in turn, can dictate choice of second line therapy.
Gleevec® (Imatinib) inhibits the BCR-ABL tyrosine kinase and is the standard first line treatment, of Ph chromosome positive (Ph+) leukemias. Lack of response due to resistance to GLEEVEC® and in some instances drug intolerance, has led to the development of newer agents including Second and Third generation Tyrosine Kinase Inhibitors (TKIs). Resistance to Gleevec® and other TKIs sharing the same therapeutic target (BCR-ABL kinase), has been attributed to point mutations in the ABL kinase domain, amplification of the BCR-ABL gene as well as other BCR- ABL independent mechanisms such as upregulation of SRC kinases. Mutation analysis at the time of TKI failure, utilizing high sensitivity sequencing techniques such as Next Generation Sequencing, can give clinically relevant information related to low level mutations and compound mutations and this information in turn, can dictate choice of second line therapy. 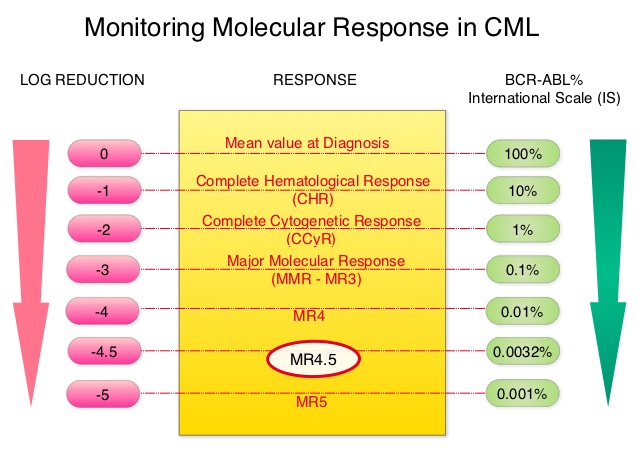 The Second generation TKIs, TASIGNA® (Nilotinib) and SPRYCEL® (Dasatinib) although initially approved for second line treatment of CML after GLEEVEC® resistance or intolerance, are now FDA approved for the treatment of newly diagnosed Chronic Phase CML. This approval was based on the rapid and superior Major Molecular Responses (MMR) noted, when compared to GLEEVEC®. Now, that the Second generation TKIs are being used as first line therapy, the choice of second line therapy after failure with Second generation TKIs has become more nebulous. It is clear however that, patients with primary cytogenetic resistance to ï¬rst and second line therapy do not beneï¬t from sequential therapy with Second generation TKIs and BCR-ABL mutation analysis should be performed in all patients who develop TKI resistant disease. Before switching from a Second to a Third generation TKI such as Ponatinib, the following considerations should be taken into account
The Second generation TKIs, TASIGNA® (Nilotinib) and SPRYCEL® (Dasatinib) although initially approved for second line treatment of CML after GLEEVEC® resistance or intolerance, are now FDA approved for the treatment of newly diagnosed Chronic Phase CML. This approval was based on the rapid and superior Major Molecular Responses (MMR) noted, when compared to GLEEVEC®. Now, that the Second generation TKIs are being used as first line therapy, the choice of second line therapy after failure with Second generation TKIs has become more nebulous. It is clear however that, patients with primary cytogenetic resistance to ï¬rst and second line therapy do not beneï¬t from sequential therapy with Second generation TKIs and BCR-ABL mutation analysis should be performed in all patients who develop TKI resistant disease. Before switching from a Second to a Third generation TKI such as Ponatinib, the following considerations should be taken into account
BCR-ABL Mutations and Sensitivity to Second Generation TKIs
1) Patients with F317L/V/I/C mutations are more sensitive to TASIGNA® (Nilotinib) or BOSULIF® (Bosutinib) than to SPRYEL® (Dasatinib)
2) Patients with V299L mutation are more sensitive to TASIGNA® than to BOSULIF® or SPRYCEL®
3) Patients with Y253F/H, E255K/V, and F359V/I/C mutations are more sensitive to SPRYCEL® or BOSULIF® than to TASIGNA®
Tolerability of Second Generation TKIs
1) Patients who experience pleural effusion during SPRYCEL® treatment might better tolerate TASIGNA® or BOSULIF®
2) Patients who experience rash during treatment with TASIGNA® or BOSULIF® could be switched to SPRYCEL®
3) Some toxicities common with other TKIs such as pleural effusion and cardiac toxicity are less common with BOSULIF® and this agent also has activity against many BCR-ABL kinase domain mutations resistant to GLEEVEC®, SPRYCEL® and TASIGNA®, with the exception of T315I mutation.
