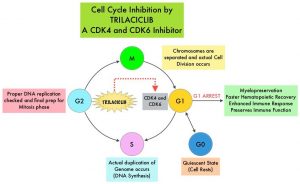
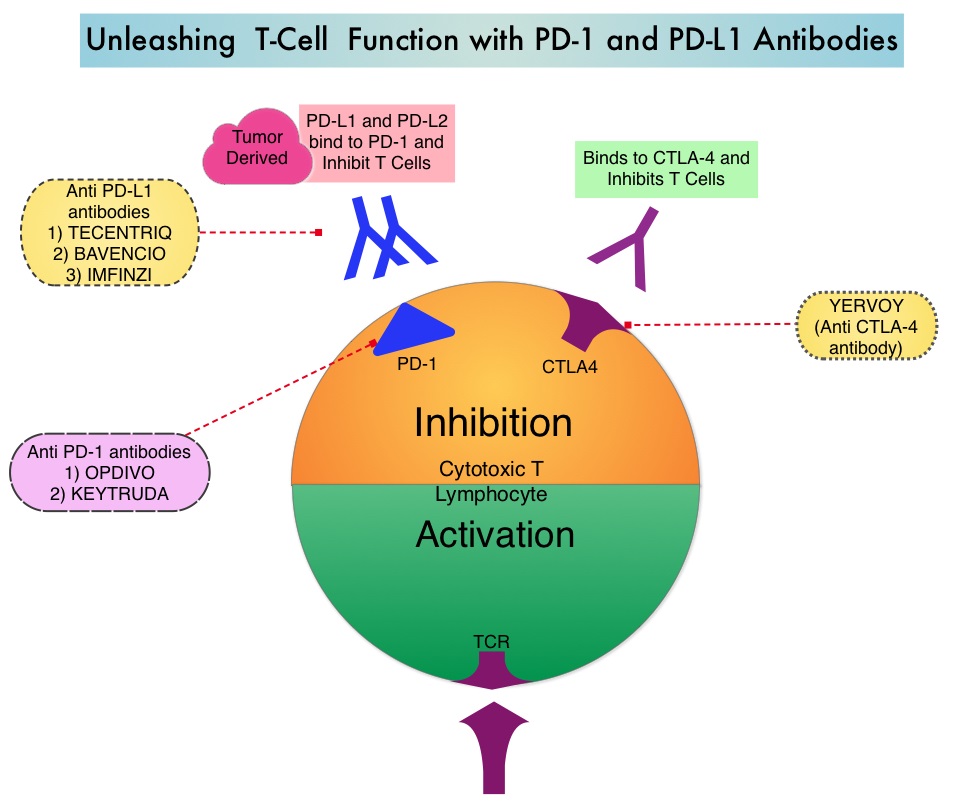
SUMMARY: Small Cell Lung Cancer (SCLC) originates from neuroendocrine cells and accounts for approximately 13-15% of all lung cancers. It is lethal and aggressive. The 5 year survival rate for Extensive Stage SCLC (ES-SCLC) is less than 5%, with a median survival of 9-10 months from the time of diagnosis. Treatment decisions was SCLC are typically based on the VA Lung Group 2-Staging system, which classifies disease as either Limited Stage (LS) or Extensive Stage (ES). In Limited Stage patients, the disease burden is limited to one hemithorax and regional nodes, without presence of extra-thoracic disease, and amenable to definitive-intent thoracic Radiation Therapy (RT). Extensive Stage encompasses all other SCLC patients.
The American Society for Radiation Oncology (ASTRO) developed an evidence-based practice guideline addressing the use of RT for patients with both Limited and ES-SCLC. The ASCO Expert Panel critically appraised the ASTRO guideline, and following minor changes, endorsed the recommendations provided in the ASTRO guideline.
Guideline Questions
♦What are the indications, appropriate dose-fractionation schedules, techniques, and timing of thoracic RT for Limited Stage (LS)-SCLC?
♦What is the role of Stereotactic Body Radiotherapy (SBRT) compared with conventional RT in Stage I or II node-negative SCLC?
♦What are the indications, appropriate dose-fractionation schedules, and timing of prophylactic cranial RT for LS-SCLC and Extensive Stage (ES) SCLC?
♦What are the indications, appropriate dose-fractionation schedules, and timing of thoracic consolidation in patients with ES-SCLC?
RECOMMENDATIONS
Thoracic RT for LS-SCLC
Recommendation 1.1. For patients with LS-SCLC who can tolerate definitive therapy, thoracic RT is recommended.
