SUMMARY: The American Cancer Society estimates that approximately 14,620 new cases of Chronic Lymphocytic Leukemia (CLL) were diagnosed in 2015 and approximately 4650 patients died from the disease. CLL is a disease of the elderly and the average age at the time of diagnosis is 72 years. There are two main types of lymphocytes, B and T lymphocytes/cells. B-cell CLL is the most common type of leukemia in adults. Normal B-cell activation and proliferation is dependent on B-cell receptor (BCR) signaling. This signaling is also important for initiation and progression of B-cell lymphoproliferative disorders. Bruton's Tyrosine Kinase (BTK) is a member of the Tec family of kinases, downstream of the B-cell receptor and is predominantly expressed in B-cells. It is a mediator of B-cell receptor signaling in normal and transformed B-cells. Following binding of antigen to the B-Cell Receptor, kinases such as Syk (Spleen Tyrosine Kinase), Lyn (member of the Src family of protein tyrosine kinases) and BTK (Bruton's Tyrosine Kinase) are activated, with subsequent propagation through PI3K/Akt, MAPK, and NF-κB pathways. This results in B-cell activation and proliferation. IMBRUVICA® (Ibrutinib) is an oral, irreversible inhibitor of BTK and inhibits cell proliferation and promotes programmed cell death (Apoptosis) by blocking B-cell activation and signaling. The FDA initially granted accelerated approval to IMBRUVICA® in February 2014 for previously treated patients with CLL and this was followed by full FDA approval and a new treatment indication for high-risk CLL patients with 17p deletions, in July 2014.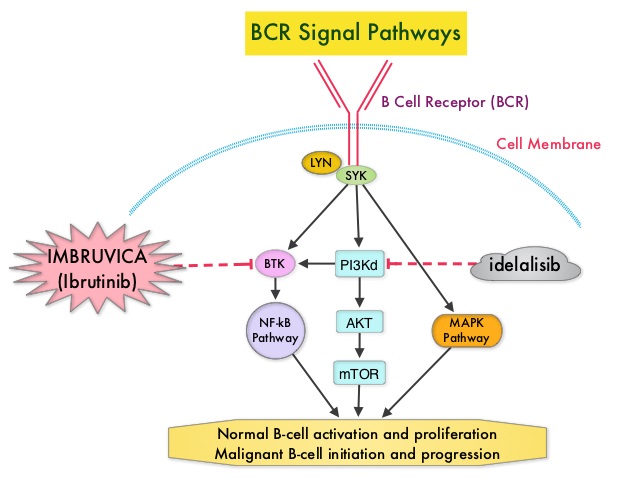
In elderly CLL patients with comorbid conditions, Chlorambucil is often considered as a standard first-line therapy because of the higher rate of toxicities associated with FLUDARA® (Fludarabine) and TREANDA® (Bendamustine). RESONATE-2 is a international, open-label, randomized, phase III trial, in which the efficacy of two oral agents , IMBRUVICA® and Chlorambucil, were compared, in previously untreated elderly patients with CLL or Small Lymphocytic Lymphoma.
In this study, 269 treatment naïve patients with CLL or Small Lymphocytic Lymphoma, who were 65 years of age or older, were randomly assigned in a 1:1 ratio, to receive IMBRUVICA® 420 mg PO once daily (N=136) or Chlorambucil at a dose of 0.5 mg/kg on days 1 and 15 of each 28 day cycle, increased to a maximum of 0.8 mg/kg, if tolerated (N=133). Patients with chromosome del (17p) were excluded. The median age was 73 years and 70% of patients were over age 70. The primary end point was Progression Free Survival (PFS) and secondary end points included, Overall Response Rate (ORR), Overall Survival (OS), and sustained hematologic improvement.
With a median follow-up of 18.4 months, patients in the IMBRUVICA® group had a significantly longer Progression Free Survival (PFS) compared to the Chlorambucil group (median not reached versus 18.9 months), with a risk of progression or death 84% lower with IMBRUVICA®, compared to Chlorambucil (HR=0.16; P<0.001). IMBRUVICA® significantly prolonged Overall Survival, with an estimated Overall Survival rate of 98% at 24 months, compared to 85% with Chlorambucil, with a 84% reduction in the risk of death (HR=0.16; P=0.001). Further, IMBRUVICA® significantly improved Overall Response Rate, compared with Chlorambucil (86% vs 35%; P< 0.001) and also significantly improved hemoglobin and platelets levels from baseline values, compared with Chlorambucil. The most common adverse events associated with IMBRUVICA® were diarrhea, fatigue, cough and nausea and were mostly Grade 1 toxicities. The authors concluded that IMBRUVICA® significantly improves PFS, OS and ORR, compared to Chlorambucil, in previously untreated patients with CLL or Small Lymphocytic Lymphoma, and should be considered for all elderly patients who are not candidates for aggressive systemic therapy. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. Burger JA, Tedeschi A, Barr PM, et al. N Engl J Med 2015; 373:2425-2437