It should be noted that Second generation TKI as third line therapy has limited value in majority of the patients with CML. ICLUSIG® (Ponatinib) is a Third generation kinase inhibitor approved for the treatment of patients with T315I positive CML or T315I-positive Philadelphia Chromosome positive Acute Lymphoblastic Leukemia (Ph+ ALL) and for whom no other TKI is indicated. Other treatment options include SYNRIBO® (Omacetaxine Mepesuccinate), a first-in-class Cephalotaxine and a semi synthetic purified Homoharringtonine (HHT) compound. Unlike Tyrosine Kinase Inhibitors , SYNRIBO® is a protein synthesis inhibitor and reduces the levels of multiple Oncoproteins including BCR-ABL, BCL-2, MCL-1 and promotes apoptosis of leukemic stem cells. This agent is presently approved for the treatment of Chronic or Accelerated phase Chronic Myeloid Leukemia (CML) with resistance and/or intolerance to two or more Tyrosine Kinase Inhibitors, with cytopenias being the most common toxicity. Allogeneic Hematopoietic Stem Cell transplantation should be considered for eligible patients with T315I mutation not responding to ICLUSIG®, those with mutations resistance to second and third generation TKIs and patients with Accelerated or Blast phase CML, following remission with TKIs. Use of Second- and Third-Generation Tyrosine Kinase Inhibitors in the Treatment of Chronic Myeloid Leukemia: An Evolving Treatment Paradigm. Jabbour E, Kantarjian H and Cortes J. Clinical Lymphoma, Myeloma & Leukemia 2015;15:323-334
Deep Molecular Response Is Reached by the Majority of Patients Treated With Imatinib, Predicts Survival, and Is Achieved More Quickly by Optimized High-Dose Imatinib Results From the Randomized CML-Study IV
SUMMARY:Chronic Myeloid Leukemia (CML) constitutes approximately 10% of all new cases of leukemia. The American Cancer Society estimates that 5,980 new CML cases will be diagnosed in the United States in 2014 and 810 people will die of the disease. The hallmark of CML, the Philadelphia Chromosome (Chromosome 22), is a result of a reciprocal translocation between chromosomes 9 and 22, wherein the ABL gene from chromosome 9 fuses with the BCR gene on chromosome 22. As a result, the auto inhibitory function of the ABL gene is lost and the BCR-ABL fusion gene is activated resulting in cell proliferation and leukemic transformation of hematopoietic stem cells. The presently available Tyrosine Kinase Inhibitors (TKI’s) approved in the United States including GLEEVEC®, share the same therapeutic target, which is BCR-ABL kinase. Resistance to TKI’s occur as a result of mutations in the BCR-ABL kinase domain or amplification of the BCR-ABL gene. With the availability of newer therapies for CML, monitoring response to treatment is important. This is best accomplished by measuring the amount of residual disease using Reverse Transcription-Polymerase Chain Reaction (RT-PCR).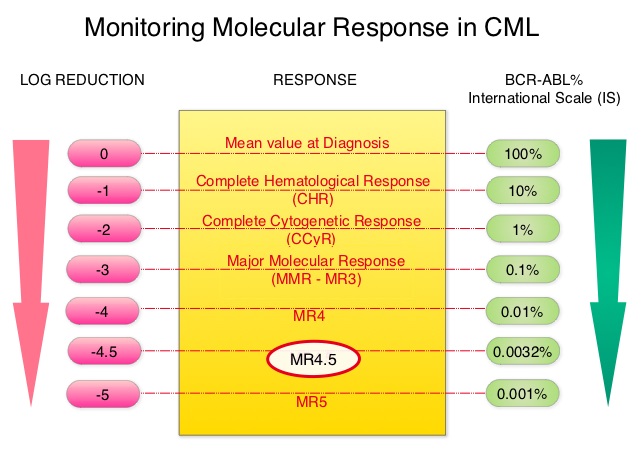 This is expressed using the International Scale (IS) as BCR-ABL%, which is the ratio between BCR-ABL and a control gene. BCR-ABL kinase domain point mutations are detected, using the mutational analysis by Sanger sequencing. Majority of the patients receiving a TKI following diagnosis of CML achieve a Complete Cytogenetic Response (CCyR) within 12 months following commencement of therapy and these patients have a life expectancy similar to that of their healthy counterparts. However, some patients have deeper responses (MR3, MR4, and MR4.5) and it is presumed that this subgroup of patients with CML may stay in unmaintained remission even after treatment discontinuation. Moreover, it is not clear what proportion of patients with CML achieve deeper responses and deeper responses have not been shown to increase survival beyond that associated with CCyR. To address these questions, the authors in this report analyzed the data from the randomized CML – Study IV to characterize the frequency and impact of deep molecular response on survival with different treatment modalities. The study is a five arm trial in which the treatment groups included high dose Imatinib (GLEEVEC® 800 mg/day), GLEEVEC® 400 mg/day, GLEEVEC® 400 mg/day in combination with Interferon