SUMMARY: The FDA on September 22, 2017 granted accelerated approval to KEYTRUDA® (Pembrolizumab), for patients with recurrent locally advanced or metastatic, Gastric or GastroEsophageal junction adenocarcinoma, whose tumors express PD-L1, as determined by an FDA-approved test. Patients must have had disease progression on or after two or more prior systemic therapies, including Fluoropyrimidine and platinum-containing chemotherapy and, if appropriate, HER2/neu-targeted therapy. Cancers of the esophagus and stomach are among the most prevalent malignancies and are a major cause of cancer-related mortality. It is estimated that in 2016 GastroEsophageal Adenocarcinoma (GEA) accounted for 43,280 new cases in the United States. The incidence of these tumors has been on the rise in the past decade. Majority of the patients with GEA have advanced disease at the time of initial presentation and have limited therapeutic options with little or no chance for cure. Patients with localized disease (stage II and stage III) are often treated with multimodality therapy and 40% of the patients may survive for 5 years or more.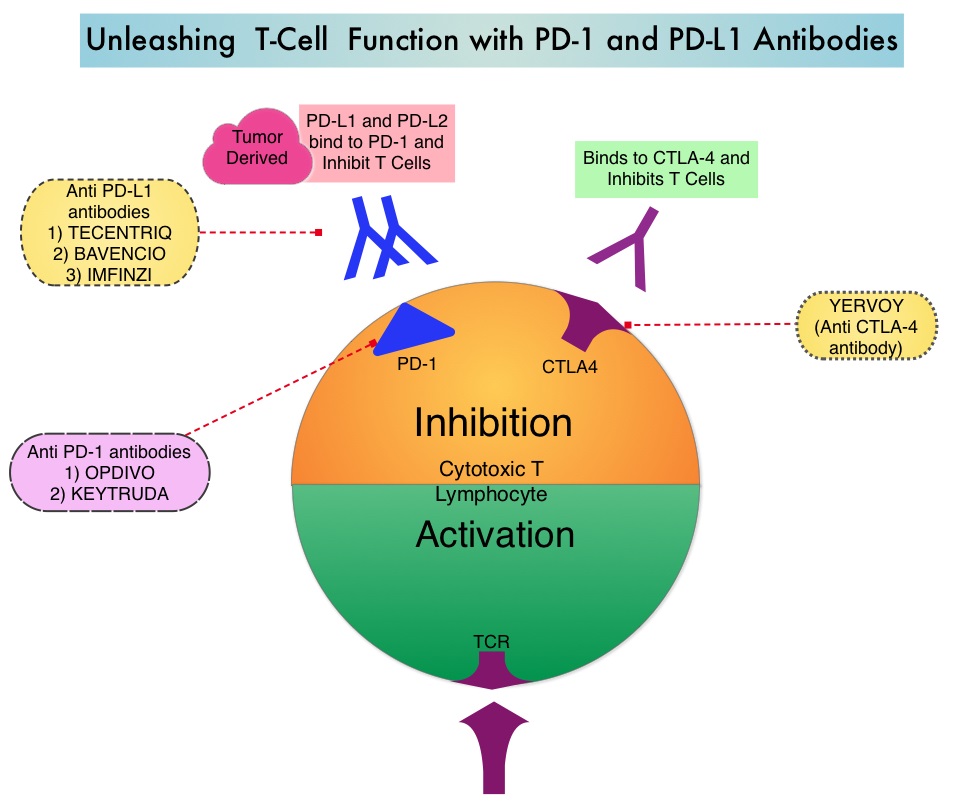
KEYTRUDA® is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2, thereby undoing PD-1 pathway-mediated inhibition of the immune response and unleashing the tumor-specific effector T cells. In previously reported Phase I study, KEYTRUDA® showed promising antitumor activity with manageable safety, in patients with previously treated advanced gastric cancer.
Based on these findings, the authors conducted a global, multicohort, Phase II study (KEYNOTE 059), in which patients with advanced gastric or gastroesophageal junction cancer received KEYTRUDA® 200 mg every 3 weeks for up to 2 years or until disease progression or unacceptable toxicity. Cohort 1 in this study enrolled 259 patients of whom 57% (N=148) had PD-L1 positive tumors and these tumors were either MicroSatellite Stable (MSS), or had undetermined MicroSatellite Instability (MSI) or MisMatch Repair (MMR) status. PD-L1 expression was evaluated by the PD-L1 IHC (22C3 antibody) and PD-L1 positivity was based on a Combined Positive Score (CPS) of 1 or more. CPS is determined by the number of PD-L1 staining cells (tumor cells, lymphocytes, macrophages) divided by total number of tumor cells evaluated, multiplied by 100. The median age was 62 years, 76% were men and over 50% of the patients received KEYTRUDA® as third line treatment or beyond. The median duration of follow up was 5.4 months. Primary end points included Objective Response Rate (ORR), safety, and tolerability.
In the group of patients with PD-L1 positive tumors, the ORR was 15.5% with 2% Complete Responses and 13.5% Partial Responses. Among the responding patients, the response duration ranged from 3-19 months, with 58% of the responding patients having response durations of 6 months or longer and 26% of the responding patients having response durations of 12 months or longer. In this cohort of 259 patients, 3% had tumors that were determined to be MSI-High. The ORR in this group was 57%, with response duration ranging from 5-14 months. In the PD-L1 negative patients however, the ORR was only 5.5%.
It was concluded that KEYTRUDA® demonstrated promising antitumor activity and durable responses in patients with advanced Gastric/GastroEsophageal Junction cancer, who had progressed on more than 2 lines of therapy, with higher Objective Response Rates noted in patients with PD-L1-positive tumors. KEYNOTE-059 cohort 1: Efficacy and safety of pembrolizumab (pembro) monotherapy in patients with previously treated advanced gastric cancer. Fuchs CS, Doi T, Jang RW-J, et al. J Clin Oncol. 2017;35 (suppl; abstr 4003).

