Elacestrant in ER-Positive, HER2-Negative, Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors. The most common subtype of metastatic breast cancer is Hormone Receptor-positive (HR-positive), HER2-negative breast cancer (65% of all metastatic breast tumors), and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients, with a median Overall Survival (OS) of 36 months. With the development of Cyclin Dependent Kinases (CDK) 4/6 inhibitors, endocrine therapy plus a CDK4/6 inhibitor is the mainstay, for the management of ER+/HER2-negative metastatic breast cancer, as first line therapy. Even with this therapeutic combination, most patients will eventually experience disease progression, including the development of ESR1 (Estrogen Receptor gene alpha) mutations.
ESR1 is the most common acquired mutation noted in breast tumors as they progress from primary to metastatic setting. These mutations promote ligand independent Estrogen Receptor activation and have been shown to promote resistance to estrogen deprivation therapy. It appears that ESR1 mutations are harbored in metastatic ER-positive breast cancers with prior Aromatase Inhibitor (AI) therapy, but not in primary breast cancers, suggesting that ESR1 mutations may be selected by prior therapy with an AI in advanced breast cancer. In a previously published study (JAMA Oncol.2016;2:1310-1315), ESR1 mutations Y537S and D538G mutations detected in baseline plasma samples from ER+/HER- advanced breast cancer patients, was associated with shorter Overall Survival. In this study it was noted that there was a three-fold increase in the prevalence of these mutations in patients who had failed first line hormonal therapy for metastatic disease, compared with those who were initiating first line therapy for advanced breast cancer (33% versus 11%).
Fulvestrant is a parenteral, Selective Estrogen Receptor Degrader (SERD) and is the only SERD approved for the treatment of postmenopausal women with HR-positive metastatic breast cancer. However, acquired ESR1 mutations can also occur following Fulvestrant treatment, possibly because of poor bioavailability and incomplete ER blockade when administered intramuscularly. There is therefore an urgent unmet need for an alternate SERD that has activity in tumors harboring ESR1 mutations, and has improved bioavailability allowing oral administration.
Elacestrant is an oral, nonsteroidal, Selective Estrogen Receptor Degrader (SERD) that degrades the Estrogen Receptor (ER) in a dose-dependent manner and inhibits estradiol-dependent functions of ER target gene transcription induction and breast cancer cell proliferation. Estradiol-stimulated tumor growth was diminished by Elacestrant in the ER+ xenograft models derived from heavily pretreated patients, including models resistant to CDK 4/6 inhibitors, Fulvestrant and those harboring ESR1 mutations Y537S and D538G. In an early Phase I trial, Elacestrant was noted to have an acceptable safety profile, and demonstrated single-agent activity with confirmed Partial Responses in heavily pretreated patients with ER+ metastatic breast cancer.
EMERALD trial is a multicenter, International, randomized, open-label, Phase III study, designed to evaluate the benefit of Elacestrant in patients with ER+/HER2- advanced or metastatic breast cancer. In this study, 477 postmenopausal women with ER+/HER2- metastatic breast cancer were randomly assigned 1:1 to receive either Elacestrant 400 mg orally daily (N=239) or the Standard of Care which included investigator’s choice of Fulvestrant or an Aromatase Inhibitor including Anastrozole, Letrozole, or Exemestane (N=238). Treatment was given until disease progression. Both treatment groups were well balanced. The median patient age was 63 years, and patients must have progressed or relapsed on or after 1 or 2 lines of endocrine therapy for advanced disease, one of which was given in combination with a CDK4/6 inhibitor, had 1 or fewer lines of chemotherapy for advanced disease, and had an ECOG performance status of 0 or 1. In the study, 48% had tumors with mutated ESR1 and 43% received two prior endocrine therapies. These patients were evenly distributed in both treatment groups. Patients were stratified by ESR1-mutation status, prior treatment with Fulvestrant, and visceral metastases. The co-Primary end points were Progression Free Survival (PFS) in the overall population, and in those with ESR1 mutations. Overall Survival (OS) was a Secondary end point.
Treatment with Elacestrant resulted in a statistically significant and clinically meaningful improvement in PFS, compared with Standard of Care. There was a 30% reduction in the risk of progression or death in the Elacestrant group for all patients (HR=0.70; P=0.002) and a 45% reduction in the risk of progression or death among those with ESR1 mutations (HR=0.55; P=0.0005). The researchers in this study used landmark analysis of PFS at 6 months and 12 months which selects for patients who are still sensitive to endocrine therapy and addresses the limited PFS benefit caused by an initial progression, in patients with complete endocrine resistance who do not respond to endocrine therapy. The PFS at 12 months with Elacestrant was 22.3% in all patients compared with 9.4% for those receiving the Standard of Care treatment. Among the ESR1 mutation group, the 12 month PFS rate was more pronounced and was 26.8% with Elacestrant, compared to 8.2% with Standard of Care. The benefits with Elacestrant compared with Standard of Care, was consistent across multiple prespecified subgroups including patients who had received prior Fulvestrant. There also was a trend toward improved Overall Survival in patients who received Elacestrant, compared with Standard of Care. The final Overall Survival data were not mature at the time of this analysis. Nausea of any grade occurred in 35% of patients receiving Elacestrant and 18.8% receiving Standard of Care treatment, and treatment discontinuations due to adverse events were 3.4% in the Elacestrant group versus 0.9% in the Standard of Care group.
It was concluded that Elacestrant is the first oral Selective Estrogen Receptor Degrader that demonstrated significant and clinically meaningful improvement in PFS, compared with Standard of Care endocrine therapy, in patients with ER+/ HER2- metastatic breast cancer, in the second/third line after treatment with a CDK4/6 inhibitor, and has the potential to become the new standard of care in this study population.
Elacestrant (oral selective estrogen receptor degrader) Versus Standard Endocrine Therapy for Estrogen Receptor–Positive, Human Epidermal Growth Factor Receptor 2–Negative Advanced Breast Cancer: Results From the Randomized Phase III EMERALD Trial. Bidard F-C, Kaklamani VG, Neven P, et al. DOI: 10.1200/JCO.22.00338 Journal of Clinical Oncology. Published online May 18, 2022.
Late Breaking Abstract - ASCO 2022: ENHERTU® for HER2-Low Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence.
It is estimated that approximately 60% of metastatic breast cancers categorized as HER2-negative express low levels of HER2, defined as a score of 1+ on ImmunoHistoChemical (IHC) analysis or as an IHC score of 2+ and negative results on In Situ Hybridization (ISH). These HER2-low breast cancer tumors are treated as HER2-negative, as currently available HER2-directed therapies have resulted in poor outcomes. These patients have limited targeted treatment options and are often treated with single agent palliative chemotherapy following progression on first line chemotherapy.
ENHERTU® (Trastuzumab Deruxtecan) is an Antibody-Drug Conjugate (ADC) composed of a humanized monoclonal antibody specifically targeting HER2, with the amino acid sequence similar to Trastuzumab, a cleavable tetrapeptide-based linker, and a potent cytotoxic Topoisomerase I inhibitor as the cytotoxic drug (payload). ENHERTU® has a favorable pharmacokinetic profile and the tetrapeptide-based linker is stable in the plasma and is selectively cleaved by cathepsins that are up-regulated in tumor cells. Unlike KADCYLA® (ado-Trastuzumab emtansine), ENHERTU® has a higher drug-to-antibody ratio (8 versus 4), released payload easily crosses the cell membrane with resulting potent cytotoxic effect on neighboring tumor cells regardless of target expression, and the released cytotoxic agent (payload) has a short half-life , thus minimizing systemic exposure. The potential activity of ENHERTU® in HER2-low breast cancer tumors is driven by the bystander antitumor effect, offered by the optimized ADC technology. Previously published Phase I and II trials have shown that ENHERTU® in heavily pretreated patients with HER2-low metastatic breast cancer resulted in an Overall Response Rate of 37%, and median Progression Free Survival ranging from 6.3 to 11.1 months.
DESTINY-Breast04 is a multicenter, randomized, open-label, Phase III trial, conducted to evaluate the efficacy and safety of ENHERTU® as compared with the physician’s choice of chemotherapy, in patients with HER2-low metastatic breast cancer. In this study, patients were randomly assigned in a 2:1 ratio to receive ENHERTU® 5.4 mg/kg IV every 3 weeks (N=373) or the physician’s choice of Capecitabine, Eribulin, Gemcitabine, Paclitaxel, or Nab-paclitaxel (N=184). Low expression of HER2 was defined as a score of 1+ on ImmunoHistoChemical (IHC) analysis or as an IHC score of 2+ and negative results on In Situ Hybridization (ISH). Randomization was stratified according to HER2-low status (IHC 1+ versus IHC 2+ and ISH-negative), the number of previous lines of chemotherapy for metastatic disease (one versus two), and Hormone Receptor (HR) status (positive versus negative) and if positive, previous CDK4/6 inhibitor therapy versus no CDK4/6 inhibitor therapy. IHC scores for HER2 expression were determined through central testing with the use of VENTANA HER2/neu investigational assay system, according to an algorithm adapted from the 2018 ASCO/CAP testing guidelines. Eligible patients must have received chemotherapy for metastatic disease or have had disease recurrence during or within 6 months after completing adjuvant chemotherapy. Patients with Hormone Receptor positive (HR-positive) disease must have received at least one line of endocrine therapy. Patients with treated, stable brain metastases were eligible. Patients were ineligible if they had a history of noninfectious interstitial lung disease treated with steroids or had suspected interstitial lung disease on imaging at screening. Both treatment groups were well balanced and approximately 89% in the ENHERTU® group and 90% in the chemotherapy group were HR-positive. The Primary end point was Progression Free Survival (PFS) among patients with HR-positive disease. Secondary end points included PFS among all patients, Overall Survival (OS) in the HR-positive cohort and among all patients, Objective Response Rate (ORR), Duration of Response, and efficacy in the HR-negative cohort. The median duration of follow up for survival was 18.4 months.
At the time of the primary efficacy analysis, the median PFS in the HR-positive cohort was 10.1 months in the ENHERTU® group and 5.4 months in the physician’s choice group (HR for disease progression or death=0.51; P<0.001). This benefit with ENHERTU® was seen consistently across all analyzed subgroups which included HER2 IHC 1+, HER2 IHC 2+ and ISH-negative, as well as those who had received previous treatment with CDK4/6 inhibitors. The median PFS among all patients was 9.9 months in the ENHERTU® group and 5.1 months in the physician’s choice group (HR for disease progression or death=0.50; P<0.001). The median PFS in the HR-negative cohort was 8.5 months in the ENHERTU® group and 2.9 months in the physician’s choice group (HR=0.46).
The median OS in the HR-positive cohort was 23.9 months in the ENHERTU® group and 17.5 months in the physician’s choice group (HR for death=0.64; P=0.003). The median OS among all patients was 23.4 months in the ENHERTU® group and 16.8 months in the physician’s choice group HR=0.64; P=0.001). The median OS in the HR-negative cohort was 18.2 months in the ENHERTU® group and 8.3 months in the physician’s choice group (HR=0.48).
The ORR in the HR-positive group was 52.6% in the ENHERTU® group and 16.3% in the physician’s choice group, and the median duration of response was 10.7 months in the ENHERTU® group and 6.8 months in the physician’s choice group. The ORR among all patients was 52.3% in the ENHERTU® group and 16.3% in the physician’s choice group. Among HR-negative cohort, the ORRs were 50% and 16.7% respectively.
Grade 3 or higher adverse events occurred in 53% of the patients who received ENHERTU® and 67.4% of those who received the physician’s choice of chemotherapy. Adjudicated, drug-related Interstitial Lung Disease or pneumonitis occurred in 12.1% of the patients who received ENHERTU®.
The authors concluded that this is the first Phase III, practice-changing trial of a HER2-directed therapy in patients with HER2-low metastatic breast cancer, to show a statistically significant and clinically meaningful benefit in PFS and OS, compared to standard chemotherapy, regardless of Hormone Receptor status, with a generally manageable safety profile. The authors added that the strong efficacy of ENHERTU® in this HER2-low patient population, with approximately 50% lower risk of disease progression and 36% lower risk of death with ENHERTU® compared to standard chemotherapy, supports the need to reclassify HER2-low as a new targetable category of metastatic breast cancer.
Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. Modi S, Jacot W, Yamashita T, et al. for the DESTINY-Breast04 Trial Investigators. N Engl J Med 2022; 387:9-20.
Late Breaking Abstract - ASCO 2022: KISQALI® with Switch Endocrine Therapy Following Progression on a Prior CDK4/6 Inhibitor in HR+/HER2-negative Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors in patients with metastatic disease are Estrogen Receptor (ER) and/or Progesterone Receptor (PR) positive and HER2-negative. These patients are often treated with single agent endocrine therapy, endocrine therapy in combination with CDK4/6 inhibitor, or single agent chemotherapy.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation and associated with increased expression of CDK4. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6, preferentially inhibiting CDK4 that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest.
MAINTAIN trial is an investigator-initiated, multicenter, Phase II, double-blind, placebo-controlled, prospective randomized study, conducted to evaluate the efficacy of Fulvestrant or Exemestane with or without KISQALI®, in patients with HR+/HER2-negative metastatic breast cancer, who had previously progressed on any CDK 4/6 inhibitor plus any endocrine therapy. In this study, 119 evaluable patients were randomized 1:1 to receive either KISQALI® 600mg orally once daily given 3 weeks on and 1 week off plus switch endocrine therapy (N= 60) or placebo plus switch endocrine therapy (N=59). Patients treated with prior Fulvestrant received Exemestane as endocrine therapy in the randomization, whereas if prior Exemestane was endocrine therapy, patients received Fulvestrant. If patients received neither as prior endocrine therapy, Fulvestrant or Exemestane was given per investigator discretion, although Fulvestrant was encouraged. Ultimately, 83% of patients received Fulvestrant and 17% received Exemestane. Eligible patients were postmenopausal, had HR+/HER2- negative metastatic breast cancer and had progressed on prior endocrine therapy and any CDK4/6 inhibitor. With regards to prior CDK 4/6 inhibitor treatment, 84% received IBRANCE® (Palbociclib), 11% received KISQALI®, 2% received VERZENIO® (Abemaciclib) and 3% received IBRANCE® and another CDK 4/6 inhibitor. The median duration of treatment with the prior CDK4/6 inhibitor was 15.5 months in the KISQALI® group and 17 months in the placebo group. Approximately 60% of patients had visceral metastasis, 45% had de novo metastasis at diagnosis, 18% had bone-only disease, 18% had received 2 or more prior endocrine therapies for metastatic disease, and 9% had received chemotherapy. The Primary end point was Progression Free Survival (PFS) and Secondary end points included Overall Response Rate (ORR), Clinical Benefit Rate, safety, and tumor and blood biomarkers. The median follow up was 18.2 months.
There was a statistically significant PFS improvement for patients randomized to KISQALI® plus endocrine therapy and the median PFS for patients in the KISQALI® plus endocrine therapy was 5.33 months, compared with 2.76 months for patients receiving placebo and endocrine therapy (HR=0.56;; P=0.004). At 12 months, the PFS rates were 24.6% in the KISQALI® group versus 7.4% in the placebo group. Similar results were noted in the subset of patients treated with Fulvestrant and KISQALI®, and the median PFS for those randomized to KISQALI® was 5.29 months versus 2.76 months in the placebo group (HR=0.59; P=0.02). The PFS benefit was more evident in KISQALI® group compared to the placebo group, especially among those who received a shorter duration of therapy with a prior CDK4/6 inhibitor (HR=0.36) and among those over age 65 years (HR=0.31). The Overall Response Rate in the KISQALI® group was 20%, compared to 11% in the placebo group, and the median Duration of Response was 18.8 months in those treated with KISQALI® and endocrine therapy, compared with 14.8 months for those treated with placebo plus endocrine therapy. There was also a significant improvement in the Clinical Benefit Rate (CBR), defined as patients who achieved Complete Response, Partial Response, or stable disease lasting at least 24 weeks. The CBR was significantly improved in the KISQALI® group, compared with the placebo group, and was 43% versus 25%, respectively (P=0.06). The most common adverse event in the KISQALI® group was neutropenia at 72%, compared to 15% in the placebo group.
