SUMMARY: Prostate cancer is the most common cancer in American men, excluding skin cancer and 1 in 7 men will be diagnosed with prostate cancer during their lifetime. It is estimated that in the United States, over 230,000 new cases of prostate cancer will be diagnosed in 2014 and close to 30,000 men will die of the disease. Over 90% of the patients with metastatic prostate cancer have bone metastases, and the tumor burden, is an independent predictor of death in this patient population. Agents such as ZOMETA® and XGEVA® can prevent or delay Skeletal Related Events (SRE’s) and External Beam Radiation Therapy (EBRT) is often utilized to treat symptomatic SRE’s. EBRT can however damage the bone marrow in the radiated field, resulting in cytopenias, and consequently can potentially preclude patients from receiving cytotoxic chemotherapy.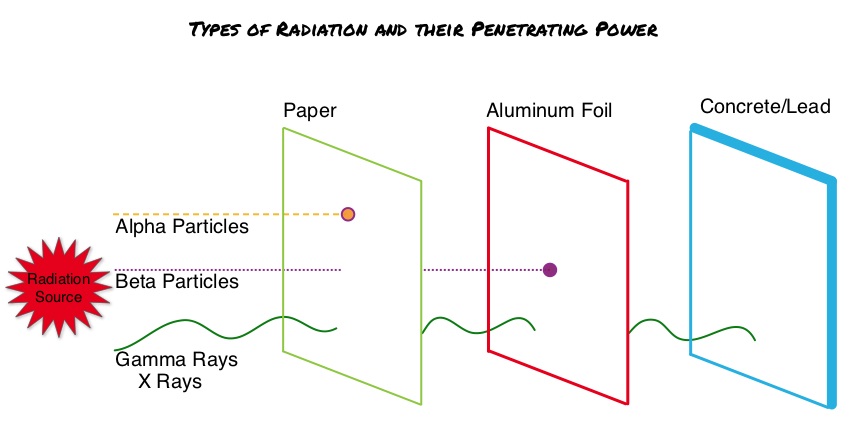 Radium Ra 223 dichloride (XOFIGO®) is a bone seeking alpha particle emitter and by virtue of its chemical similarity to calcium is preferentially taken up by the bone and forms complexes with bone mineral, hydroxyapatite, in areas where there is increased bone turnover, such as bone metastases. XOFIGO® induces double stranded DNA breaks resulting in antitumor effects and has a very short range in tissues (around 2 and 10 cells), quickly losing energy, compared to beta or gamma radiation. The end result is less damage to the adjacent healthy tissues. Further, unlike Ra-226 which was first isolated by Madame Curie, XOFIGO® has a short half life of 11.4 days and rapidly decays, preventing significant radiation exposure. The ALSYMPCA (ALpharadin in SYMptomatic Prostate CAncer patients) study is a randomized, double-blind, phase III trial, in which 921 patients with Castrate Resistant Prostate Cancer (CRPC) with 2 or more symptomatic bone metastases and no known visceral metastases, were randomly assigned in a 2:1 ratio to receive either XOFIGO® along with best supportive care (N=614) or placebo with best supportive care (N=307). Enrolled patients either progressed on or had not received TAXOTERE® (Docetaxel) for a variety of reasons and 42% of the enrolled patients were chemotherapy naïve.