alfa (IFN), GLEEVEC® 400 mg/day in combination with Cytarabine, and GLEEVEC® 400 mg/day after IFN failure. The analysis included a total of 1538 patients and the principal objective of CML – Study IV was to determine the impact of MMR (Major Molecular Response) on survival, remission rates and survival probabilities. After a median follow up of 67.5 months, 5 year overall survival was 90%, 8 year overall survival was 86% and 5 year PFS was 87.5%. The cumulative rate of MR4.5, irrespective of treatment group (defined as 4.5 or more log reduction in BCR-ABL transcripts), was 66% at 8 years and 70% at 9 years and the median time to reaching MR4.5 was 4.9 years. High dose GLEEVEC® therapy and early Major Molecular Remission predicted deep molecular response (MR4.5). High dose GLEEVEC® resulted in a more rapid MR4.5 than with GLEEVEC® 400 mg/day (P = .016). Finally, this analysis showed that a confirmed MR4.5 at 4 years predicted significantly higher 8 year overall survival probability compared to CCyR (Complete Cytogenetic response: IS 1%) or MMR (major molecular response: IS 0.1%) – 92% versus 83%, P=0.047. The authors concluded that deep molecular response (MR4.5) is a new molecular predictor of long term survival in CML patients and is achieved in a majority of patients treated with GLEEVEC®, and is achieved more rapidly with optimized high-dose GLEEVEC®. The authors further pointed out that none of the patients with confirmed MR4.5 had disease progression and this may therefore provide a therapeutic rationale for discontinuing treatment in this subset of patients with CML. These findings may also justify the use of more effective second generation TKI’s to induce early and deep molecular responses. Hehlmann R, Müller MC, Lauseker M, et al. J Clin Oncol 2014;32:415-423
This is expressed using the International Scale (IS) as BCR-ABL%, which is the ratio between BCR-ABL and a control gene. BCR-ABL kinase domain point mutations are detected, using the mutational analysis by Sanger sequencing. Majority of the patients receiving a TKI following diagnosis of CML achieve a Complete Cytogenetic Response (CCyR) within 12 months following commencement of therapy and these patients have a life expectancy similar to that of their healthy counterparts. However, some patients have deeper responses (MR3, MR4, and MR4.5) and it is presumed that this subgroup of patients with CML may stay in unmaintained remission even after treatment discontinuation. Moreover, it is not clear what proportion of patients with CML achieve deeper responses and deeper responses have not been shown to increase survival beyond that associated with CCyR. To address these questions, the authors in this report analyzed the data from the randomized CML – Study IV to characterize the frequency and impact of deep molecular response on survival with different treatment modalities. The study is a five arm trial in which the treatment groups included high dose Imatinib (GLEEVEC® 800 mg/day), GLEEVEC® 400 mg/day, GLEEVEC® 400 mg/day in combination with Interferon alfa (IFN), GLEEVEC® 400 mg/day in combination with Cytarabine, and GLEEVEC® 400 mg/day after IFN failure. The analysis included a total of 1538 patients and the principal objective of CML – Study IV was to determine the impact of MMR (Major Molecular Response) on survival, remission rates and survival probabilities. After a median follow up of 67.5 months, 5 year overall survival was 90%, 8 year overall survival was 86% and 5 year PFS was 87.5%. The cumulative rate of MR4.5, irrespective of treatment group (defined as 4.5 or more log reduction in BCR-ABL transcripts), was 66% at 8 years and 70% at 9 years and the median time to reaching MR4.5 was 4.9 years. High dose GLEEVEC® therapy and early Major Molecular Remission predicted deep molecular response (MR4.5). High dose GLEEVEC® resulted in a more rapid MR4.5 than with GLEEVEC® 400 mg/day (P = .016). Finally, this analysis showed that a confirmed MR4.5 at 4 years predicted significantly higher 8 year overall survival probability compared to CCyR (Complete Cytogenetic response: IS 1%) or MMR (major molecular response: IS 0.1%) – 92% versus 83%, P=0.047. The authors