It was concluded from this randomized, placebo-controlled trial that, treatment with KISQALI® and an alternate endocrine therapy, after progression on a prior CDK4/6 inhibitor, showed a 43% reduction in the risk of progression or death, compared with placebo and endocrine therapy, in patients with HR+/HER2-negative metastatic breast cancer.
A randomized, phase II trial of fulvestrant or exemestane with or without ribociclib after progression on anti-estrogen therapy plus cyclin-dependent kinase 4/6 inhibition (CDK 4/6i) in patients (pts) with unresectable or hormone receptor–positive (HR+), HER2-negative metastatic breast cancer (MBC): MAINTAIN trial. Kalinsky K, Accordino MK, Chiuzan C, et al. J Clin Oncol 40, 2022 (suppl 17; abstr LBA1004). DOI: 10.1200/JCO.2022.40.17_suppl.LBA1004.
Late Breaking Abstract – ASCO 2022: Survival Benefit with TRODELVY® in Hormone Receptor Positive/HER2-Negative Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors in patients with metastatic disease are Estrogen Receptor (ER) and/or Progesterone Receptor (PR) positive and HER2-negative. These patients are often treated with single agent endocrine therapy, endocrine therapy in combination with CDK4/6 inhibitor, or single agent chemotherapy. Resistance to hormonal therapy occurs in a majority of the patients and there is therefore an unmet need for agents with novel mechanisms of action.
TRODELVY® (Sacituzumab govitecan) is an Antibody-Drug Conjugate (ADC) in which SN-38, an active metabolite of Irinotecan, a Topoisomerase I inhibitor, is coupled to the humanized Anti-Trophoblast cell-surface antigen 2 (Trop-2) monoclonal antibody (hRS7 IgG1κ), through the cleavable CL2A linker. SN-38 cannot be given directly to patients because of its toxicity and poor solubility. Trop-2, a transmembrane calcium signal transducer, stimulates cancer-cell growth, and this cell surface receptor is overexpressed in several epithelial cancers including cancers of the breast, colon and lung, and has limited expression in normal human tissues. Trop-2 is expressed in more than 85% of breast tumors including Triple Negative Breast Cancer. Upon binding to Trop-2, the anti-TROP-2 monoclonal antibody is internalized and delivers SN-38 directly into the tumor cell, making it a suitable transporter for the delivery of cytotoxic drugs. Further, the cleavable linker enables SN-38 to be released both intracellularly into the tumor cells, as well as the tumor microenvironment, thereby allowing for the delivery of therapeutic concentrations of the active drug in bystander cells to which the conjugate has not bound. Thus, TRODELVY®-bound tumor cells are killed by intracellular uptake of SN-38, whereas the adjacent tumor cells are killed by the extracellular release of SN-38.
TRODELVY® was approved by the FDA in 2021 for patients with unresectable, locally advanced or metastatic Triple Negative Breast Cancer, who have received two or more prior systemic therapies, at least one of them for metastatic disease. In the IMMU-132 Phase I/II study, the Hormone Receptor positive (HR+)/HER2-negative cohort of patients with metastatic breast cancer patients had an Objective Response Rate (ORR) of 31.5%, median Progression Free Survival (PFS) of 5.5 months and median Overall Survival (OS) of 12 months, with manageable toxicities, when treated with TRODELVY®.
TROPiCS-02 is a global, open-label, randomized, Phase III study, conducted to confirm the benefit of TRODELVY® in HR+/HER2- negative advanced breast cancer. In this study, 543 patients with HR+/HER2-negative, unresectable, locally advanced or metastatic breast cancer, were randomly assigned 1:1 to receive TRODELVY® 10 mg/kg IV on D1 and 8, every 21 days (N=272), or treatment of physician’s choice, which included single agent treatment with either Capecitabine, Eribulin, Vinorelbine, or Gemcitabine (N=271). Treatment was continued until disease progression or unacceptable toxicity. Both treatment groups were well balanced. Eligible patients had 3 median prior chemotherapy regimens for metastatic breast cancer, and one prior therapy for metastatic breast cancer was allowed if disease progressed in 12 months or less after neoadjuvant chemotherapy. Patients were required to have received endocrine therapy, a CDK4/6 inhibitor and at least one prior therapy with a Taxane in any setting. Majority of patients had visceral metastases (95%), 86% had prior endocrine therapy for metastatic breast cancer for at least 6 months, and 60% and 38% received prior CDK4/6 inhibitors for 12 months or less, and for more than 12 months, respectively. The Primary endpoint was Progression Free Survival (PFS) by blinded Independent Central Review (final analysis) and key Secondary endpoint was Overall Survival (OS) at the first planned interim analysis.
The median Progression Free Survival was 5.5 months with TRODELVY® versus 4 months with standard chemotherapy (HR=0.66; P=0.0003), representing a 34% improvement in PFS with TRODELVY®. This benefit was seen across all treatment subgroups including those who were 65 years or older, those who were heavily pretreated, as well as those with visceral metastases. The Objective Response Rate (ORR) was 21% with TRODELVY® versus 14% with standard chemotherapy. The Clinical Benefit Rate was also higher with TRODELVY® versus standard chemotherapy (34% versus 22%) and median duration of response was 7.4 months and 5.6 months respectively. Overall Survival data were immature, but there was a numerical, non-significant improvement in the median Overall Survival noted in the TRODELVY® group, compared to the standard chemotherapy group (13.9 months versus 12.3 months; HR=0.84; P=0.14), respectively. Treatment with TRODELVY® also resulted in an overall health-related Quality of Life benefit over chemotherapy, with delayed deterioration in fatigue and global health status/ Quality of Life scales, according to the researchers. Grade 3 or more adverse events were observed in 74% of patients receiving TRODELVY® and in 60% of those receiving chemotherapy, and the most common toxicities associated with TRODELVY® were diarrhea and neutropenia.
It was concluded from this landmark analysis that treatment with TRODELVY® resulted in a statistically significant and clinically meaningful improvement in Progression Free Survival, compared to standard chemotherapy, in heavily pre-treated patients with HR+/HER2-negative, endocrine-resistant, unresectable, locally advanced or metastatic breast cancer, and should therefore be considered as a new treatment option for this patient population.
Primary results from TROPiCS-02: A randomized phase 3 study of sacituzumab govitecan (SG) versus treatment of physician’s choice (TPC) in patients (Pts) with hormone receptor–positive/HER2-negative (HR+/HER2-) advanced breast cancer. Rugo HS, Bardia A, Marmé F, et al. J Clin Oncol 40, 2022 (suppl 17; abstr LBA1001)
Late Breaking Abstract – ASCO 2022: Adjuvant Radiotherapy May Be Omitted in Select Patients with Luminal A Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence.
Patient undergoing breast conserving surgery, often receive adjuvant breast radiation therapy to reduce the risk of local recurrence. Radiation therapy however is inconvenient, expensive and is associated with acute and late toxicities. Previously published study by Kunkler IH, et al. (Lancet Oncol. 2015;16:266-273) concluded that radiotherapy could be avoided in a subset of elderly patients with low risk breast cancer following breast conserving surgery.
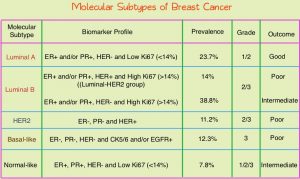 Conventional clinical pathological factors have limited ability to identify breast cancer patients with low risk disease, who could avoid radiation therapy. Molecular defined intrinsic subtypes of breast cancer can provide additional prognostic information. Breast cancer is heterogeneous malignancy and using global gene expression analyses, 5 breast cancer intrinsic subtypes have been established. They include Luminal A, Luminal B, HER2-enriched, Basal-like, and Normal breast-like group. Luminal A breast cancer patients have the lowest risk of recurrence. In a retrospective analysis of women over age 60 years, with Luminal A, Grade 1-2, T1N0 breast cancer, treated with breast conserving surgery and endocrine therapy alone, the local recurrence rate was low (JCO 2015; 33:2035). However, the utility of combining molecular subtype (Luminal A subtype) with clinical pathological factors, to guide radiotherapy decision-making, has not been prospectively evaluated.
Conventional clinical pathological factors have limited ability to identify breast cancer patients with low risk disease, who could avoid radiation therapy. Molecular defined intrinsic subtypes of breast cancer can provide additional prognostic information. Breast cancer is heterogeneous malignancy and using global gene expression analyses, 5 breast cancer intrinsic subtypes have been established. They include Luminal A, Luminal B, HER2-enriched, Basal-like, and Normal breast-like group. Luminal A breast cancer patients have the lowest risk of recurrence. In a retrospective analysis of women over age 60 years, with Luminal A, Grade 1-2, T1N0 breast cancer, treated with breast conserving surgery and endocrine therapy alone, the local recurrence rate was low (JCO 2015; 33:2035). However, the utility of combining molecular subtype (Luminal A subtype) with clinical pathological factors, to guide radiotherapy decision-making, has not been prospectively evaluated.
LUMINA is a prospective multicenter single-arm, cohort study, in which 501 women, 55 years and older, who had undergone breast conserving surgery for breast cancer, were enrolled. Eligible patients had invasive ductal T1N0, Grade 1-2, Luminal A breast cancer, had undergone breast conserving surgery, with excision margins of at least 1 mm and sentinel lymph node biopsy, omitted radiotherapy, and had received adjuvant endocrine therapy for at least 5 years. Luminal A subtype was defined as ER 1% or more, PR more than 20%, HER2 negative and Ki67 13.25% or less. Ki67 immunohistochemistry was performed centrally in one of three Canadian laboratories using International Ki67 Working Group methods. The median patient age was 67 years, 66% had Grade 1 tumors, 88% of patients were less than 75 years, and the median tumor size was 1.1 cm. Patients were followed every six months for the first two years and then yearly. The Primary outcome was local recurrence defined as time from enrollment to any invasive or non-invasive cancer in the ipsilateral breast. Secondary endpoints included contralateral breast cancer, Relapse Free Survival (RFS) based on any recurrence, Disease Free Survival, and Overall Survival.
At a median follow up of 5 years, the local recurrence rate was 2.3% and the rate of contralateral breast cancer was 1.9%. The 5-year Relapse Free Survival, Disease Free Survival and Survival rate was 97.3%, 89.9% and 97.2% respectively.
The authors concluded that among women 55 years of age and over, with low grade Luminal A breast cancer, omission of radiation therapy following breast conserving surgery and treatment with endocrine therapy alone for 5 years or more, resulted in very low rates of local recurrence at 5 years. The researchers added that approximately 30,000-40,000 women per year in North America, predominantly in the US, could avoid the morbidity, expense, and inconvenience of radiotherapy.
LUMINA: A prospective trial omitting radiotherapy (RT) following breast conserving surgery (BCS) in T1N0 luminal A breast cancer (BC). Whelan TJ, Smith S, Nielsen TO, et al. J Clin Oncol. 2022;40(suppl 17):LBA501. doi:10.1200/JCO.2022.40.17_suppl.LBA501
FDA Grants Regular Approval to ENHERTU® for Breast Cancer
SUMMARY: The FDA on May 4, 2022, approved ENHERTU® (Trastuzumab Deruxtecan) for adult patients with unresectable or metastatic HER2-positive breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within 6 months of completing therapy. In 2019, ENHERTU® received accelerated approval for adult patients with unresectable or metastatic HER2-positive breast cancer who have received two or more prior anti-HER2-based regimens in the metastatic setting. The following trial was the confirmatory trial for the accelerated approval. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence.
The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA® given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.
ENHERTU® (Trastuzumab Deruxtecan) is an Antibody-Drug Conjugate (ADC) composed of a humanized monoclonal antibody specifically targeting HER2, with the amino acid sequence similar to Trastuzumab, a cleavable tetrapeptide-based linker, and a potent cytotoxic Topoisomerase I inhibitor as the cytotoxic drug (payload). ENHERTU® has a favorable pharmacokinetic profile and the tetrapeptide-based linker is stable in the plasma and is selectively cleaved by cathepsins that are up-regulated in tumor cells. Unlike KADCYLA®, ENHERTU® has a higher drug-to-antibody ratio (8 versus 4), released payload easily crosses the cell membrane with resulting potent cytotoxic effect on neighboring tumor cells regardless of target expression, and the released cytotoxic agent (payload) has a short half-life , thus minimizing systemic exposure. In the DESTINY-Breast 01 Phase II registration trial involving patients with HER2-positive metastatic breast cancer, who had received two or more prior HER2 targeted therapies including KADCYLA®, the Objective Response Rate (ORR) was 60.9%, with 6% Complete Responses and 54.9% Partial Response, with a median response duration of 14.8 months. The median PFS was 16.4 months. This benefit was consistent across all key subgroups, including patients who had previously received PERJETA® therapy.
The present FDA approval was based on DESTINY-Breast 03, which is a global, multicenter, open-label, randomized Phase III study, in which the efficacy and safety of ENHERTU® was compared with KADCYLA®, in patients with HER2-positive metastatic breast cancer previously treated with Trastuzumab and a Taxane or developed disease recurrence during or within 6 months of completing neoadjuvant or adjuvant therapy. In this study, 524 pts were randomized 1:1 to receive ENHERTU® 5.4 mg/kg (N=261) or KADCYLA® 3.6 mg/kg (N=263) once every 3 weeks. Randomization was stratified by hormone receptor status, prior treatment with Pertuzumab, and history of visceral disease. The median patient age was 54 years and patients in both treatment groups were comparable in terms of baseline characteristics including age, HER2-positivity status, ECOG Performance Status, prior treatment for breast cancer, brain metastases, and prior cancer therapy with agents including Trastuzumab. The Primary endpoint was Progression Free Survival (PFS) by Blinded Independent Central Review (BICR). Secondary endpoints include Overall Survival (OS), Objective Response Rate (ORR), Duration of Response, PFS by investigator, and Safety.
At the time of the prespecified interim analysis of this study, the median follow up was approximately 16 months and the median PFS by BICR review was Not Reached with ENHERTU® and was 6.8 months with KADCYLA® (HR=0.28; P= <0.0001). This represented a very statistically significant 72% reduction in the risk for progression or death with ENHERTU® compared to KADCYLA®. The investigator-assessed PFS was similar (25.1 versus 7.2 months, HR=0.26, P<0.0001). This PFS benefit was observed as early as 4 weeks and remained consistent throughout the follow up period. PFS was significantly higher with ENHERTU® in all prespecified key subgroups, including Hormone Receptor status, prior treatment with PERJETA®, visceral disease, number of prior lines of therapy, and the presence or absence of brain metastases. Majority of patients in the ENHERTU® group experienced a reduction in tumor size, and the ORR was significantly higher among patients in the ENHERTU® compared to those who received KADCYLA® (82.7% versus 36.1%; P<0.0001), with a near doubling of the Complete Response rate in the ENHERTU® group, at 16.1% compared to 8.7% in the KADCYLA® group. The estimated 12-month Overall Survival rate was 94.1% versus 85.9% respectively (HR=0.56; P=0.007), but was not considered significant as it did not cross the prespecified boundary for significance, likely due to the immaturity of the dataset.