Radium Ra 223 dichloride (XOFIGO®) is a bone seeking alpha particle emitter and by virtue of its chemical similarity to calcium is preferentially taken up by the bone and forms complexes with bone mineral, hydroxyapatite, in areas where there is increased bone turnover, such as bone metastases. XOFIGO® induces double stranded DNA breaks resulting in antitumor effects and has a very short range in tissues (around 2 and 10 cells), quickly losing energy, compared to beta or gamma radiation. The end result is less damage to the adjacent healthy tissues. Further, unlike Ra-226 which was first isolated by Madame Curie, XOFIGO® has a short half life of 11.4 days and rapidly decays, preventing significant radiation exposure. The ALSYMPCA (ALpharadin in SYMptomatic Prostate CAncer patients) study is a randomized, double-blind, phase III trial, in which 921 patients with Castrate Resistant Prostate Cancer (CRPC) with 2 or more symptomatic bone metastases and no known visceral metastases, were randomly assigned in a 2:1 ratio to receive either XOFIGO® along with best supportive care (N=614) or placebo with best supportive care (N=307). Enrolled patients either progressed on or had not received TAXOTERE® (Docetaxel) for a variety of reasons and 42% of the enrolled patients were chemotherapy naïve.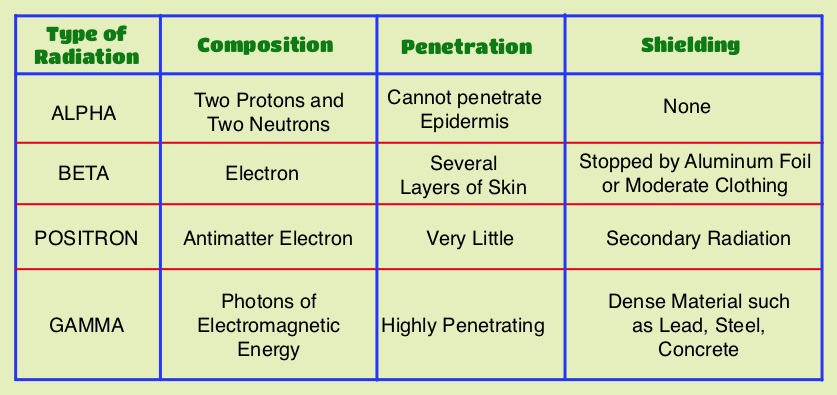 The primary endpoint was Overall Survival and secondary endpoints included time to first symptomatic skeletal event, time to increase in total alkaline phosphatase level and PSA level. This study was stopped earlier than planned, as there was a significant increase in the median overall survival in the XOFIGO® group compared to placebo group, with a 30% reduction in the risk of death (14.9 months vs 11.3 months, HR=0.70, P<0.001). All secondary endpoints favored XOFIGO® as well. The most common adverse events were cytopenias, majority of which were Grades 1 and 2 and discontinuation rates due to adverse events were lower in the XOFIGO® group (17%) compared to 21% for the Placebo group. At 1.5 years, the incidence of myelosuppression was 3%, there were no grade 3 or 4 non-hematologic toxicities and no reports of secondary malignancies in the XOFIGO® group. The authors concluded that based on the long term safety data, XOFIGO® has an excellent side-effect profile in patients with CRPC. Unlike the bone seeking beta emitters such as Strontium-89 and Samarium-153, XOFIGO®, an alpha emitter, is the only agent that has been shown to improve overall survival. Studies are underway evaluating the efficacy of chemotherapy in combination with XOFIGO®, in patients with CRPC and associated bone metastases. Nilsson S, Vogelzang NJ, Sartor AO, et al. J Clin Oncol 32, 2014 (suppl 4; abstr 9)
The primary endpoint was Overall Survival and secondary endpoints included time to first symptomatic skeletal event, time to increase in total alkaline phosphatase level and PSA level. This study was stopped earlier than planned, as there was a significant increase in the median overall survival in the XOFIGO® group compared to placebo group, with a 30% reduction in the risk of death (14.9 months vs 11.3 months, HR=0.70, P<0.001). All secondary endpoints favored XOFIGO® as well. The most common adverse events were cytopenias, majority of which were Grades 1 and 2 and discontinuation rates due to adverse events were lower in the XOFIGO® group (17%) compared to 21% for the Placebo group. At 1.5 years, the incidence of myelosuppression was 3%, there were no grade 3 or 4 non-hematologic toxicities and no reports of secondary malignancies in the XOFIGO® group. The authors concluded that based on the long term safety data, XOFIGO® has an excellent side-effect profile in patients with CRPC. Unlike the bone seeking beta emitters such as Strontium-89 and Samarium-153, XOFIGO®, an alpha emitter, is the only agent that has been shown to improve overall survival. Studies are underway evaluating the efficacy of chemotherapy in combination with XOFIGO®, in patients with CRPC and associated bone metastases. Nilsson S, Vogelzang NJ, Sartor AO, et al. J Clin Oncol 32, 2014 (suppl 4; abstr 9)

