SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. About 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Approximately 50% of all breast cancers are Estrogen Receptor (ER) positive, HER2-negative, axillary node-negative tumors. Patients with early stage breast cancer often receive adjuvant chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for Hormone Receptor positive, HER negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
Oncotype Dx assay categorizes patients on the basis of Recurrence Scores into Low risk (less than 18), Intermediate risk (18-30), and High risk (31 or more). It has been unclear whether patients in the Intermediate risk group benefited from the addition of chemotherapy to endocrine therapy. TAILORx was specifically designed to address this question and provide a very definitive answer. In this study, the Intermediate risk Recurrent Score (18-30) was changed to 11-25, to account for exclusion of higher-risk patients with HER2-positive disease and to minimize the potential for under treatment.
TAILORx ((Trial Assigning Individualized Options for Treatment) is a phase III, randomized, prospective, non-inferiority trial, and is the largest breast cancer treatment trial ever conducted, and the first precision medicine trial ever done, according to the authors. In this study, 10,273 women, 18-75 years of age, with hormone receptor-positive, HER2-negative, axillary node-negative breast cancer were enrolled. Patients had tumors 1.1-5.0 cm in size (or 0.6-1.0 cm and intermediate/high grade). Patients were divided into three groups based on their Recurrence Score. Women with a Low Recurrence Score of 0-10 received endocrine therapy alone and those with a High Recurrence Score of 26-100 received endocrine therapy in combination with standard adjuvant chemotherapy. Patient with Intermediate Recurrence Score of 11-25 (N=6711) were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. Patients were followed up for 9 years. The Primary endpoint was invasive Disease Free Survival, defined as recurrence of cancer in the breast, regional lymph nodes, and/or distant organs, a second primary cancer in the opposite breast or another organ, or death from any cause. Results reported in June 2018 showed that while most women with an Intermediate Recurrence Score of 11-25 did not benefit from chemotherapy, women 50 years or younger with a Recurrence Score of 16-25 did indeed benefit from adjuvant chemotherapy.
The authors in this publication provided additional results from the same data set showing that adding “Clinical Risk” provides additional prognostic information. The investigators used a binary classification system from the MINDACT trial (Microarray in Node-Negative Disease May Avoid Chemotherapy), which used a 70-gene assay, and divided patients into high or low “Clinical Risk” based on tumor size and histologic grade. Clinical Risk was defined as low if the tumor was 3 cm or less in diameter and had a low histologic grade, 2 cm or less and had an intermediate histologic grade, or 1 cm or less in diameter and had a high grade. The Clinical Risk was defined as high if the low-risk criteria were not met. This additional reporting provided prognostic information about recurrent risk, but not benefits of chemotherapy particularly in the Intermediate Recurrence Score group.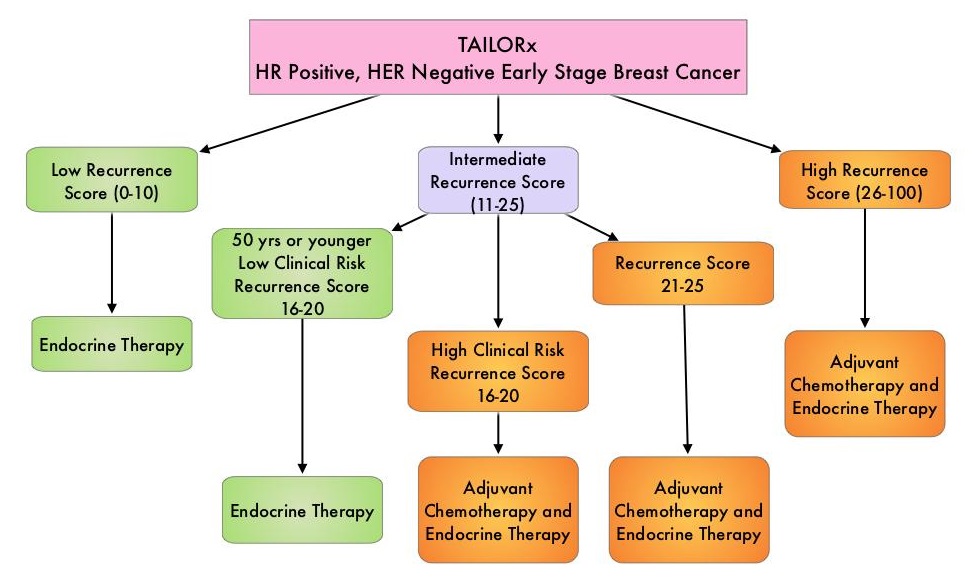
Among women who were 50 years of age or younger with a low Recurrence Score, the distant recurrence rate at 9 years was less than 5%, irrespective of Clinical Risk, and about 5% among those with an intermediate Recurrence Score with low Clinical Risk. However in sharp contrast, among women 50 years of age or younger, with high Clinical Risk and an intermediate Recurrence Score who had received endocrine therapy alone, the rate of distant recurrence at 9 years was 12.3%, compared with 6.1% among women who had received adjuvant chemotherapy. It is possible that younger women with a Recurrence Score of 11-25 and high Clinical Risk, receiving endocrine therapy alone, may have been undertreated with Tamoxifen, and the authors speculated that based on previously published studies, adding Ovarian Suppression and an Aromatase Inhibitor might result in risk reduction, equivalent to that observed using adjuvant chemotherapy.
The authors concluded that these new findings complement the original, definitive TAILORx conclusion and integration of genomic (Recurrence Score) and Clinical Risk may provide a more accurate estimate of prognosis for individual patients, than could be provided by either the genomic or clinical information alone. They added that this Clinical Risk stratification facilitates more refined estimates of absolute chemotherapy benefit for women 50 years of age or younger, with a Recurrence Score 16-25. Clinical and Genomic Risk to Guide the Use of Adjuvant Therapy for Breast Cancer. Sparano JA, Gray RJ, Ravdin PM, et al. N Engl J Med 2019;380:2395-2405