Adjudicated treatment related Interstitial Lung Disease/pneumonitis was more common in the ENHERTU® compared with the KADCYLA® treatment arm, at rates of 10.5% and 1.9%, respectively and most of the events were Grade 1 or 2 in severity, and none at Grade 4 or 5 in either treatment group. Interstitial Lung Disease profile was of less concern, than was seen in previous trials of ENHERTU® in more heavily pretreated patients. All Left Ventricular Ejection Fraction decreases were Grade 1 or 2 and were seen in 2.7% of the ENHERTU® group and in 0.4% of KADCYLA® group. Other serious adverse reactions in patients who received ENHERTU® included, vomiting, pyrexia, and urinary tract infection.
The researchers concluded that ENHERTU® demonstrated a highly statistically significant and clinically meaningful improvement in Progression Free Survival, when compared to KADCYLA®, in patients previously treated with Trastuzumab and Taxane for HER2-positive metastatic Breast cancer, with manageable toxicities.
Trastuzumab deruxtecan (T-DXd) vs trastuzumab emtansine (T-DM1) in patients (Pts) with HER2+ metastatic breast cancer (mBC): Results of the randomized phase III DESTINY-Breast03 study. Cortés J, Kim SB, Chung WP, et al. Presented at: European Society for Medical Oncology 2021 Virtual Congress. September 16-21, 2021; virtual. Abstract LBA1.
TUKYSA® in Pretreated HER2-positive Metastatic Breast Cancer With and Without Brain Metastases: Final Overall Survival Analysis
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence.
The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab), KADCYLA® (ado-Trastuzumab emtansine), ENHERTU® (Trastuzumab deruxtecan) and MARGENZA® (Margetuximab). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2-positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.
It is estimated that close to 50% of patients with HER2-positive metastatic breast cancer develop brain metastases. Systemic HER2-targeted agents, including Tyrosine Kinase Inhibitors, as well as chemotherapy have limited antitumor activity in the brain. This is therefore an area of high unmet need. Local therapeutic interventions for brain metastases include neurosurgical resection and Stereotactic or Whole-Brain Radiation Therapy.
TUKYSA® (Tucatinib) is an oral Tyrosine Kinase Inhibitor that is highly selective for the kinase domain of HER2 with minimal inhibition of Epidermal Growth Factor Receptor. In a Phase 1b dose-escalation trial, TUKYSA® in combination with HERCEPTIN® and XELODA® (Capecitabine) showed encouraging antitumor activity in patients with HER2-positive metastatic breast cancer, including those with brain metastases.
HER2CLIMB is an international, randomized, double-blind, placebo-controlled trial in which the combination of TUKYSA® plus HERCEPTIN® and XELODA® was compared with placebo plus HERCEPTIN® and XELODA®. A total of 612 patients with unresectable locally advanced or metastatic HER2-positive breast cancer, who were previously treated with HERCEPTIN®, PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine) were enrolled. Patients were randomly assigned in a 2:1 ratio to receive either TUKYSA® 300 mg orally twice daily throughout the treatment period (N=410) or placebo orally twice daily (N=201), in combination with HERCEPTIN® 6 mg/kg IV once every 21 days, following an initial loading dose of 8 mg/kg, and XELODA® 1000 mg/m2 orally twice daily on days 1 to 14 of each 21-day cycle. Stratification factors included presence or absence of brain metastases, ECOG Performance Status and geographic region. The median patient age was 54 years and patient demographic as well as disease characteristics at baseline were well balanced between the two treatment groups. In the total treatment population, 47.5% had brain metastases at baseline, 48.3% in the TUKYSA® combination group and 46% in the placebo combination group. The Primary endpoint was Progression Free Survival (PFS). Secondary end points included Overall Survival (OS), PFS among patients with brain metastases, confirmed Objective Response Rate (ORR), and safety.
In the primary analysis, at a median follow-up of 14 months, TUKYSA® added to HERCEPTIN® and XELODA®, significantly improved Overall Survival (OS) and Progression Free Survival (PFS) in patients with HER2-positive metastatic breast cancer. After the primary analysis, the protocol was amended to allow unblinding and cross-over from the placebo combination to the TUKYSA® combination. Protocol prespecified descriptive analyses of OS, PFS and safety were carried out at about 2 years from the last patient randomized. The researchers in this publication reported the final efficacy and safety outcomes after an additional 15.6 months follow up (total follow up of 29.6 months) in patients from the HER2CLIMB trial.
At a median follow up of 29.6 months, the median duration of OS was 24.7 months for the TUKYSA® combination group versus 19.2 months in the placebo combination group (HR for death=0.73; P=0.004). The estimated OS rate at 2 years was 51% in the TUKYSA® combination group and 40% in the placebo combination group. The OS benefit with the TUKYSA® combination was noted across all prespecified subgroups in the overall study population and was consistent with the primary analysis. The median duration of PFS was 7.6 months for the TUKYSA® combination group versus 4.9 months for the placebo combination group (HR for progression or death=0.57; P<0.00001), and PFS at 1 year was 29% and 14%, respectively.
Systemic treatment with TUKYSA® in combination with HERCEPTIN® and XELODA® provided consistent clinical benefit to patients with and without brain metastases. TUKYSA® combination doubled the intracranial Objective Response Rate, reduced the risk of intracranial progression or death by two-thirds in all patients with brain metastases. In this study population, the estimated 1-year intracranial PFS was 40% in the TUKYSA® group and 0% in the control group. In patients with untreated or treated and progressing (active) brain metastases, the estimated 1-year intracranial PFS was 35% in the TUKYSA® group, 0% in the control group, and in patients with treated (stable) brain metastases, was 53% in the TUKYSA® group and 0% in the control group. The TUKYSA® combination was well tolerated with a low rate of discontinuation due to toxicities. Common adverse events in the TUKYSA® group included diarrhea, Palmar-Plantar Erythrodysesthesia syndrome, nausea, vomiting and fatigue. Diarrhea and abnormal liver function tests were more common in the TUKYSA® group than in the control group.
It was concluded that with additional follow up, TUKYSA® in combination with HERCEPTIN® and XELODA® provided a clinically meaningful survival benefit, including those with brain metastases, supporting the use of this combination in patients with previously treated HER2-positive metastatic breast cancer, after progression on two HER2-targeted therapies.
Tucatinib versus placebo added to trastuzumab and capecitabine for patients with pretreated HER2+ metastatic breast cancer with and without brain metastases (HER2CLIMB): final overall survival analysis. Curigliano G, Mueller V, Borges V, et al. Ann Oncol. 2022;33:321-329.
KISQALI® Plus FEMARA® Improves Overall Survival in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation and associated with increased expression of CDK4. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.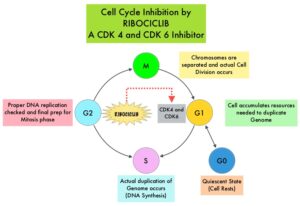
KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6, preferentially inhibiting CDK4, that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest. In a phase 1b study involving postmenopausal women with ER positive, HER2-negative advanced breast cancer, KISQALI® in combination with FEMARA® (Letrozole) demonstrated an Overall Response Rate (ORR) of 46% and a Clinical Benefit Rate of 79%, in treatment-naïve patients with advanced breast cancer. This led to the design of MONALEESA-2 trial.
MONALEESA-2 trial is a randomized, double-blind, placebo-controlled, Phase III study in which 668 patients were randomly assigned in a 1:1 ratio to receive either KISQALI® plus FEMARA® or placebo plus FEMARA®. Eligible patients included post-menopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer who had received no prior therapy for advanced disease. Treatment consisted of oral KISQALI® 600 mg daily on a 3-weeks on and 1-week off schedule, in 28-day treatment cycles plus FEMARA® 2.5 mg orally daily on a continuous schedule or placebo plus FEMARA®. Patients were stratified according to the presence or absence of liver or lung metastases and treatment was continued until disease progression or unacceptable toxicity. No treatment crossover was allowed. The median age was 62 years, close to 60% of the patients had visceral metastases, and patients were stratified according to the presence or absence of liver or lung metastases. The Primary end point was Progression Free Survival (PFS) and Secondary end points included Overall Survival (OS), Overall Response Rate (ORR), Clinical Benefit Rate (Overall Response plus Stable disease lasting 24 weeks or more), Safety, and Quality of Life assessments.
In the primary and updated analyses of the MONALEESA-2 trial, PFS was significantly longer with KISQALI® plus FEMARA® than with placebo plus FEMARA® (25.3 months versus 16.0 months; HR for disease progression or death=0.57; P<0.001). The Overall Survival data were immature at the time of the primary and updated analyses. The authors have now reported the findings from the protocol-specified final analysis of Overall Survival, which is a key Secondary end point.
After a median follow up of 6.6 years, a significant Overall Survival benefit was observed with KISQALI® plus FEMARA®, compared to placebo plus FEMARA®. The median Overall Survival was 63.9 months with KISQALI® plus FEMARA® and 51.4 months with placebo plus FEMARA® (HR=0.76; two-sided P=0.008). This Overall Survival benefit was consistent across all prespecified subgroups. The median time to first subsequent chemotherapy was 50.6 months in the KISQALI® group and 38.9 months in the placebo group (HR for receipt of first chemotherapy=0.74). No new safety signals were observed.
It was concluded from the analysis of the MONALEESA-2 trial that first line therapy with KISQALI® plus FEMARA® showed a significant Overall Survival benefit as compared with placebo plus FEMARA®, in patients with HR-positive, HER2-negative advanced breast cancer, with a 24% relative reduction in the risk of death. The authors added that MONALEESA trials of KISQALI® have shown a consistent Overall Survival benefit regardless of accompanying endocrine therapy, line of therapy, or menopausal status.
Overall Survival with Ribociclib plus Letrozole in Advanced Breast Cancer. Hortobagyi GN, Stemmer SM, Burris HA, et al. N Engl J Med 2022; 386:942-950
Overall Survival Benefit with the Addition of Capecitabine to Adjuvant Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence.
Patients with early stage breast cancer often receive adjuvant chemotherapy to improve Overall Survival (OS), and this is even more so true for HER positive and triple negative (ER, PR and HER negative) breast cancer patients, who are at an increased risk to develop recurrent disease. Meta-analyses conducted by the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) has shown a 20-25% relative risk reduction in breast cancer mortality with first-generation adjuvant chemotherapy regimens such as CMF (Cyclophosphamide/Methotrexate/Fluorouracil) and additional survival benefit with the Anthracyclines and Taxane based regimens. This benefit is dependent on the type of chemotherapy administered and chemotherapy dose intensity.
XELODA® (Capecitabine) is an oral prodrug of fluorouracil which is presently approved for the treatment of advanced breast cancer, but NOT for neoadjuvant or adjuvant treatment of early breast cancer. Meta-analysis of randomized trials has found that addition of Capecitabine to standard adjuvant chemotherapy regimens prolongs Disease Free Survival (DFS), whereas replacing a standard agent with Capecitabine did not improve DFS. Preclinical models have suggested that chemotherapy agents such as taxanes, and Cyclophosphamide increase thymidine phosphorylase concentration in the cancer cell, potentially leading to improved conversion of Capecitabine to fluorouracil within the tumor, suggesting that concomitant administration of Capecitabine with these agents improves efficacy, compared with single-agent Capecitabine. The researchers in this publication addressed the question whether addition of Capecitabine to these regimens could lead to improved survival outcomes.
The Finland Capecitabine Trial (FinXX) is a randomized, open-label, multicenter, Phase III trial that evaluated the addition of Capecitabine to an adjuvant chemotherapy regimen containing a taxane and an anthracycline for the treatment of early breast cancer. In this study, 1,500 patients with axillary node-positive or high-risk node-negative early breast cancer were randomly assigned to 6 cycles of either the Capecitabine arm- TX-CEX (N=753) or to the control group-T-CEF (N=747). TX-CEF consisted of Docetaxel 60 mg/m2 IV day 1 and Capecitabine 900 mg/m2 PO BID on days 1-15 of a 21-day cycle for 3 cycles followed by CEX consisting of Cyclophosphamide 600 mg/m2 IV Day 1, Epirubicin 75 mg/m2 IV on Day 1 and Capecitabine 900 mg/m2 PO BID on days 1-15 of a 21 day cycle for 3 cycles. T-CEF consisted of Docetaxel 80 mg/m2 IV Day 1, every 3 weeks for 3 cycles followed by CEF consisting of Cyclophosphamide 600 mg/m2 IV, Epirubicin 75 mg/m2 IV and Fluorouracil 600 mg/m2 IV, all administered on Day 1, every 3 weeks for 3 cycles. Adjuvant endocrine therapy was initiated within 2 months after completion of chemotherapy if the tumor was ER or PR-positive. Radiotherapy was given after completion of chemotherapy according to each institution's practice. Adjuvant Trastuzumab was approved while the trial accrual was ongoing and was allowed for women with HER2-positive cancer after May 2005, and adjuvant Trastuzumab was administered to 13% patients assigned to TX-CEF and 11% patients assigned to T-CEF. The median patient age was 52.5 yrs, 76% of patients had ER-positive tumors, 19% had HER2-positive cancer, 13% had Triple Negative Breast Cancer, more than 90% had T1 or T2 tumors, and 89% were node positive. The researchers then performed a protocol-scheduled analysis of Overall Survival on the basis of approximately 15-year follow up of the patients.
At a median follow up of 15.3 years, the Overall Survival was 77.6% in the TX-CEX group and 73.3% in the T-CEF group (HR=0.81; P=0.037). Exploratory subgroup analysis suggested that patients with ER-negative disease and those with Triple Negative Breast Cancer lived longer with the addition of Capecitabine (TX-CEX regimen), than those treated with T-CEF.
It was concluded that the addition of Capecitabine to a chemotherapy regimen significantly improved Overall Survival at median follow up of 15 years in a patient population with early breast cancer, suggesting that Capecitabine may be an important addition to adjuvant chemotherapy in patients with high risk disease.
Adjuvant Capecitabine for Early Breast Cancer: 15-Year Overall Survival Results from a Randomized Trial. Joensuu H, Kellokumpu-Lehtinen P-L , Huovinen R, et al. DOI: 10.1200/JCO.21.02054 Journal of Clinical Oncology. Published online January 12, 2022.
Adjuvant VERZENIO® in High Risk Early Stage Breast Cancer: Updated Efficacy and Ki-67 Analysis
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors, and Hormone Receptor (HR)-positive/HER2-negative breast cancer is the most frequently diagnosed molecular subtype. Majority of these patients are diagnosed with early stage disease and are often cured with a combination of surgery, radiotherapy, chemotherapy, and hormone therapy. However approximately 20% of patients will experience local recurrence or distant relapse during the first 10 years of treatment. This may be more relevant for those with high risk disease, among whom the risk of recurrence is even greater during the first 2 years while on adjuvant endocrine therapy, due to primary endocrine resistance. More than 75% of the early recurrences are seen at distant sites.
Cyclin Dependent Kinases (CDKs) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. CDK 4 and 6 phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity. Phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in HR-positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of CDKs in the cell cycle, has paved the way for the development of CDK inhibitors.
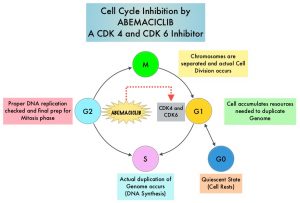 VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against Cyclin D1/CDK 4 and Cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against Cyclin D1/CDK 4 and Cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
VERZENIO® is presently approved by the FDA as monotherapy as well as in combination with endocrine therapy for patients with HR-positive, HER2- negative advanced breast cancer. The addition of VERZENIO® to FASLODEX® (Fulvestrant) resulted in a statistically significant improvement in Overall Survival (OS) among patients with HR-positive, HER2-negative advanced breast cancer, who had progressed on prior endocrine therapy. The goal of monarchE was to evaluate the additional benefit of adding a CDK4/6 inhibitor to endocrine therapy in the adjuvant setting, for patients with HR-positive, HER2-negative, high risk, early breast cancer.
