SUMMARY: The American Cancer Society estimates that over 21,000 women will be diagnosed with ovarian cancer in the United States for 2015 and over 14,000 will die of the disease. Ovarian cancer ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. Intraperitoneal (IP) delivery of antineoplatic drugs ("Belly Bath") for ovarian cancer dates back to the late 1970’s and 1980’s. This strategy for ovarian cancer was based on the fact that the peritoneal cavity is the primary site of spread and failure in most cases of advanced ovarian cancer. 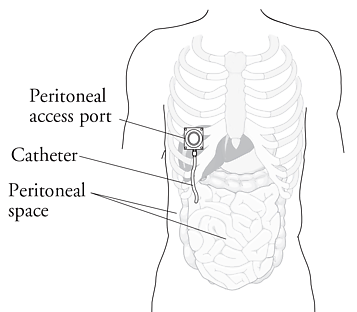 IP chemotherapy for ovarian cancer facilitates the exposure of tumors in the peritoneal cavity to 10-20 fold greater concentration of Cisplatin and Carboplatin and 1000 fold greater concentration of Paclitaxel, compared to IV administration, thus allowing continuous and prolonged exposure of the tumor to high drug concentrations, without systemic toxicities. Even though three Intergroup Phase III trials demonstrated the superiority of IP therapy over IV therapy, it has not been widely accepted in the US and abroad. Barriers to IP therapy have included inconvenience, IP catheter related complications, higher toxicities, lack of knowledge regarding patient selection for IP therapy as well as minimum number of cycles of IP therapy to administer and uncertain long term benefit.
IP chemotherapy for ovarian cancer facilitates the exposure of tumors in the peritoneal cavity to 10-20 fold greater concentration of Cisplatin and Carboplatin and 1000 fold greater concentration of Paclitaxel, compared to IV administration, thus allowing continuous and prolonged exposure of the tumor to high drug concentrations, without systemic toxicities. Even though three Intergroup Phase III trials demonstrated the superiority of IP therapy over IV therapy, it has not been widely accepted in the US and abroad. Barriers to IP therapy have included inconvenience, IP catheter related complications, higher toxicities, lack of knowledge regarding patient selection for IP therapy as well as minimum number of cycles of IP therapy to administer and uncertain long term benefit.
The authors in this study retrospectively analyzed data from 876 patients in the two phase III, Gynecologic Oncology Group trials (GOG#114 and GOG#172). The purpose of this study was to determine the long-term survival and associated prognostic factors following IP chemotherapy, in patients with advanced ovarian cancer. In both studies, patients were randomly assigned to IP (combined N=440) or IV (combined N=436) chemotherapy. In GOG#114 trial, the two treatment groups were Paclitaxel at 135 mg/m2 IV followed by Cisplatin 75 mg/m2 IV for 6 cycles or Carboplatin IV for 2 courses followed by Paclitaxel 135 mg/m2 IV dose on day 1 and Cisplatin 100 mg/m2 IP on day 8, for 6 cycles. In GOG#172 trial, the two treatment groups (IV vs IP) were Paclitaxel at 135 mg/m2 IV followed by Cisplatin 75 mg/m2 IV on day 2 for 6 cycles or Cisplatin 100mg/m2 IP on day 2 and Paclitaxel 60 mg/m2 IP on day 8, for 6 cycles. Patients in the IP and IV groups were well balanced for baseline characteristics. At a median follow up of 10.7 years, the median Overall Survival with IP chemotherapy was 61.8 months compared with 51.4 months for IV chemotherapy and IP chemotherapy resulted in a 23% reduction in the risk of death (HR=0.77; P=0.002). IP chemotherapy was also associated with improved survival among those patients with gross residual disease ie.1 cm or less (HR = 0.75; P=0.006). The risk for death decreased by 12% for each cycle of IP chemotherapy that patients completed (HR=0.88; P<0.001). Factors significantly associated with poorer Overall Survival included clear/mucinous vs serous histology (HR=2.79; P <0 .001), gross residual vs no visible disease (HR=1.89; P< 0.001), and fewer vs more cycles of IP chemotherapy (HR=0.88; P<0.001). Younger patients were more likely to complete IP chemotherapy, with probability of completion decreasing by 5% with each additional year of age (P<0.001). The authors concluded that IP chemotherapy was associated with significantly prolonged Overall Survival in women with advanced ovarian cancer, including those with gross residual disease, when compared with IV chemotherapy. This benefit extends beyond 10 years and Overall Survival improved with increasing number of IP chemotherapy cycles administered. Long-Term Survival Advantage and Prognostic Factors Associated With Intraperitoneal Chemotherapy Treatment in Advanced Ovarian Cancer: A Gynecologic Oncology Group Study . Tewari D, Java J, Salani R, et al. JCO published online on March 23, 2015; DOI:10.1200/JCO.2014.55.9898.

