SUMMARY: Chronic Lymphocytic leukemia (CLL) is a disease of the elderly with a median age at diagnosis of 72 years. Given the age at diagnosis, it is not uncommon for these patients to have multiple comorbidities. The authors in this trial attempted to study a new agent, Obinutuzumab or GAZYVA® (GA101) specifically in this patient population. 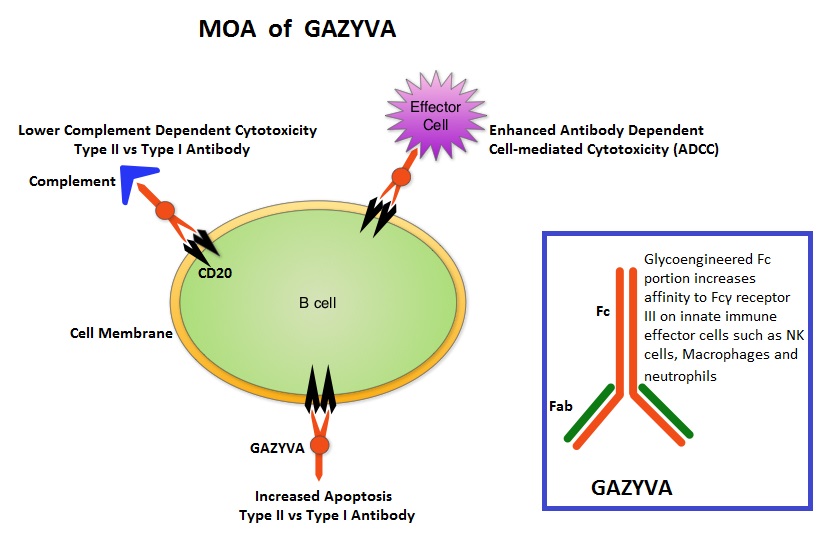 GAZYVA® is glycoengineered, fully humanized, third generation, type II anti-CD20 antibody (IgG1 monoclonal antibody) that selectivity binds to the extracellular domain of the CD20 antigen on malignant human B cells. By virtue of binding affinity of the glycoengineered Fc portion of GAZYVA® to Fcγ receptor III on innate immune effector cells such as natural killer cells, macrophages and neutrophils, antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis is significantly enhanced, whereas it induces very little complement-dependent cytotoxicity. This is in contrast to RITUXAN® (Rituximab), which is a first generation type I, chimeric anti-CD20 targeted monoclonal antibody that kills CLL cells primarily by complement-dependent cytotoxicity and also ADCC. In this phase III trial, LEUKERAN® (Chlorambucil) was compared with a combination of GAZYVA® plus LEUKERAN® and a combination of RITUXAN® plus LEUKERAN®. Five Hundred and eighty nine (589) treatment naïve CLL patients over 70 years of age with comorbidities were enrolled of whom 118 patients received LEUKERAN® alone, 238 received GAZYVA® plus LEUKERAN® and 233 received RITUXAN® plus LEUKERAN®. The primary endpoint was Progression-Free Survival (PFS). Chemoimmunotherapy with both GAZYVA® plus LEUKERAN® and RITUXAN® plus LEUKERAN® significantly prolonged PFS compared to LEUKERAN® alone. The median PFS was 11.1 months with LEUKERAN® alone compared to 26.7 months for GAZYVA® plus LEUKERAN® (HR=0.18, P<0.001) and 16.3 months for RITUXAN® plus LEUKERAN® (HR=0.44, P<0.001). This benefit was seen in all subgroups except those with del(17) and quality of life in those who received antibody along with LEUKERAN® was not compromised. The combination of GAZYVA® and LEUKERAN®, also prolonged overall survival when compared to LEUKERAN® alone (HR=0.41; P=0.002). This benefit however was not noted with the RITUXAN® plus LEUKERAN® combination. Treatment with GAZYVA® plus LEUKERAN® when compared with RITUXAN® plus LEUKERAN®, resulted in a longer PFS (26.7 vs15.2 months; HR=0.39; P<0.001), higher complete response rates (20.7% vs. 7.0%) and deeper molecular responses. Infusion related reactions were more common in the GAZYVA® plus LEUKERAN® group without increase in the risk for infections. The authors concluded that a combination of GAZYVA® and LEUKERAN® when given to elderly patients with comorbid conditions improved overall survival compared to LEUKERAN® alone and resulted in higher response rates and longer PFS than RITUXAN® plus LEUKERAN®. Goede V, Fischer K, Busch R, et al. N Engl J Med 2014; 370:1101-1110
GAZYVA® is glycoengineered, fully humanized, third generation, type II anti-CD20 antibody (IgG1 monoclonal antibody) that selectivity binds to the extracellular domain of the CD20 antigen on malignant human B cells. By virtue of binding affinity of the glycoengineered Fc portion of GAZYVA® to Fcγ receptor III on innate immune effector cells such as natural killer cells, macrophages and neutrophils, antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis is significantly enhanced, whereas it induces very little complement-dependent cytotoxicity. This is in contrast to RITUXAN® (Rituximab), which is a first generation type I, chimeric anti-CD20 targeted monoclonal antibody that kills CLL cells primarily by complement-dependent cytotoxicity and also ADCC. In this phase III trial, LEUKERAN® (Chlorambucil) was compared with a combination of GAZYVA® plus LEUKERAN® and a combination of RITUXAN® plus LEUKERAN®. Five Hundred and eighty nine (589) treatment naïve CLL patients over 70 years of age with comorbidities were enrolled of whom 118 patients received LEUKERAN® alone, 238 received GAZYVA® plus LEUKERAN® and 233 received RITUXAN® plus LEUKERAN®. The primary endpoint was Progression-Free Survival (PFS). Chemoimmunotherapy with both GAZYVA® plus LEUKERAN® and RITUXAN® plus LEUKERAN® significantly prolonged PFS compared to LEUKERAN® alone. The median PFS was 11.1 months with LEUKERAN® alone compared to 26.7 months for GAZYVA® plus LEUKERAN® (HR=0.18, P<0.001) and 16.3 months for RITUXAN® plus LEUKERAN® (HR=0.44, P<0.001). This benefit was seen in all subgroups except those with del(17) and quality of life in those who received antibody along with LEUKERAN® was not compromised. The combination of GAZYVA® and LEUKERAN®, also prolonged overall survival when compared to LEUKERAN® alone (HR=0.41; P=0.002). This benefit however was not noted with the RITUXAN® plus LEUKERAN® combination. Treatment with GAZYVA® plus LEUKERAN® when compared with RITUXAN® plus LEUKERAN®, resulted in a longer PFS (26.7 vs15.2 months; HR=0.39; P<0.001), higher complete response rates (20.7% vs. 7.0%) and deeper molecular responses. Infusion related reactions were more common in the GAZYVA® plus LEUKERAN® group without increase in the risk for infections. The authors concluded that a combination of GAZYVA® and LEUKERAN® when given to elderly patients with comorbid conditions improved overall survival compared to LEUKERAN® alone and resulted in higher response rates and longer PFS than RITUXAN® plus LEUKERAN®. Goede V, Fischer K, Busch R, et al. N Engl J Med 2014; 370:1101-1110