The International monarchE trial, is an open-label, randomized, Phase III study, which included 5637 patients, who were pre- and postmenopausal, with HR-positive, HER2-negative early breast cancer, and with clinical and/or pathologic risk factors that rendered them at high risk for relapse. The researchers defined high risk as the presence of four or more positive axillary lymph nodes, or 1-3 three positive axillary lymph nodes, with either a tumor size of 5 cm or more, histologic Grade 3, or centrally tested high proliferation rate (Ki-67 of 20% or more). Following completion of primary therapy which included both adjuvant and neoadjuvant chemotherapy and radiotherapy, patients were randomly assigned (1:1) to VERZENIO® 150 mg orally twice daily for 2 years plus 5-10 years of physicians choice of endocrine therapy as clinically indicated (N=2808), or endocrine therapy alone (N=2829). The median patient age was 51 years, about 43% of the patients were premenopausal, and 95% of patients had prior chemotherapy. Approximately 60% of patients had 4 or more positive lymph nodes. The Primary endpoint was Invasive Disease Free Survival (IDFS), and Secondary end points included Distant Relapse Free Survival (DRFS), Overall Survival (OS), and Safety. The researchers provided updated results from the prespecified Primary outcome analysis, additional follow-up analysis conducted at regulatory request, as well as outcomes from prespecified subpopulations, based on Ki-67 levels.
At the time of Primary outcome analysis, with a median follow up of 19 months, 1,437 patients (25.5%) had completed the 2 year treatment period and 3,281 patients (58.2%) were in the 2 year treatment period. The combination of VERZENIO® plus endocrine therapy demonstrated superior Invasive Disease Free Survival (IDFS) compared to endocrine therapy alone, with a 29% reduction in the risk of developing invasive disease (P=0.0009; HR=0.71). The 2-year IDFS in the combination group was 92.3% and 89.3% in the endocrine therapy alone treatment group, with an absolute improvement of 3.0%. Further, there was an improvement in the 2-year distant Relapse Free Survival (DRFS) rate among patients who received the combination treatment compared with those who received endocrine therapy alone, corresponding to an absolute difference of 3.0% at 2 years (93.8% versus 90.8%, respectively; HR=0.69; P<0.001).
With 8 months of additional follow up, at a median of 27 months and with 90% of patients off treatment, the benefit with the combination of VERZENIO® plus endocrine therapy was maintained for IDFS (HR=0.70; P<0.0001) and DRFS (HR=0.69; P<0.0001), demonstrating a 30% risk reduction for IDFS and 31% risk reduction for DRFS. There was continued treatment benefit over time that extended beyond the 2-year treatment period of VERZENIO®. With more patients at risk for recurrence at 3 years, the data demonstrated absolute improvements in 3-year IDFS and DRFS rates of 5.4% and 4.2%, respectively. This treatment benefit in IDFS and DRFS was noted across prespecified subgroups. Further, the benefit with VERZENIO® was consistent, regardless of Ki-67 index. Overall Survival data was immature at the time of this analysis.
It was concluded that adjuvant VERZENIO® combined with endocrine therapy continued to demonstrate statistically significant and clinically meaningful improvement in Invasive Disease Free Survival and Distant Relapse Free Survival, among patients with HR-positive, HER2-negative, node-positive, high risk, early breast cancer, regardless of Ki-67 status.
Adjuvant abemaciclib combined with endocrine therapy for high-risk early breast cancer: updated efficacy and Ki-67 analysis from the monarchE study. Harbeck N, Rastogi P, Martin M, et al. Annals of Oncology 2021;32: 1457-1459.
Elacestrant in Metastatic Breast Cancer Progressing on CDK4/6 Therapy and ESR1-Mutant Subtype
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 290,560 new cases of breast cancer will be diagnosed in 2022 and about 43,780 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors. The most common subtype of metastatic breast cancer is Hormone Receptor-positive (HR-positive), HER2-negative breast cancer (65% of all metastatic breast tumors), and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients, with a median Overall Survival (OS) of 36 months. With the development of Cyclin Dependent Kinases (CDK) 4/6 inhibitors, endocrine therapy plus a CDK4/6 inhibitor is the mainstay for the management of ER+/HER2- metastatic breast cancer as first-line therapy. Even with this therapeutic combination, most patients will eventually experience disease progression, including development of ESR1 (Estrogen Receptor gene alpha) mutations.
ESR1 is the most common acquired mutation noted in breast tumors as they progress from primary to metastatic setting. These mutations promote ligand independent Estrogen Receptor activation and have been shown to promote resistance to estrogen deprivation therapy. It appears that ESR1 mutations are harbored in metastatic ER+ breast cancers with prior Aromatase Inhibitor (AI) therapy, but not in primary breast cancers, suggesting that ESR1 mutations may be selected by prior therapy with an AI, in advanced breast cancer. In a recently published study (JAMA Oncol.2016;2:1310-1315), ESR1 mutations Y537S and D538G mutations detected in baseline plasma samples from ER+/HER- advanced breast cancer patients, was associated with shorter Overall Survival. In this study it was noted that there was a three-fold increase in the prevalence of these mutations in patients who had failed first line hormonal therapy for metastatic disease, compared with those who were initiating first line therapy for advanced breast cancer (33% versus 11%).
Fulvestrant is a parenteral, Selective Estrogen Receptor Degrader (SERD) and is the only SERD approved for the treatment of postmenopausal women with HR-positive metastatic breast cancer. However, acquired ESR1 mutations can also occur following Fulvestrant treatment, possibly because of poor bioavailability and incomplete ER blockade when administered intramuscularly. There is therefore an urgent unmet need for an alternate SERD that has activity in tumors harboring ESR1 mutations, and has improved bioavailability allowing oral administration.
Elacestrant is an oral, nonsteroidal, Selective Estrogen Receptor Degrader (SERD) that degrades the Estrogen Receptor (ER) in a dose-dependent manner and inhibits estradiol-dependent functions of ER target gene transcription induction and breast cancer cell proliferation. Estradiol-stimulated tumor growth was diminished by Elacestrant in the ER+ xenograft models derived from heavily pretreated patients, including models resistant to CDK 4/6 inhibitors, Fulvestrant and those harboring ESR1 mutations Y537S and D538G. In an early Phase I trial, Elacestrant was noted to have an acceptable safety profile, and demonstrated single-agent activity with confirmed Partial Responses in heavily pretreated patients with ER+ metastatic breast cancer.
EMERALD trial is a multicenter, International, randomized, open-label, Phase III study designed to evaluate the benefit of Elacestrant in patients with ER+ HER2- advanced or metastatic breast cancer. In this study, 477 postmenopausal women with ER+/HER2- metastatic breast cancer were randomly assigned 1:1 to receive either Elacestrant 400 mg orally daily (N=239) or the Standard of Care which included investigator’s choice of Fulvestrant or an Aromatase Inhibitor including Anastrozole, Letrozole, or Exemestane (N=238). Treatment was given until disease progression. Both treatment groups were well balanced. The median patient age was 63 years, and patients must have progressed or relapsed on or after 1 or 2 lines of endocrine therapy for advanced disease, one of which was given in combination with a CDK4/6 inhibitor, had 1 or fewer lines of chemotherapy for advanced disease, and had an ECOG performance status of 0 or 1. In the study, 48% had tumors with mutated ESR1 and these patients were evenly distributed in both treatment groups. Patients were stratified by ESR1-mutation status, prior treatment with Fulvestrant, and visceral metastases. The co-Primary end points were Progression Free Survival (PFS) in the overall population, and in those with ESR1 mutations. Overall Survival (OS) was a Secondary end point.
Treatment with Elacestrant resulted in a statistically significant and clinically meaningful improvement in PFS, compared with Standard of Care. There was a 31% reduction in the risk of progression or death in the Elacestrant group for all patients (HR=0.69; P=0.0018) and a 45% reduction in the risk of progression or death among those with ESR1 mutations (HR=0.55; P=0.0005).
The PFS at 12 months with Elacestrant was 22.3% in all patients compared with 9.4% for those receiving the Standard of Care treatment. Among the ESR1 mutation group, the 12 month PFS rate was more pronounced and was 26.8% with Elacestrant, compared to 8.2% with Standard of Care. The benefits with Elacestrant compared with Standard of Care, was consistent across multiple prespecified subgroups including patients who had received prior Fulvestrant. There also was a trend toward improved Overall Survival in patients who received Elacestrant, compared with Standard of Care. The final OS results however are not expected until late 2022. Elacestrant was well tolerated and treatment discontinuation rate was not significantly different between the two treatment groups.
It was concluded that Elacestrant is the first oral Selective Estrogen Receptor Degrader that demonstrated significant and clinically meaningful improvement in PFS compared with Standard of Care endocrine therapy in patients with ER+/ HER2- metastatic breast cancer in the second/third line after treatment with a CDK4/6 inhibitor, and has the potential to become the new standard of care in the study population.
Elacestrant, an oral selective estrogen receptor degrader (SERD), vs investigator’s choice of endocrine monotherapy for ER+/HER2- advanced/metastatic breast cancer (mBC) following progression on prior endocrine and CDK4/6 inhibitor therapy: Results of the EMERALD phase 3 trial. Bardia A, Neven P, Streich G, et al. Presented at 2021 San Antonio Breast Cancer Symposium; December 7-10, 2021; San Antonio, TX. Abstract GS2-02.
Defining Patient Groups With Triple Negative Breast Cancer Who Derive Benefit From KEYTRUDA® plus Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers.
The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. It appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition, and inhibition of PI3K pathway. Previously published studies have shown that presence of tumor-infiltrating lymphocytes was associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response, and unleashes the tumor-specific effector T cells. The rationale for combining chemotherapy with immunotherapy is that cytotoxic chemotherapy releases tumor-specific antigens, and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity. Single agent KEYTRUDA® in metastatic TNBC demonstrated durable antitumor activity in several studies, with Objective Response Rates (ORRs) ranging from 10-21% and improved clinical responses in patients with higher PD-L1 expression. When given along with chemotherapy as a neoadjuvant treatment for patients with high-risk, early-stage TNBC, KEYTRUDA® combination achieved Pathological Complete Response rate of 65%, regardless of PD-L1 expression. Based on this data, KEYTRUDA® in combination with chemotherapy was studied, for first line treatment of advanced TNBC.
KEYNOTE-355 is a randomized, double-blind, Phase III study, which evaluated the benefit of KEYTRUDA® in combination with one of the three different chemotherapy regimens, nab-Paclitaxel, Paclitaxel, or the non-taxane containing Gemzar/Carboplatin, versus placebo plus one of the three chemotherapy regimens, in patients with previously untreated or locally recurrent inoperable metastatic TNBC. In this study, 847 patients were randomized 2:1 to receive either KEYTRUDA® 200 mg IV on day 1 of each 21-day cycle along with either nab-Paclitaxel 100 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, Paclitaxel 90 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, or Gemcitabine 1000 mg/m2 IV plus Carboplatin AUC 2, IV on days 1 and 8 of each 21-day cycle (N= 566) or placebo along with one of the three chemotherapy regimens (N= 281). This study was not designed to compare the efficacy of the different chemotherapy regimens. Treatment was continued until disease progression. Patients were stratified by chemotherapy, PD-L1 tumor expression (CPS-Combined Positive Score of 1 or higher versus CPS of less than 1), and prior treatment with the same class of neoadjuvant/adjuvant chemotherapy (Yes versus No). The baseline characteristics of treatment groups were well-balanced. The co-Primary end points of the trial were Progression Free Survival (PFS) and Overall Survival (OS) in patients with PD-L1-positive tumors, and in all patients. Secondary end points were Objective Response Rate (ORR), Duration of Response, Disease Control Rate, and Safety.
In the primary analysis of the KEYNOTE-355 trial, the Overall Survival results after a median follow up of 44.1 months in the subgroup of patients with PD-L1 CPS (Combined Positive Score) of 10 or more was significantly better with first line KEYTRUDA® plus chemotherapy versus placebo plus chemotherapy (23.0 months versus 16.1 months, respectively; HR=0.73; P=0.0093). This represented a 27% reduction in the risk of death with the KEYTRUDA® combination. KEYTRUDA® in combination with chemotherapy, also significantly improved PFS in patients with CPS (Combined Positive Score) of 10 or greater. The median PFS was 9.7 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for placebo plus chemotherapy (HR=0.65, P=0.0012). This represented a 35% reduction in the risk of disease progression. However, among patients with CPS of 1 or greater, the median PFS was not considered statistically significant, based on prespecified statistical criteria.
The researchers here in presented the results of a subgroup analysis, stratified by levels of PD-L1 expression, as assessed by CPS score. In the subgroups with CPS scores of less than 1 and 1-9, Overall Survival was similar for KEYTRUDA® plus chemotherapy and placebo plus chemotherapy. However, in subgroups with CPS 10-19 and CPS 20 or more, there was sustained separation of the Overall survival curves starting at approximately 10 months and the survival was improved by about 28%.
The authors noted that the general trend for PFS was consistent with that observed for Overall Survival, with improving PFS trend among those subgroups with PD-L1 enriched CPS of 10 or more. In the subgroup of patients with a CPS of 10-19 and CPS of 20 or more, the addition of KEYTRUDA® to chemotherapy resulted in a more sustained separation of PFS curves, beginning at approximately 4 months, compared with placebo plus chemotherapy. The Hazard Ratios for these two groups were 0.70 and 0.62, respectively. Toxicities of any grade were reported in 96% of the experimental group and 95% of the placebo plus chemotherapy group. The rate of Grades 3-5 treatment-related adverse events was 68.1% and 66.9%, respectively and the majority of treatment discontinuations in this study were for progressive disease.
The researchers based on this subgroup analyses concluded that a CPS of 10 or more is a reasonable cutoff to define the population of women with metastatic Triple Negative Breast Cancer, expected to derive treatment benefit from KEYTRUDA® plus chemotherapy, lending further support to KEYTRUDA® plus chemotherapy as a standard of care treatment regimen for this group of patients.
Final results of KEYNOTE-355: randomized, double-blind, phase 3 study of pembrolizumab + chemotherapy vs placebo + chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer. Cortés J, Cescon DW, Rugo HS, et al. Presented at: 2021 San Antonio Breast Cancer Symposium; December 7-10, 2021; San Antonio, TX. Abstract GS1-02.
Postmenopausal Women with Node Positive Breast Cancer May Not Benefit From Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Approximately 25% of patients with Hormone Receptor (HR)-positive, HER2-negative early breast cancer have metastatic lymph node involvement and two third of these patients are postmenopausal. Majority of these patients currently receive adjuvant chemotherapy.
The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for early stage, HR-positive, HER-negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
In the ground-breaking TAILORx (Trial Assigning Individualized Options for Treatment) study which enrolled 10,273 patients with HR-positive, HER2-negative, axillary node-negative breast cancer, patients were divided into three groups based on their Recurrence Score. Patient with Intermediate Recurrence Score of 11-25 were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. There was no benefit noted from adding chemotherapy to endocrine therapy, for women older than 50 years in this Intermediate RS group, suggesting that a significant percentage of women with node-negative breast cancer do not achieve substantial benefit from chemotherapy. For women 50 years old or younger who received chemotherapy and had a Recurrence Score of at least 16, there was a lower rate of distant recurrence, and the absolute benefit increased with increasing recurrence score. Further, the risk of recurrence and benefit of chemotherapy was further influenced by the tumor size and grade.
Whether the results of TAILORx can be extrapolated to women with node-positive breast cancer has remained unclear. It is estimated that approximately 85% of women with node-positive disease have Recurrence Score results of 0-25. The RxPONDER (A Clinical Trial RX for Positive Node, Endocrine Responsive Breast Cancer) trial was designed to determine the benefit of chemotherapy, in patients with HR-positive, HER2-negative breast cancer and 1-3 positive axillary lymph nodes (nodal stage N1), who had a Recurrence Score of 0-25. This trial did not include pre and postmenopausal women with Recurrence Score results 26-100, based on previously published studies suggesting that this patient group benefited from chemotherapy.