concluded that deep molecular response (MR4.5) is a new molecular predictor of long term survival in CML patients and is achieved in a majority of patients treated with GLEEVEC®, and is achieved more rapidly with optimized high-dose GLEEVEC®. The authors further pointed out that none of the patients with confirmed MR4.5 had disease progression and this may therefore provide a therapeutic rationale for discontinuing treatment in this subset of patients with CML. These findings may also justify the use of more effective second generation TKI’s to induce early and deep molecular responses. Hehlmann R, Müller MC, Lauseker M, et al. J Clin Oncol 2014;32:415-423
Subcutaneous Omacetaxine Mepesuccinate in Patients With Chronic-Phase Chronic Myeloid Leukemia Previously Treated With 2 or More Tyrosine Kinase Inhibitors Including Imatinib
SUMMARY: Omacetaxine Mepesuccinate (SYNRIBO®) is a first-in-class cephalotaxine and is a semi synthetic purified Homoharringtonine (HHT) compound. HHT is a plant alkaloid derived from Cephalotaxus fortunei, a coniferous bush, also called Japanese Plum Yew. With over 40 years of drug development history since its discovery, SYNRIBO®, unlike Tyrosine Kinase Inhibitors (TKI), is a protein synthesis inhibitor and reduces the levels of multiple Oncoproteins including BCR-ABL, BCL-2, MCL-1 and promotes apoptosis of leukemic stem cells. The following is the pooled data from 2 phase II trials. Treatment population included patients with Chronic Myeloid Leukemia – Chronic Phase (CML-CP), resistant or intolerant to GLEEVEC® (Imatinib) and at least one other TKI such as SPRYCEL® (Dasatinib) and/or TASIGNA® (Nilotinib). All patients had prior treatment with GLEEVEC®, 85% had prior treatment with SPRYCEL® and 59% had prior treatment with TASGNA®. The primary end point was Major Cytogenetic Response (MCyR) and 81 patients with a median age of 59 years were included in this analysis. Treatment consisted of subcutaneous SYNRIBO®, administered at 1.25 mg/m2 twice daily for 14 consecutive days every 4 weeks until response, then for 7 days every 4 weeks as maintenance treatment. The median duration of treatment was 7.5 months. MCyR was noted in 20% of the patients and the median response duration was 17.7 months. Hematologic response was seen in 69% of the patients and the median response duration was 12.2 months. The median failure-free survival was 9.6 months and overall survival was 34 months. The most common grade 3/4 toxicities were cytopenias. The authors concluded that SYNRIBO® has clinical activity in a heavily pretreated population of patients with CML-CP and should therefore be considered for patients with CML-CP with resistance or intolerance to 2 or more TKI’s. By virtue of its mechanism of action, patients with T315I BCR-ABL mutation may potentially benefit from this unique compound. Cortes JE, Nicolini FE, Wetzler M, et al. Clin Lymphoma Myeloma Leuk. 2013;13:584-591.
ICLUSIG® (Ponatinib)
ICLUSIG® (Ponatinib): The FDA on December 17, 2012 granted accelerated approval to ICLUSIG® for the treatment of adult patients with chronic phase, accelerated phase, or blast phase chronic myeloid leukemia (CML) that is resistant or intolerant to prior tyrosine kinase inhibitor therapy or Philadelphia chromosome positive acute lymphoblastic leukemia (Ph+ALL) that is resistant or intolerant to prior tyrosine kinase inhibitor therapy. ICLUSIG® is available as oral tablets and is a product of ARIAD Pharmaceuticals, Inc.
SYNRIBO® (Omacetaxine mepesuccinate)
The FDA on October 26, 2012 granted accelerated approval to SYNRIBO® for the treatment of adult patients with chronic or accelerated phase chronic myeloid leukemia (CML) with resistance and/or intolerance to two or more tyrosine kinase inhibitors (TKIs). SYNRIBO® is administered as a subcutaneous injection and is a product of Teva Pharmaceutical Industries Ltd.
BOSULIF® (Bosutinib)
The FDA on September 4, 2012 approved BOSULIF® (Bosutinib) for the treatment of chronic, accelerated, or blast phase Philadelphia chromosome positive (Ph+) chronic myelogenous leukemia (CML) in adult patients with resistance or intolerance to prior therapy. BOSULIF® tablets are a product of Pfizer, Inc.