Recommendation 1.2. For patients with LS-SCLC receiving chemotherapy and RT, thoracic RT should begin with cycle 1 or 2 of chemotherapy. It is important to maintain the dosage and timing of chemotherapy with RT based on trial data. Timing is more critical for accelerated dose-intensive RT including twice-daily, hyperfractionated regimens.
Recommendation 1.3. For postoperative patients with LS-SCLC and R1 or R2 resection, postoperative RT is conditionally recommended.
Recommendation 1.4. For postoperative patients with LS-SCLC that is clinically node-negative and pathologically N2-positive, mediastinal RT is conditionally recommended.
Recommendation 1.5. For patients with LS-SCLC, twice-daily RT in 150-cGy fractions to 4,500 cGy is recommended.
Recommendation 1.6. For patients with LS-SCLC, daily RT in 200-cGy fractions to 6,000-7,000 cGy is conditionally recommended as an acceptable alternative to twice-daily RT.
Recommendation 1.7. For patients with LS-SCLC, involved-field RT is recommended as the standard of care. (defined as FDG avid on PET, enlarged on CT, and/or biopsy-positive)
Recommendation 1.8. For tumors that experience shrinkage with chemotherapy in patients with LS-SCLC, treating all involved nodal stations (at time of diagnosis) and postchemotherapy lung parenchymal tumor is recommended.
Recommendation 1.9. For patients with LS-SCLC, highly conformal techniques are recommended to minimize normal tissue dose.
Role of SBRT in Stage I or II Node-Negative SCLC
Recommendation 2.1. For patients with Stage I or II node-negative LS-SCLC who are medically inoperable, either SBRT or conventional fractionation is recommended. Ideally, the node-negative status should be confirmed by invasive nodal staging. Ultracentral tumors, meaning those with the planning target volume touching or overlapping the proximal bronchial tree, esophagus, or trachea, may be more appropriately treated with conventional fractionation schema.
Recommendation 2.2. For patients with Stage I or II node-negative LS-SCLC receiving SBRT, chemotherapy should be delivered to patients in whom it is medically tolerated.
Prophylactic Cranial RT
Recommendation 3.1. For patients with SCLC who respond to initial therapy, restaging with brain MRI to guide decision making regarding PCI is recommended.
Recommendation 3.2. For patients with Stage I SCLC, PCI is conditionally not recommended. In lieu of PCI, surveillance using brain MRI with contrast can serve as an alternative.
Recommendation 3.3. For patients with Stage II-III LS-SCLC who are less than 70 years of age with good performance status (ECOG 0-2) and respond to thoracic chemoradiation, PCI is recommended.
Recommendation 3.4. For patients with LS-SCLC who have limited Performance Status, older age, and/or significant comorbidities, shared decision making on PCI (considering patient- and disease-specific characteristics) is recommended.
Recommendation 3.5. For patients with LS-SCLC receiving PCI, 2,500 cGy in 10 fractions is recommended.
Recommendation 3.6. For patients with ES-SCLC who respond to chemotherapy, consultation with a Radiation Oncologist to enhance shared decision making on PCI versus MRI surveillance (considering patient- and disease-specific characteristics) is recommended.
Recommendation 3.7. For patients with ES-SCLC who elect PCI, 2,500 cGy in 10 fractions or 2,000 cGy in 5 fractions is recommended.
Thoracic Consolidation for ES-SCLC
Recommendation 4.1. For patients with ES-SCLC with a response to chemotherapy alone but residual tumor in the thorax, thoracic RT is recommended.
Recommendation 4.2. For patients with ES-SCLC with a response to chemotherapy alone, thoracic RT to a dose of 3,000 cGy in 10 fractions is conditionally recommended. In patients expected to have a prolonged survival, higher doses may be appropriate.
Recommendation 4.3. For patients with ES-SCLC who will receive thoracic RT, the treatment should be given after completion of chemotherapy alone.
Recommendation 4.4. For patients with ES-SCLC with a response to chemotherapy and immunotherapy and residual disease in the thorax, thoracic RT to 3,000 cGy in 10 fractions within 6-8 weeks of completion of the chemotherapy prior to maintenance immunotherapy, is conditionally recommended.
Radiation Therapy for Small-Cell Lung Cancer: ASCO Guideline Endorsement of an ASTRO Guideline. Daly ME, NIsmaila N , Decker RH, et al. J Clin Oncol. 2021;39:931-939.