SWOG S1007 (RxPONDER) is an multicenter, international, prospective, randomized, Phase III trial, in which patients with HR-positive, HER2-negative breast cancer with 1-3 positive axillary lymph nodes were included, to determine which patients would benefit from chemotherapy and which patients could safely avoid it. In this study, a total of 5083 HR-positive, HER2-negative breast cancer patients with 1-3 positive lymph nodes and Oncotype DX Recurrence Score of less than 25 were randomly assigned 1:1 to receive chemotherapy plus endocrine therapy (N=2547) or endocrine therapy alone (N=2536). The median patient age was 57.5 years and approximately two-thirds of patients were postmenopausal and one-third were premenopausal and had no contraindications to taxane and/or anthracycline based chemotherapy. Patients were stratified by Recurrence Score (0-13 versus 14-25), menopausal status, and axillary nodal dissection versus sentinel node biopsy. The Primary endpoint was Invasive Disease Free Survival (IDFS), defined as local, regional, or distant recurrence, any second invasive cancer, or death from any cause, and whether the effect depended on the Recurrence Score. Secondary endpoints included distant Relapse Free Survival (RFS) and Overall Survival (OS).
At a median follow up of 6.1 years, the chemotherapy benefit with respect to increasing invasive DFS differed according to menopausal status. Among postmenopausal women, in this updated analysis with longer follow up, the invasive DFS at 5 years was 91.9% in the endocrine therapy alone group, and was 91.3% in those treated with chemotherapy plus endocrine therapy (HR=1.02; P=0.89). Postmenopausal women with recurrence scores of 0 to 25 continued to NOT benefit from adjuvant chemotherapy.
Among premenopausal women however, the invasive DFS at 5 years was 89% in the endocrine therapy alone group and 93.9% % in those treated with chemotherapy plus endocrine therapy (HR=0.64; P=0.004). There was a 5-year absolute benefit of 4.9% for invasive DFS with chemotherapy among premenopausal women. There was a similar increase noted in the distant Relapse Free Survival (HR=0.58; P=0.009). The relative chemotherapy benefit did not increase as the Recurrence Score increased.
It was concluded from this practice-changing study that postmenopausal women with HR-positive, HER2-negative breast cancer with 1-3 positive nodes and Oncotype DX Recurrence Score of 25 or less, can safely avoid receiving adjuvant chemotherapy, whereas premenopausal patients with 1-3 positive nodes and a Recurrence Score of 25 or less benefited from chemotherapy plus endocrine therapy and had a longer invasive Disease Free Survival and distant Relapse Free Survival, than those who received endocrine therapy alone.
21-Gene Assay to Inform Chemotherapy Benefit in Node-Positive Breast Cancer. Kalinsky K, Barlow WE, Gralow JR, et al. N Engl J Med 2021;385:2336-2347
Risk of Cardiovascular Diseases among Older Breast Cancer Survivors
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Significant progress in breast cancer screening techniques, as well as new and novel therapies, have resulted in early cancer detection and improvement in the breast cancer 5-year survival rate in the US from 75% in the 1970s to 91% in the 2010s. Cardiovascular Disease (CVD) is the most frequent cause of noncancer-related death, and cardiotoxicities associated with cancer treatments may increase cardiovascular risk in this population of breast cancer survivors. However, few studies have in detail quantified the risks of the different clinically important cardiovascular outcomes. The authors therefore assessed the prevalence of the different clinically specific cardiovascular outcomes at breast cancer diagnosis, and their incidence after diagnosis, among survivors 65 years or older in the US, and compared this with similar women without cancer.
The researchers performed a matched cohort study using prospectively collected data from the SEER-Medicare linked claims-based database and identified all women older than 65 years of age with an incident Stage I-III breast cancer diagnosis in 2004 through 2013. Each patient with breast cancer was matched at diagnosis with 5 cancer-free female counterparts. Baseline prevalence of specific cardiovascular outcomes was measured, and the risk for individual cardiovascular outcomes during follow up was calculated, taking into consideration time since diagnosis, race/ethnicity, prior Cardiovascular Disease (CVD), and age. This study included a total of 91,473 women with breast cancer and 454,197 without breast cancer.
It was noted that women with breast cancer had lower baseline prevalence of all CVDs. Breast cancer survivors had substantially increased risks of Deep Vein Thrombosis and pericarditis, compared with cancer-free female counterparts. There was also evidence of smaller increased risks of sudden cardiac arrest, arrhythmia, heart failure, and valvular heart disease. The increased risks of arrhythmia, heart failure, pericarditis, and Deep Vein Thrombosis were most pronounced in the first year and persisted for more than 5 years after cancer diagnosis. There was evidence of a decreased risk of incident angina, myocardial infarction, revascularization, peripheral vascular disease, and stroke in breast cancer survivors, although this was not constant over time.
The CVD risk during follow up was consistently higher in African American women diagnosed with breast cancer compared with Caucasian women, regardless of whether there was an overall increased or decreased risk of outcomes during the entire follow up period, and this is consistent with racial differences in overall CVD risk in the US.
Finally, there was consistently a greater risk of all cardiovascular outcomes in those diagnosed with Stage III, Grade 3, and ER/PR-negative breast cancer, which may be a reflection of the more aggressive cancer treatment regimens used in these subtypes.
The authors concluded that there is evidence of increased risk of several cardiovascular diseases in elderly women diagnosed with breast cancer in the US, compared with similar women without cancer, with this risk persisting for several years after diagnosis. They added that these results highlight the importance of periodic cardiovascular evaluation throughout the long term follow up of women diagnosed with breast cancer.
Risk of Cardiovascular Diseases Among Older Breast Cancer Survivors in the United States: A Matched Cohort Study. Matthews AA, Hinton SP, Stanway S, et al. J Natl Compr Canc Netw 2021;19:275-284.
Duration of Adjuvant Aromatase Inhibitor Therapy in Postmenopausal Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Luminal breast cancer is the most prevalent molecular subtype in postmenopausal females, accounting for over 70%. Despite substantial improvements in adjuvant therapies, the risk of disease recurrence continues indefinitely, with more than half the recurrences occurring after the first 5 years following diagnosis. Following initial adjuvant endocrine therapy with Tamoxifen for 5 years, the addition of extended adjuvant therapy has resulted in 40% longer Disease Free Survival (DFS), when compared to placebo or no extended therapy. However the benefit of extending adjuvant Aromatase Inhibitor therapy for 5 years beyond the initial 5-year duration regimen is less well established. Further, the most effective duration of such extended adjuvant endocrine therapy remains unclear. Added to this dilemma are the side effects associated with Aromatase Inhibitor therapy including hot flushes, arthralgia, and bone pain, as well as treatment-induced osteoporosis, which can have a significant impact on patient’s quality of life. Researchers in the Secondary Adjuvant Long-Term Study with Arimidex [Anastrozole] (SALSA) prospectively investigated whether an additional 2 years or 5 years of Anastrozole therapy would result in better outcomes, following the initial 5 years of endocrine therapy, in postmenopausal women with Hormone Receptor-positive breast cancer.
The authors conducted a prospective, multicenter, randomized, Phase III trial, which included 3,470 eligible postmenopausal women with Stages I, II or III early stage breast cancer with no evidence of recurrence. Enrolled patients had invasive Hormone Receptor-positive breast cancer, and had received 5 years (plus or minus 12 months) of adjuvant endocrine therapy with Tamoxifen, Aromatase Inhibitors, or both sequentially, up until 12 months before randomization. Patients were randomly assigned 1:1 to receive Anastrozole 1 mg, orally daily, for either 2 additional years for a total of 7 years (N=1732) or 5 additional years for a total of 10 years (N=1738). The two treatment groups were well balanced. The median age at the time of randomization was 64 years, 72% of patients had tumors that were smaller than 2 cm, 66% had node-negative disease, and 19% had high-grade tumors. Stratification criteria included pathological tumor stage, pathological node stage, primary adjuvant endocrine therapy and adjuvant chemotherapy.
The primary analysis included all the patients who were still participating in the study (N=3208), including 1,603 in the 2-year group versus 1,605 in the 5-year group. In the primary analysis population of 3208 patients, 51% had received Tamoxifen alone for the initial 5 years, 7.3% had received an Aromatase Inhibitor alone, and 41.7% had received an Aromatase Inhibitor in combination with Tamoxifen. The Primary end point was Disease Free Survival (DFS). Secondary end points were Overall Survival (OS), contralateral breast cancer, second primary cancer, and clinical bone fracture. The median follow-up after randomization was 118 months.
The researchers observed no difference in DFS with 2 versus 5 additional years of adjuvant endocrine therapy with Anastrozole. The DFS 10 years since randomization was 73.6% in the 2-year group versus 73.9% in the 5-year group (HR=0.99; P=0.90). Contralateral breast cancer occurred in 2.2% versus 2.1% of patients (HR= 1.15), and local recurrence occurred in 3% versus 2.4% in the 2 year and 5 year groups, respectively. There was no difference noted for Overall Survival at 8 years between the two treatment groups (87.5% in the 2-year group and 87.3% in the 5-year group, HR for death from any cause=1.02). The risk of clinical bone fracture however was higher in the 5-year group than in the 2-year group (HR=1.35).
It was concluded from this study that in postmenopausal women with Hormone Receptor positive breast cancer who had received 5 years of adjuvant endocrine therapy, extending endocrine therapy with an Aromatase Inhibitor by an additional 5 years provided no benefit over a 2-year extension, but was associated with a greater risk of bone fracture.
Duration of Adjuvant Aromatase-Inhibitor Therapy in Postmenopausal Breast Cancer. Gnant M, Fitzal F, Rinnerthaler G, et al. N Engl J Med 2021; 385:395-405.
Genetic Testing in Older Women with Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Genetic testing for cancer susceptibility with multigene testing panels is now becoming widely available and affordable. Identification of Pathogenic Variants in predisposition genes such as BRCA1 and BRCA2 among carriers has provided benefit through early intervention. There are 12 established breast cancer predisposition genes implicated among breast cancer cases, unselected for family history or young age at breast cancer diagnosis. Only loss-of-function through protein truncation and missense variants labeled as pathogenic, are classified as Pathogenic Variants-PV in the ClinVar database. They include ATM, BARD1, BRCA1, BRCA2, CDH1, CHEK2, NF1, PALB2, PTEN, RAD51C, RAD51D, and TP53. Among these genes, BRCA1, BRCA2, and PALB2 are high risk genes, whereas the other genes are considered moderate risk. Identifying high risk genes in women with breast cancer is relevant both for prevention and treatment. Breast cancer prevention opportunities include contralateral prophylactic mastectomy or surveillance with MRI of the breast, prophylactic salpingo-oophorectomy for BRCA mutation carriers, avoidance of radiation in TP53 mutation carriers, and genetic testing for family members. PARP inhibitor LYNPARZA® (Olaparib) is indicated for the treatment of patients with deleterious or suspected deleterious germline BRCA1/2 mutated, HER2-negative metastatic breast cancer who have been treated with chemotherapy in the neoadjuvant, adjuvant or metastatic setting. Younger age at diagnosis, strong family history of breast and/or ovarian cancer, Ashkenazi Jewish ancestry, or Triple Negative Breast Cancer (TNBC), are all associated with hereditary breast cancer and approximately 10% of these patients carry a Pathogenic Variant in a breast cancer predisposition gene.
According to the NCCN guidelines, hereditary cancer testing for women with breast cancer diagnosed at age greater than 65 years, without specific risk factors such as Ashkenazi Jewish ancestry or family history of cancer, has limited clinical utility as these women have less than 2.5% chance of having a Pathogenic Variant in a high risk gene. However, few studies have specifically evaluated breast cancer predisposition genes in women over age 65 years.
The authors conducted this study to determine the prevalence of Pathogenic Variants in established breast cancer predisposition genes, and to estimate remaining lifetime risks of breast cancer associated with Pathogenic Variants, among women over age 65 years in the general population. A total of 26,707 women over age 65 years from population-based studies (N=13,762, 51.5% with breast cancer and N=12,945, 48.5% age and study matched unaffected women controls) were tested for Pathogenic Variants in germline breast cancer predisposition gene. The researchers then assessed the frequencies of Pathogenic Variants and associations between Pathogenic Variants in each gene and breast cancer, and estimated the remaining lifetime breast cancer risks for non-Hispanic White women with Pathogenic Variants.
The researchers noted that the frequency of Pathogenic Variants in established breast cancer predisposition genes were identified in 3.18% of 13,762 women with breast cancer and 1.48% of the 12,945 age-matched unaffected controls. Pathogenic Variants in the high risk BRCA1, BRCA2, and PALB2 genes were found in 3.42% of women diagnosed with ER-negative breast cancer and 3.01% of women with Triple Negative Breast Cancer. The frequency of Pathogenic Variants in the high risk genes was low among women with no first degree relatives with breast cancer and ER-positive breast cancer. Pathogenic Variants in BRCA1, BRCA2, PALB2 and CHEK2 were associated with increased risks (Odds Ratio=2.9-4.0) of breast cancer. The remaining lifetime risk of breast cancer from age 66 to 85 years was more than 15% or more for those with Pathogenic Variants in BRCA1, BRCA2, and PALB2.
The authors concluded that based on this largest study to date of population-based US women over age 65 years, diagnosed with breast cancer, all women diagnosed with Triple Negative Breast Cancer or ER-negative breast cancer should receive genetic testing and that women over age 65 years with BRCA1 and BRCA2 Pathogenic Variants and perhaps with PALB2 and CHEK2 Pathogenic Variants should be considered for breast MRI screening, as they continue to be at an increased risk of breast cancer.
Risk of Late-Onset Breast Cancer in Genetically Predisposed Women. Boddicker NJ, Hu C, Weitzel JN, et al. J Clin Oncol 2021;39:3430-3440.
Duration of Extended Adjuvant Letrozole after Tamoxifen in Postmenopausal Women with Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Approximately 75% of patients with breast cancer are Hormone Receptor (HR) - positive (Estrogen Receptor/Progesterone Receptor positive), and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues.
It has been well established that treatment with 5 years of endocrine therapy in early stage, HR-positive breast cancer, significantly reduces the risks of locoregional and distant recurrence, contralateral breast cancer, death from breast cancer, and therefore death from any cause. Extended adjuvant endocrine therapy with either Tamoxifen or an Aromatase Inhibitor (AI) beyond 5 years can further reduce breast cancer recurrence. This however can result in treatment related side effects. Therefore, when considering extended adjuvant endocrine therapy beyond 5 years, the potential benefits should be weighed against the associated risk with such therapy. The absolute benefit of continuing endocrine therapy after 5 years depends on the absolute risk of later recurrence, if patient’s receives no further therapy.
Third generation Aromatase Inhibitors (AIs), including Anastrozole, Exemestane and Letrozole, have demonstrated improved efficacy, when compared to Tamoxifen, for the adjuvant endocrine treatment of postmenopausal patients with HR-positive breast cancer. Randomized trials such as the Intergroup Exemestane Study have shown improvements in Disease Free Survival (DFS) among patients who after 2-3 years on Tamoxifen treatment switch to Exemestane for the remainder of a 5-year endocrine treatment period, with a modest improvement in Overall Survival (OS). Whether there is added benefit by extending Aromatase Inhibitor therapy beyond 5 years has remained controversial.
The present study was conducted to compare extended therapy with Letrozole for 5 years versus the standard duration of 2-3 years of Letrozole, in postmenopausal patients with breast cancer, who had already received 2-3 years of Tamoxifen. This multicentre, open-label, randomized, Phase III trial included 2056 postmenopausal women patients with Stage I-III operable, invasive, HR-positive breast cancer, who had received adjuvant Tamoxifen therapy for at least 2 years but no longer than 3 years and 3 months, and had no signs of recurrent disease. Patients were randomly assigned (1:1) to receive Letrozole 2.5 mg orally once a day for 2-3 years (control group; N=1030) or Letrozole 2.5 mg orally once a day for 5 years (extended group; N=1026). Approximately 41% of patients had node-positive disease, 21% had Grade 3 tumors, 6% had HER2-positive disease and 55% had prior chemotherapy. The median duration of treatment with adjuvant Tamoxifen was about 2.5 years. The Primary endpoint was invasive Disease Free Survival. Safety analysis was done for patients who received at least 1 month of study treatment. About 80% of patients in the control group and 63% in the extended treatment group completed treatment. The median duration of Letrozole treatment was 5.0 years in the extended treatment group and 2.4 years in the control group. About 16% of patients in the extended treatment group and 11.7% in the control group received bisphosphonate treatment respectively.
After a median follow-up of 11.7 years, the 12-year Disease Free Survival was 62% in the control group and 67% in the extended treatment group (HR=0.78; P=0.0064). This benefit was seen across all patient subgroups. The Overall Survival was also significantly improved at 12 years, and was 84% in the control group versus 88% in the extended treatment group (HR=0.77; P=0.036).
With regards to Adverse Events, there was a slightly higher incidence of arthralgia, myalgia and osteoporosis in the extended treatment group, but there was no significant difference observed between the groups in the incidence of Skeletal Related Events.
It was concluded from this landmark study that, in postmenopausal patients with breast cancer who received 2-3 years of Tamoxifen, extended treatment with 5 years of Letrozole resulted in a significant improvement in Disease Free Survival, compared with the standard 2-3 years of Letrozole. The authors added that sequential endocrine therapy with Tamoxifen for 2-3 years followed by Letrozole for 5 years should be considered as one of the optimal standard endocrine treatments for postmenopausal patients with Hormone Receptor-positive breast cancer.
Extended therapy with letrozole as adjuvant treatment of postmenopausal patients with early-stage breast cancer: a multicentre, open-label, randomised, phase 3 trial. Del Mastro L, Mansutti M, Bisagni G, et al. Lancet Oncology. Published: September 17, 2021. DOI: https://doi.org/10.1016/S1470-2045(21)00352-1
Omitting Axillary Lymph Node Dissection in Patients with Clinically Positive Axillary Lymph Nodes Treated with Neoadjuvant Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Patients with locally advanced breast cancer with clinically positive axillary lymph nodes often receive chemotherapy in the neoadjuvant settings, and approximately 30-40% of patients achieve a pathological Complete Response (pCR), with eradication of disease in the axilla. In addition to increasing the likelihood of tumor resectability and breast preservation, patients achieving a pCR following neoadjuvant chemotherapy have a longer Event Free Survival (EFS) and Overall Survival (OS).
Previously published prospective trials have demonstrated that breast cancer patients with clinically positive axillary lymph nodes, who disease following neoadjuvant therapy is converted to clinically node negative disease, can safely undergo Sentinel Lymph Node Biopsy (SLNB) rather than axillary lymph node dissection, as the false negative rates are less than 10%, when 3 or more sentinel lymph nodes are retrieved. However, the rates of axillary lymph node recurrence in this population, has remained unclear. The purpose of this study was to evaluate axillary nodal recurrence rates in a consecutive cohort of breast cancer patients with clinically positive axillary lymph nodes, treated with neoadjuvant chemotherapy, who had negative disease following treatment, on Sentinel Lymph Node Biopsy.
This study included 769 patients with Stage II-III, biopsy-proven, node-positive breast cancer, of whom 610 patients were eligible for Sentinel Lymph Node Biopsy following neoadjuvant chemotherapy. Ninety one percent (N=555) converted to clinical node negative disease on physical examination, following neoadjuvant chemotherapy and 513 patients had 3 or more SLNs retrieved. Overall Axillary Lymph Node Dissection was avoided in 234 patients with 3 or more pathologically negative sentinel lymph nodes. The median patient age in this study cohort of 234 patients was 49 years. Median tumor size was 3 cm, 62% were HER2-positive, and 18% were triple negative. Majority of the patients (91%) received Doxorubicin-based neoadjuvant chemotherapy, 88% received adjuvant Radiotherapy (RT), and 70% of these patients also received nodal RT. The Primary outcome was the nodal recurrence rate among breast cancer patients with clinically positive axillary lymph nodes, treated with Sentinel Lymph Node Biopsy alone after neoadjuvant chemotherapy. Nodal recurrence was defined as a recurrence in the ipsilateral axillary, supraclavicular, or internal mammary nodal basins. Local recurrence was defined as an ipsilateral breast tumor recurrence. Distant failure included any distant metastases.
At a median follow up of 40 months, there was 1 axillary nodal recurrence, synchronous with local recurrence, in a patient who refused Radiation Therapy. Among patients who received Radiation Therapy (N=205), there were no nodal recurrences. The 5-year distant Recurrence Free Survival was 92.7%. The 5-year Overall Survival was 94.2%.
It was concluded from this study that in patients with clinically positive axillary lymph nodes, rendered clinically node negative with neoadjuvant chemotherapy, with 3 or more pathologically negative sentinel lymph nodes on Sentinel Lymph Node Biopsy alone, nodal recurrence rates were low without routine Axillary Lymph Node Dissection. These findings support surgical de-escalation by omitting Axillary Lymph Node Dissection in patients with clinically positive axillary lymph nodes, treated with neoadjuvant chemotherapy.
Nodal Recurrence in Patients with Node-Positive Breast Cancer Treated With Sentinel Node Biopsy Alone After Neoadjuvant Chemotherapy—A Rare Event. Barrio AV, Montagna G, Mamtani A, et al. JAMA Oncol. Published online October 7, 2021. doi:10.1001/jamaoncol.2021.4394
Late Breaking Abstract - ESMO 2021: ENHERTU® Superior to KADCYLA® in Patients with HER2 Positive Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA® given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.
ENHERTU® (Trastuzumab Deruxtecan) is an Antibody-Drug Conjugate (ADC) composed of a humanized monoclonal antibody specifically targeting HER2, with the amino acid sequence similar to Trastuzumab, a cleavable tetrapeptide-based linker, and a potent cytotoxic Topoisomerase I inhibitor as the cytotoxic drug (payload). ENHERTU® has a favorable pharmacokinetic profile and the tetrapeptide-based linker is stable in the plasma and is selectively cleaved by cathepsins that are up-regulated in tumor cells. Unlike KADCYLA®, ENHERTU® has a higher drug-to-antibody ratio (8 versus 4), released payload easily crosses the cell membrane with resulting potent cytotoxic effect on neighboring tumor cells regardless of target expression, and the released cytotoxic agent (payload) has a short half-life , thus minimizing systemic exposure.
In the DESTINY-Breast 01 Phase II registration trial involving patients with HER2-positive metastatic breast cancer, who had received two or more prior HER2 targeted therapies including KADCYLA®, the Objective Response Rate (ORR) was 60.9%, with 6% Complete Responses and 54.9% Partial Response, with a median response duration of 14.8 months. The median PFS was 16.4 months. This benefit was consistent across all key subgroups, including patients who had previously received PERJETA® therapy.
DESTINY-Breast 03 is a global, multicenter, open-label, randomized Phase III study, in which the efficacy and safety of ENHERTU® was compared with KADCYLA®, in patients with HER2-positive metastatic breast cancer previously treated with Trastuzumab and a Taxane. In this study, 524 pts were randomized 1:1 to receive ENHERTU® 5.4 mg/kg (N=261) or KADCYLA® 3.6 mg/kg (N=263) once every 3 weeks. The median patient age was 54 years and patients in both treatment groups were comparable in terms of baseline characteristics including age, HER2-positivity status, ECOG Performance Status, prior treatment for breast cancer, brain metastases, and prior cancer therapy with agents including Trastuzumab. The Primary endpoint was Progression Free Survival (PFS) by Blinded Independent Central Review (BICR). Secondary endpoints include Overall Survival (OS), Objective Response Rate (ORR), Duration of Response, PFS by investigator, and Safety.
At the time of the prespecified interim analysis of this study, the median follow up was approximately 16 months and the median PFS by BICR review was Not Reached with ENHERTU® and was 6.8 months with KADCYLA® (HR=0.28; P= 7.8 × 10−22). This represented a very statistically significant 72% reduction in the risk for progression or death with ENHERTU® compared to KADCYLA®. The investigator-assessed PFS was similar (25.1 versus 7.2 months, HR=0.26, P<0.0001). This PFS benefit was observed as early as 4 weeks and remained consistent throughout the follow up period. PFS was significantly higher with ENHERTU® in all prespecified key subgroups, including Hormone Receptor status, prior treatment with PERJETA®, visceral disease, number of prior lines of therapy, and the presence or absence of brain metastases. Majority of patients in the ENHERTU® group experienced a reduction in tumor size, and the ORR was significantly higher among patients in the ENHERTU® compared to those who received KADCYLA® (79.7% versus 34.2%; P<0.0001), with a near doubling of the Complete Response rate in the ENHERTU® group, at 16.1% compared to 8.7% in the KADCYLA® group. The estimated 12-month Overall Survival rate was 94.1% versus 85.9% respectively (HR=0.56; P=0.007), but was not considered significant as it did not cross the prespecified boundary for significance, likely due to the immaturity of the dataset.
Adjudicated treatment related Interstitial Lung Disease/pneumonitis was more common in the ENHERTU® compared with the KADCYLA® treatment arm, at rates of 10.5% and 1.9%, respectively and most of the events were Grade 1 or 2 in severity, and none at Grade 4 or 5 in either treatment group. Interstitial Lung Disease profile was of less concern, than was seen in previous trials of ENHERTU® in more heavily pretreated patients. All Left Ventricular Ejection Fraction decreases were Grade 1 or 2 and were seen in 2.7% of the ENHERTU® group and in 0.4% of KADCYLA® group.
The researchers concluded that ENHERTU® demonstrated a highly statistically significant and clinically meaningful improvement in Progression Free Survival, when compared to KADCYLA®, in patients previously treated with Trastuzumab and Taxane for HER2-positive metastatic Breast cancer, with manageable toxicity and a significant improvement in Interstitial Lung Disease profile. The authors added that these data support ENHERTU® becoming the standard of care for second line treatment of HER2-positive metastatic breast cancer.
Trastuzumab deruxtecan (T-DXd) vs trastuzumab emtansine (T-DM1) in patients (Pts) with HER2+ metastatic breast cancer (mBC): Results of the randomized phase III DESTINY-Breast03 study. Cortés J, Kim SB, Chung WP, et al. Presented at: European Society for Medical Oncology 2021 Virtual Congress. September 16-21, 2021; virtual. Abstract LBA1.
KEYTRUDA® in Combination with Chemotherapy Improves Overall Survival in Triple Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers.
The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. It appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway. Previously published studies have shown that presence of tumor-infiltrating lymphocytes was associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response, and unleashes the tumor-specific effector T cells. The rationale for combining chemotherapy with immunotherapy is that cytotoxic chemotherapy releases tumor-specific antigens, and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity. Single agent KEYTRUDA® in metastatic TNBC demonstrated durable antitumor activity in several studies, with Objective Response Rates (ORRs) ranging from 10-21% and improved clinical responses in patients with higher PD-L1 expression. When given along with chemotherapy as a neoadjuvant treatment for patients with high-risk, early-stage TNBC, KEYTRUDA® combination achieved Pathological Complete Response rate of 65%, regardless of PD-L1 expression. Based on this data, KEYTRUDA® in combination with chemotherapy was studied, for first line treatment of advanced TNBC.
KEYNOTE-355 is a randomized, double-blind, Phase III study, which evaluated the benefit of KEYTRUDA® in combination with one of the three different chemotherapy regimens, nab-Paclitaxel, Paclitaxel, or the non-taxane containing Gemzar/Carboplatin, versus placebo plus one of the three chemotherapy regimens, in patients with previously untreated or locally recurrent inoperable metastatic TNBC. In this study, 847 patients were randomized 2:1 to receive either KEYTRUDA® 200 mg IV on day 1 of each 21-day cycle along with either nab-Paclitaxel 100 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, Paclitaxel 90 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, or Gemcitabine 1000 mg/m2 IV plus Carboplatin AUC 2, IV on days 1 and 8 of each 21-day cycle (N= 566) or placebo along with one of the three chemotherapy regimens (N= 281). This study was not designed to compare the efficacy of the different chemotherapy regimens. Treatment was continued until disease progression. Patients were stratified by chemotherapy, PD-L1 tumor expression (CPS of 1 or higher versus CPS of less than 1), and prior treatment with the same class of neoadjuvant/adjuvant chemotherapy (Yes versus No). The baseline characteristics of treatment groups were well-balanced. The co-Primary end points of the trial were Progression Free Survival (PFS) and Overall Survival (OS) in patients with PD-L1-positive tumors, and in all patients. Secondary end points were Objective Response Rate (ORR), Duration of Response, Disease Control Rate, and Safety.
The authors had previously reported that KEYTRUDA® in combination with chemotherapy, significantly improved PFS in patients with CPS (Combined Positive Score) of 10 or greater. The median PFS was 9.7 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for placebo plus chemotherapy (HR=0.65, P=0.0012). This represented a 35% reduction in the risk of disease progression. Among patients with CPS of 1 or greater, the median PFS was 7.6 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for the placebo plus chemotherapy arm (HR= 0.74; P=0.0014). This however based on prespecified statistical criteria, was not considered statistically significant. Among the entire Intention-To-Treat (ITT) population, the median PFS was 7.5 months in the KEYTRUDA® plus chemotherapy group, compared with 5.6 months for chemotherapy plus placebo group (HR=0.82).
The researchers have now reported the Overall Survival results after a median follow up of 44.1 months. The OS in the subgroup of patients with PD-L1 CPS of 10 or more was significantly better with first line KEYTRUDA® plus chemotherapy versus placebo plus chemotherapy (23.0 months versus 16.1 months, respectively; HR=0.73; P=0.0093). This represented a 27% reduction in the risk of death with the KEYTRUDA® combination. Among this subgroup, factors significantly associated with superior outcomes included age 65 yrs and older, use of Paclitaxel as the on-study chemotherapy, no prior adjuvant therapy, de novo metastasis, Disease Free Interval of 12 months or more, and less than 3 metastatic sites. However, this OS benefit was not noted in the subgroup of patients with PD-L1 CPS scores of 1 or less. In this subgroup, the median OS was 17.6 months in the KEYTRUDA® group and 16 months in the placebo group (HR=0.86; P=Not Significant). The same was true among the Intention-To-Treat (ITT) population, including all randomized patients regardless of PD-L1 tumor status. In this patient group, the median OS was 17.2 months in the KEYTRUDA® group and 15.5 months in the placebo group, and this was not statistically significant (HR=0.89).
The authors concluded that these updated results support KEYTRUDA® in combination with chemotherapy as a new standard-of-care treatment regimen for patients with locally recurrent unresectable or metastatic Triple Negative Breast Cancer, whose tumors express PD-L1, with CPS of 10 or more.
KEYNOTE-355: Final results from a randomized, double-blind phase 3 study of first-line pembrolizumab + chemotherapy vs placebo + chemotherapy for metastatic TNBC. Cortes J, Cescon DW, Rugo HS. et al. European Society for Medical Oncology (ESMO) Annual Meeting 2021: Abstract LBA16. Presented on September 19, 2021.
Updated Overall Survival with KISQALI® plus FASLODEX® in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors. The most common subtype of metastatic breast cancer is Hormone Receptor-positive (HR-positive), HER2-negative breast cancer (65% of all metastatic breast tumors), and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients, with a median Overall Survival (OS) of 36 months. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies.
Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6, that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest.
In the MONALEESA-2 Phase III trial, KISQALI® in combination with FEMARA® (Letrozole) was compared to FEMARA® alone, in postmenopausal women with HR-positive, HER2-negative advanced breast cancer, who received no prior therapy for their advanced breast cancer. The addition of KISQALI® to FEMARA® significantly prolonged Progression Free Survival (PFS), compared to FEMARA® alone. In the MONALEESA-7 Phase III study, KISQALI® in combination with Tamoxifen or a Non-Steroidal Aromatase Inhibitor plus ZOLADEX® (Goserelin) was compared with placebo in combination with Tamoxifen or an Aromatase Inhibitor plus ZOLADEX®, in premenopausal or perimenopausal women with HR-positive, HER2- negative advanced breast cancer, who had not previously received endocrine therapy for advanced disease. In this study of premenopausal women, KISQALI® plus endocrine therapy significantly improved PFS and OS, compared with placebo plus endocrine therapy. The MONALEESA-7 trial recently reported an exploratory updated OS analysis with a median follow up of 53.5 months. In this analysis, KISQALI® plus endocrine therapy showed a clinically relevant and significant median OS benefit of 58.7 months compared with 48.0 months in the placebo plus endocrine therapy group.
MONALEESA-3 is a multicenter, international, randomized, double-blind, placebo-controlled Phase III study which compared the efficacy of KISQALI® in combination with FASLODEX® with FASLODEX® alone, among postmenopausal women with HR-positive, HER2-negative advanced breast cancer, who received no prior endocrine therapy or only one line of prior endocrine therapy for advanced disease. In this trial, 726 women were randomized, of whom 367 were treatment-naïve and 345 patients had received up to one line of prior endocrine therapy for advanced disease. Patients were randomized 2:1 to receive KISQALI® plus FASLODEX® (N=484) or placebo plus FASLODEX® (N=242). Treatment consisted of KISQALI® 600 mg orally daily 3 weeks on and 1 week off and FASLODEX® 500 mg IM on day 1 of each 28-day cycle, with an additional dose given on day 15 of cycle 1. Patients were stratified by the presence or absence of lung or liver metastases and prior endocrine therapy (first line versus second line). The median age in both groups was 63 years. The Primary endpoint was Progression Free Survival (PFS). Secondary end points included Overall Survival (OS), Overall Response Rate (ORR), and Safety. The authors had previously reported that in the MONALEESA-3 trial, there was a significant OS benefit for KISQALI® plus FASLODEX® versus placebo plus FASLODEX®, with an median OS of Not Reached (NR) versus 40.0 months (HR=0.72; P=0.00455).
The researchers in this publication reported an exploratory OS analysis update for MONALEESA-3, with an extended follow up (median, 56.3 months) in order to analyze the long-term OS benefit of KISQALI® plus FASLODEX® versus placebo plus FASLODEX®, similar to MONALEESA-7 trial. There was a significant OS benefit with a median OS of 53.7 months in the KISQALI® group versus 41.5 months in the placebo group (HR=0.73). The estimated 4-year survival rates were 54% and 45% for KISQALI® and placebo, respectively, while the 5-year survival rates were estimated to be 46% and 31% respectively. Subgroup analysis according to prior lines of endocrine therapy showed that patients in the first line subgroup had an median OS of Not Reached in the KISQALI® group and 51.8 months in the placebo group (HR=0.64). In the second line setting, the median OS was 39.7 months in the KISQALI® group versus 33.7 months in the placebo group (HR=0.78). No new safety signals were observed.
It was concluded that this extended follow up analysis of MONALEESA-3 is the longest reported follow up for any CDK4/6 inhibitor clinical trial in the postmenopausal women, and demonstrated a durable Overall Survival benefit with KISQALI® plus FASLODEX®, compared to FASLODEX® alone, in patients with HR-positive, HER2-negative advanced breast cancer. Further, this benefit was maintained when KISQALI® was given both as first line as well as second line therapy, and across subgroups of patients studied.
Ribociclib plus fulvestrant for postmenopausal women with hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer in the phase III randomized MONALEESA-3 trial: updated overall survival. Slamon DJ, Neven P, Chia S, et al. Annals of Oncology 2021; 32:1015-1024.
Duration of Adjuvant Aromatase Inhibitor Therapy in Postmenopausal Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Luminal breast cancer is the most prevalent molecular subtype in postmenopausal females, accounting for over 70%. Despite substantial improvements in adjuvant therapies, the risk of disease recurrence continues indefinitely, with more than half the recurrences occurring after the first 5 years following diagnosis. Following initial adjuvant endocrine therapy with Tamoxifen for 5 years, the addition of extended adjuvant therapy has resulted in 40% longer Disease Free Survival (DFS), when compared to placebo or no extended therapy. However the benefit of extending adjuvant Aromatase Inhibitor therapy for 5 years beyond the initial 5-year duration regimen is less well established. Further, the most effective duration of such extended adjuvant endocrine therapy remains unclear. Added to this dilemma are the side effects associated with Aromatase Inhibitor therapy including hot flushes, arthralgia, and bone pain, as well as treatment-induced osteoporosis, which can have a significant impact on patient’s quality of life. Researchers in the Secondary Adjuvant Long-Term Study with Arimidex [Anastrozole] (SALSA) prospectively investigated whether an additional 2 years or 5 years of Anastrozole therapy would result in better outcomes, following the initial 5 years of endocrine therapy, in postmenopausal women with Hormone Receptor-positive breast cancer.
The authors conducted a prospective, multicenter, randomized, Phase III trial, which included 3,470 eligible postmenopausal women with Stages I, II or III early stage breast cancer with no evidence of recurrence. Enrolled patients had invasive Hormone Receptor-positive breast cancer, and had received 5 years (plus or minus 12 months) of adjuvant endocrine therapy with Tamoxifen, Aromatase Inhibitors, or both sequentially, up until 12 months before randomization. Patients were randomly assigned 1:1 to receive Anastrozole 1 mg, orally daily, for either 2 additional years for a total of 7 years (N=1732) or 5 additional years for a total of 10 years (N=1738). The two treatment groups were well balanced. The median age at the time of randomization was 64 years, 72% of patients had tumors that were smaller than 2 cm, 66% had node-negative disease, and 19% had high-grade tumors. Stratification criteria included pathological tumor stage, pathological node stage, primary adjuvant endocrine therapy and adjuvant chemotherapy.
The primary analysis included all the patients who were still participating in the study (N=3208), including 1,603 in the 2-year group versus 1,605 in the 5-year group. In the primary analysis population of 3208 patients, 51% had received Tamoxifen alone for the initial 5 years, 7.3% had received an Aromatase Inhibitor alone, and 41.7% had received an Aromatase Inhibitor in combination with Tamoxifen. The Primary end point was Disease Free Survival (DFS). Secondary end points were Overall Survival (OS), contralateral breast cancer, second primary cancer, and clinical bone fracture. The median follow-up after randomization was 118 months.
The researchers observed no difference in DFS with 2 versus 5 additional years of adjuvant endocrine therapy with Anastrozole. The DFS 10 years since randomization was 73.6% in the 2-year group versus 73.9% in the 5-year group (HR=0.99; P=0.90). Contralateral breast cancer occurred in 2.2% versus 2.1% of patients (HR= 1.15), and local recurrence occurred in 3% versus 2.4% in the 2 year and 5 year groups, respectively. There was no difference noted for Overall Survival at 8 years between the two treatment groups (87.5% in the 2-year group and 87.3% in the 5-year group, HR for death from any cause=1.02). The risk of clinical bone fracture however was higher in the 5-year group than in the 2-year group (HR=1.35).
It was concluded from this study that in postmenopausal women with Hormone Receptor positive breast cancer who had received 5 years of adjuvant endocrine therapy, extending endocrine therapy with an Aromatase Inhibitor by an additional 5 years provided no benefit over a 2-year extension, but was associated with a greater risk of bone fracture.
Duration of Adjuvant Aromatase-Inhibitor Therapy in Postmenopausal Breast Cancer. Gnant M, Fitzal F, Rinnerthaler G, et al. N Engl J Med 2021; 385:395-405.
FDA Approves KEYTRUDA® for High Risk Early Stage Triple Negative Breast Cancer
SUMMARY: The FDA on July 26, 2021, approved KEYTRUDA® (Pembrolizumab) for high risk, early stage, Triple Negative Breast Cancer (TNBC), in combination with chemotherapy, as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment following surgery. The FDA also granted regular approval to KEYTRUDA® in combination with chemotherapy for patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (Combined Positive Score - CPS 10 or more), as determined by an FDA approved test. FDA granted accelerated approval to KEYTRUDA® for this indication in November 2020.
Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers.
The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. In addition to increasing the likelihood of tumor resectability and breast preservation, patients achieving a pCR following neoadjuvant chemotherapy have a longer Event Free Survival (EFS) and Overall Survival (OS). Those who do not achieve a pathological Complete Response tend to have a poor prognosis. For all these reasons, pCR is considered a valid endpoint for clinical testing of neoadjuvant therapy in patients with early stage TNBC. It appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway. Previously published studies have shown that presence of tumor-infiltrating lymphocytes was associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response and unleashes the tumor-specific effector T cells. Cytotoxic chemotherapy releases tumor-specific antigens and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity. Preliminary results from Phase I and II trials have shown that in patients with TNBC, KEYTRUDA® given along with chemotherapy in a neoadjuvant setting resulted in a high rate of pCR.
The present FDA approvals were based on KEYNOTE-522, which is an international, randomized, multicenter, double-blind, placebo controlled Phase III trial, conducted to evaluate the safety and efficacy of neoadjuvant KEYTRUDA® plus chemotherapy followed by adjuvant KEYTRUDA® or placebo, in patients with early stage TNBC. In this study, 1,174 patients were randomly assigned in a 2:1 ratio to receive neoadjuvant KEYTRUDA® 200 mg IV every 3 weeks (N=784) or placebo (N=390). All patients received 4 cycles of Carboplatin plus Paclitaxel, followed by 4 cycles of Doxorubicin or Epirubicin plus Cyclophosphamide, in the neoadjuvant setting. Following definitive surgery, adjuvant KEYTRUDA® or placebo was continued every 3 weeks for 9 cycles or until disease recurrence or unacceptable toxicity. Enrolled TNBC patients were newly diagnosed, early stage, high risk, treatment naïve, and included both node-negative and node-positive patients with nonmetastatic disease (Tumor Stage T1c, Nodal Stage N1-N2 or Tumor Stage T2-T4, Nodal Stage N0-N2, per AJCC criteria). Patients were enrolled regardless of tumor PD-L1 expression. Treatment groups were well balanced and patients were stratified according to nodal status, tumor size, and Carboplatin schedule (weekly versus every 3 weeks). The two Primary endpoints were pathological Complete Response (pCR) at the time of definitive surgery and Event Free Survival (EFS).
At the first interim analysis, at a median follow up of 15.5 months, the pCR among the first 602 patients who underwent randomization was 64.8% in the KEYTRUDA® plus chemotherapy group, compared with 51.2% in the placebo plus chemotherapy group (HR=0.63; P<0.001). At the median follow-up of 39 months, EFS data were made available, and this showed that KEYTRUDA® demonstrated a statistically significant EFS benefit compared with chemotherapy alone. The number of patients who experienced an EFS event was 16% and 24%, respectively (HR=0.63; P=0.00031). Among patients who were in the PD-L1 positive, defined as those with a CPS of 1 or higher, there was a 33% reduced risk of EFS events with KEYTRUDA® compared with the placebo group (HR=0.67). In the PD-L1 negative group, patients receiving the KEYTRUDA® combination had a reduced risk for EFS events by 52% compared with the placebo-chemotherapy group (HR=0.48). Across all treatment phases, Grade 3 or higher treatment-related toxicities were 78.0% in the KEYTRUDA® plus chemotherapy group and 73.0% in the placebo plus chemotherapy group
It can be concluded from this study that among patients with early stage Triple Negative Breast Cancer, the addition of KEYTRUDA® to neoadjuvant chemotherapy significantly increased the pathological Complete Response rate, compared to those who received placebo plus neoadjuvant chemotherapy, with a statistically significant Event Free Survival benefit. This KEYTRUDA® combination therapy is a meaningful milestone for breast cancer patients, and is the first immunotherapy regimen to be approved in high risk, early stage Triple Negative Breast Cancer.
https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pembrolizumab-high-risk-early-stage-triple-negative-breast-cancer.
De-escalated Neoadjuvant Dual Anti-HER2 Blockade and Survival Outcomes in Early Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Adjuvant and neoadjuvant chemotherapy given along with anti-HER2 targeted therapy reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage, as well as advanced metastatic breast cancer.
Trastuzumab is a humanized monoclonal antibody targeting HER2. It binds to the extracellular subdomain IV of the receptor and disrupts ligand independent HER2 downstream cell signaling pathways. Pertuzumab is a recombinant, humanized, monoclonal antibody that binds to the HER2 subdomain II and blocks ligand dependent HER2 heterodimerization with other HER receptors. Thus Trastuzumab along with Pertuzumab provide a more comprehensive blockade of HER2 driven signaling pathways. Dual HER2 blockade with Trastuzumab and Pertuzumab, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies.
Pathological Complete Response (pCR) after neoadjuvant therapy has strong prognostic significance in HER2+ breast cancer and pCR rates in HER2+/HR− tumors exceed those in HER2+/HR+ tumors, and this in turn correlates with superior Event Free Survival. The FDA approved anti-HER2 dual blockade with Pertuzumab and Trastuzumab, given along with chemotherapy for the neoadjuvant treatment of patients with HER2-positive, locally advanced, inflammatory, or early stage breast cancer, based on the NeoSphere trial, and for metastatic disease based on positive survival results in the CLEOPATRA trial. The role of chemotherapy free anti-HER2 dual blockade however has remained unclear.
ADAPT (Adjuvant Dynamic marker-Adjusted Personalized Therapy) is one of the first new generation neoadjuvant trials addressing individualization of neoadjuvant therapy in early breast cancer and was initiated to establish early predictive surrogate markers such as Ki-67 for therapy response following a short course of induction treatment, in order to maximally individualize therapy and avoid unnecessary toxicity by ineffective treatment. West German Study Group (WSG)-ADAPT trials were designed separately for HER2+/HR− and HER2+/HR+ breast cancer sub groups as they are biologically distinct, with HER2+/HR− subgroup being more aggressive but also more chemosensitive, as evidenced by response to neoadjuvant therapy and subsequent survival rates.
The WSG-ADAPT HER2+/HR- trial is a multicenter, prospective Phase II/III trial which assessed whether patients with strong early response to dual HER2 blockade alone might achieve pathological Complete Response (pCR), comparable to that of patients receiving dual HER2 blockade and chemotherapy. In this study, 134 patients (N=134) with cT1-cT4c, cN0-3 HER2+/HR- early breast cancer were randomly assigned 5:2 to either receive Trastuzumab and Pertuzumab alone in arm A or with added Paclitaxel in arm B. In Arm A, patients (N = 92) received a loading dose of Trastuzumab 8 mg/kg IV and then 6 mg/kg three weekly along with Pertuzumab 840 mg loading dose and then 420 mg 3 weekly, for a total of 12 weeks. In Arm B (N=42), in addition to Trastuzumab and Pertuzumab as administered in Arm A, Paclitaxel 80 mg/m2 IV was given every week for 12 weeks. Eligible patients had tumors that were ER and PR negative (less than 1%) and HER2-positive (IHC 2+ and FISH positive or IHC 3+ positive). After neoadjuvant treatment, surgery or histological confirmation of non-pCR by core needle biopsy within 3 weeks was mandatory. Pathological Complete Response (pCR) at surgery was defined as no invasive tumor residuals in breast and lymph nodes.
The Primary endpoint was pCR and omission of further chemotherapy was allowed in patients with pCR, and Secondary endpoints included 5 year distant Disease Free Survival (DFS) and Overall Survival (OS). An important objective of this study was to identify an early-responder population with regard to pCR after Trastuzumab and Pertuzumab alone treatment, which is strongly associated with long-term prognosis particularly in HER2+/HR− breast cancer, and assess whether this population might be spared neoadjuvant chemotherapy. Early response was defined as proliferation decrease 30% or more of Ki-67 (compared to baseline) or low cellularity (less than 500 invasive tumor cells) in the 3-week biopsy. The trial was stopped early due to the observed pCR superiority in the dual HER2 blockade plus chemotherapy group. The authors previously reported that the pCR rate in the dual HER2 blockade plus chemotherapy group was 90.5%, compared with 36.3% in the dual HER2 blockade alone. The authors have now reported the first survival data.
After a median follow-up of 5 years, there were no significant differences in DFS, distant DFS and OS between the 2 study groups. The invasive DFS rate was 98% with dual HER2 blockade and chemotherapy and 87% with dual HER2 blockade alone (HR=0.32; P=0.144). Distant DFS was 98% and 92%, respectively (HR=0.34; P=0.313) and Overall Survival was 98% and 94% (HR=0.41; P=0.422). The achievement of a pathologic Complete Response following 12 weeks of treatment was strongly associated with improved invasive DFS at 5 years, irrespective of study group (98.5% versus 82%; HR=0.14; P=0.011). The researchers also examined the benefit of neoadjuvant chemotherapy-free dual HER2 blockade alone and noted that no pCR was observed in patients with low HER2 expression (IHC 1+ or 2+ and FISH positive) and/or basal-like subtype as detected by PAM50 assay. In the total study population, low HER2 expression and/or no early response was strongly associated with worse distant DFS (P=0.029) and invasive DFS (P=0.068).
The authors concluded that excellent pCR and Survival can be accomplished in patients treated by de-escalated 12-week neoadjuvant weekly Paclitaxel and dual HER2 blockade, irrespective of additional chemotherapy use. They added that early pCR after only 12 weeks of neoadjuvant Paclitaxel plus dual HER2 blockade was strongly associated with improved outcome, and may thus serve as a predictive clinical marker for further treatment de-escalation.
De-escalated neoadjuvant pertuzumab+trastuzumab with or without paclitaxel weekly in HR-/HER2+ early breast cancer: ADAPT-HR-/HER2+ biomarker and survival results. Harbeck N, Gluz O, Christgen M, et al. J Clin Oncol 39, 2021 (suppl 15; abstr 503)
Platinum Chemotherapy Inferior to XELODA® in Triple Negative Breast Cancer Following Neoadjuvant Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Breast cancer is heterogeneous malignancy and using global gene expression analyses, 6 breast cancer intrinsic subtypes have been established. They include Luminal A, Luminal B, HER2-enriched, Claudin-low, Basal-like, and a Normal breast-like group. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients and African American females. It is a surrogate for the inherently aggressive Basal-like breast cancer subtype. This group has the worse prognosis compared to other breast cancer subtypes and is usually aggressive, and tumors tend to be high grade. Patients with TNBC are at a higher risk for both local and distant recurrence and often develop visceral metastases. Those with metastatic disease have one of the worst prognoses of all cancers, with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. Basal-like breast cancer subtype is also a marker of hereditary breast cancer susceptibility. Multiparity may increase the risk of TNBC and reduce the likelihood of developing ER-positive breast cancer. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge.
Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. In addition to increasing the likelihood of tumor resectability and breast preservation, patients achieving a pCR following neoadjuvant chemotherapy have a longer Event Free Survival (EFS) and Overall Survival (OS). Those who do not achieve a pathological Complete Response tend to have a poor prognosis. For all these reasons, pCR is considered a valid endpoint for clinical testing of neoadjuvant therapy in patients with early stage TNBC. Patients with TNBC are at a high risk for recurrence if they have residual invasive disease, following completion of standard neoadjuvant chemotherapy.
In the Phase III CREATE-X trial, the addition of adjuvant XELODA® (Capecitabine) therapy was found to be safe and effective in prolonging Disease Free Survival (DFS) and Overall Survival (OS) among patients with HER2-negative breast cancer, who had residual invasive disease on pathological evaluation, following neoadjuvant chemotherapy (N Engl J Med 2017;376:2147-2159).
Based on the preclinical models supporting the use of platinum agents in the TNBC Basal-like subtype, the EA1131 trial was conducted to test the hypothesis that adjuvant platinum chemotherapy would improve invasive DFS compared with XELODA®, in patients with clinical Stage II-III TNBC, who had Basal-like subtype invasive residual disease in the breast, following neoadjuvant chemotherapy. The aim of this study was to assess whether platinum chemotherapy would be as effective, or more effective than XELODA® (noninferiority design with superiority alternative - Hybrid design).
In this study, 410 patients with clinical Stage II or III TNBC who had completed at least one full cycle of taxane with or without anthracycline-containing neoadjuvant chemotherapy were randomly assigned to receive XELODA® 1000 mg/m2 orally twice daily, days 1-14, every 3 weeks, for a total of six cycles, or a platinum agent (treating physician choice of Cisplatin 75 mg/m2 or Carboplatin AUC 6 on day 1) IV, once every 3 weeks, for a total of four cycles. Radiation Therapy before or after study treatment completion, was required for all patients after breast-conservation surgery. Postmastectomy Radiation Therapy was required for patients with primary tumors more than 5 cm or those with 4 or more positive axillary lymph nodes. TNBC subtype (Basal versus non-Basal) was determined by PAM50 in the residual disease. The Primary end point was invasive DFS (time from random assignment to the earliest disease recurrence, invasive contralateral cancer, second primary cancer, or death) in patients with Basal subtype TNBC.
After a recent interim analysis, the Data and Safety Monitoring Committee recommended stopping the trial, as it was unlikely that further follow up would show noninferiority or superiority of platinum chemotherapy. After a median follow up of 20 months, the 3-year invasive DFS among the 308 patients with Basal subtype TNBC for platinum chemotherapy was 42% versus 49% for XELODA®. Further, Grade 3 and 4 toxicities were more common in the platinum chemotherapy group. The 3-year Relapse Free Survival as well as Overall Survival was also in favor of XELODA® group versus Platinum group. There was no benefit noted with platinum chemotherapy in any of the subsets of randomized patients.
It was concluded from this study that platinum agents do not improve outcomes in patients with Basal subtype TNBC, who have residual disease following neoadjuvant chemotherapy, and are associated with more severe toxicities, when compared with XELODA®. All participants in this study had a lower than expected 3-year invasive DFS regardless of study treatment, highlighting the need for better therapies in this high-risk population. The authors added that these study findings have an immediate impact in clinical practice, and adjuvant use of platinum agents in this patient population should only be considered in the context of a clinical trial.
Randomized Phase III Postoperative Trial of Platinum-Based Chemotherapy Versus Capecitabine in Patients With Residual Triple-Negative Breast Cancer Following Neoadjuvant Chemotherapy: ECOG-ACRIN EA1131. Mayer IA, Zhao F, Arteaga CL, et al. DOI: 10.1200/JCO.21.00976 Journal of Clinical Oncology. Published online June 06, 2021.
Late Breaking Abstract – ASCO 2021: Adjuvant LYNPARZA® Improves Disease Free Survival in BRCA Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. Mutations in BRCA1 and BRCA2 account for about 20 to 25 percent of hereditary breast cancers and about 5 to 10 percent of all breast cancers. These mutations can be inherited from either of the parents and a child has a 50 percent chance of inheriting this mutation, and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal. Patients with BRCA mutations can present with aggressive, high-risk disease and are at a high risk of recurrence following completion of multimodality therapy including surgery, radiation, and chemotherapy. This is an area of unmet need, warranting identification of additional novel and effective therapies.
BRCA1 and BRCA2 are tumor suppressor genes and they recognize and repair double strand DNA breaks via Homologous Recombination (HR) pathway. Homologous Recombination is a DNA repair pathway utilized by cells to accurately repair DNA double-stranded breaks during the S and G2 phases of the cell cycle, and thereby maintain genomic integrity. The PARP (Poly ADP Ribose Polymerase) family of enzymes include PARP1 and PARP2, and is a related enzymatic pathway that repairs single strand breaks in DNA. In a BRCA mutant, the cancer cell relies solely on PARP pathway for DNA repair to survive.
LYNPARZA® is a PARP inhibitor, that traps PARP onto DNA at sites of single-strand breaks, thereby preventing their repair and generate double-strand breaks. These breaks cannot be repaired accurately in tumors harboring defects in Homologous Recombination Repair pathway genes, such as BRCA1 or BRCA2 mutations, and this leads to cumulative DNA damage and tumor cell death. LYNPARZA® is presently approved by the FDA for metastatic HER2-negative breast cancer with BRCA1/2 germline mutation. The researchers in this study evaluated the benefit of LYNPARZA® in patients with germline BRCA-mutated, HER2-negative, early stage breast cancer.
OlympiA is a multicenter, randomized, placebo-controlled, double-blind, Phase III trial of adjuvant LYNPARZA® after neoadjuvant/adjuvant chemotherapy, in patients with germline BRCA1/2 mutations, and high-risk HER2-negative early breast cancer. This trial enrolled 1836 patients, including triple negative and hormone receptor positive breast cancer. All enrolled patients had already received standard adjuvant or neoadjuvant chemotherapy, surgery and if needed, radiation therapy, for early stage breast cancer (Stage II-III). Inclusion criteria also required that patients have a high risk of disease recurrence and those with lower risk of invasive disease recurrence were excluded. For example, patients with hormone receptor positive breast cancer had 4 or more positive lymph nodes prior to adjuvant chemotherapy. Patients were randomized 1:1 to receive LYNPARZA® 300 mg PO BID continuously for 1 year (N=921) or placebo (N=915). Endocrine therapy and bisphosphonates were allowed. The Primary endpoint was invasive Disease Free Survival (IDFS) and Secondary endpoints included distant DFS (DDFS), Overall Survival (OS) and Safety. The study results were reported early, at a median follow up of 2.5 years, after a planned interim analysis was reviewed by an Independent Data Monitoring Committee.
At the pre-specified interim analysis (2.5 years), the estimated 3-year invasive DFS (IDFS) was 85.9% for patients who received LYNPARZA® compared with 77.1% for those who received placebo (HR=0.58; P<0.001), representing a 42% reduction in the risk of IDFS with LYNPARZA® compared to placebo. The estimated 3-year distant DFS (DDFS) was 87.5% versus 80.4% respectively (HR=0.57; P<0.001). This represented a 43% reduction in DDFS with adjuvant LYNPARZA® compared to placebo. At the time of this interim analysis, Overall Survival data were considered immature. The side effects were consistent with the known safety profile of LYNPARZA®, and no new safety signals were noted during the trial.
The authors concluded that adjuvant LYNPARZA® following adjuvant or neoadjuvant chemotherapy significantly improved invasive DFS and distant DFS with acceptable toxicity, in patients with germline BRCA mutated, and high risk HER-2 negative early stage breast cancer. The authors added that this is the first study to report the benefit of a PARP inhibitor given as adjuvant therapy on survival endpoints, in this patient group. Overall Survival data are awaited, as follow up data matures.
Adjuvant Olaparib for Patients with BRCA1- or BRCA2-Mutated Breast Cancer. Tutt AJ, Garber JE, Kaufman B, et al. June 3, 2021, DOI: 10.1056/NEJMoa2105215.
70-Gene Risk Signature May Determine Long Term Benefit of Endocrine Therapy in Premenopausal Breast Cancer Patients
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. Approximately 75% of patients with breast cancer are Hormone Receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues.
ZOLADEX® (Goserelin) is a potent synthetic analogue of Luteinizing Hormone-Releasing Hormone (LHRH), also known as a Gonadotropin Releasing Hormone (GnRH) agonist analogue. It stimulates the production of the sex hormones Testosterone and Estrogen in a non-pulsatile (non-physiological) manner, resulting in the disruption of the endogenous hormonal feedback systems, and down-regulation of Testosterone and Estrogen production. Given that premenopausal patients with breast cancer have their disease diagnosed earlier in life, they are at an increased risk of fatal disease during their lifetime. The long term benefit of endocrine therapy, including ovarian suppression, has not been studied in this patient population.
Zipp-trial (STO-5) in one of three well defined randomized, controlled, clinical studies of adjuvant endocrine therapy, conducted between 1990 and 1997 by the Stockholm Breast Cancer Study Group. This trial included premenopausal patients with invasive breast cancer treated with a modified radical mastectomy or breast conserving surgery and axillary lymph node dissection or biopsy. Patients undergoing breast conserving surgery also received adjuvant radiotherapy to the breast (50 Gy over 5 weeks). Patients were included in study irrespective of ER status. All patients with node positive disease electively received adjuvant cytotoxic chemotherapy and those with four or more lymph node metastases received radiotherapy as well.
The Stockholm part of the Zoladex In Premenopausal Patients (ZIPP-trial, STO-5) included 924 patients and the purpose of this analysis was to examine the long-term 20-year benefit of ZOLADEX® and Tamoxifen, stratified by the molecular 70-gene risk prediction signature in this patient population. Patients were stratified by lymph node status and divided into 3 groups: patients with lymph node-negative status, those with 1-3 positive lymph nodes who had received chemotherapy, and those with 4 or more positive lymph nodes who received chemotherapy and locoregional radiotherapy. All of these patients were included in a 2X2 factorial randomization to receive ZOLADEX® 3.6 mg Subcutaneously every 28 days (N=230), Tamoxifen 40 mg orally daily (N=231), ZOLADEX® plus Tamoxifen (N=230), or no endocrine therapy (N = 233), for 2 years. Node-positive patients received adjuvant chemotherapy in addition to endocrine therapy. The median age was 46 years.
The researchers identified clinically relevant breast cancer markers by immunohistochemistry in 729 patients, of whom 610 patients had Hormone Receptor-positive tumors. Molecular risk classification data were available in 465 patients and the 70-gene signature classified patients into groups with either Low risk (N=306) or High risk of disease recurrence (N=159). Of the 610 patients with Hormone Receptor-positive tumors, 160 received ZOLADEX®, 142 received Tamoxifen, 156 received the combination, and 152 received no endocrine therapy (control group).
The researchers noted that the 20-year risk for distant recurrence was significantly reduced in the patients who received ZOLADEX®, Tamoxifen or both, compared with those who did not receive endocrine therapy. The respective Hazard Ratios (HRs) were 0.48, 0.59, and 0.67 after adjusting for prior therapy and tumor characteristics. Stratification by the 70-gene signature demonstrated that Low risk patients derived a significant benefit from Tamoxifen therapy (HR=0.38), whereas ZOLADEX® plus Tamoxifen provided less benefit to these patients ((HR=0.80 and 0.72, respectively). In contrast, patients at High risk had significant benefit from ZOLADEX® therapy (HR=0.22), whereas less benefit was observed with Tamoxifen or ZOLADEX® plus Tamoxifen (HR=0.69 and 0.64, respectively).
The authors concluded that long term endocrine therapy benefit in premenopausal patients is influenced by molecular risk classification, with significant benefit from ZOLADEX® noted in High risk patients, whereas Low risk patients benefit from Tamoxifen.
LBA1 - 20-year benefit of endocrine therapy in premenopausal breast cancer patients by the 70-gene risk signature. Johansson A, Dar H, Van ‘T Veer L, et al. DOI:https://doi.org/10.1016/j.annonc.2021.03.210
Role of Chemotherapy in Postmenopausal Women with Node Positive Early Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer were diagnosed in 2020 and about 42,170 women died of the disease. Approximately 25% of patients with Hormone Receptor (HR)-positive, HER2-negative early breast cancer have metastatic lymph node involvement and two third of these patients are postmenopausal. Majority of these patients currently receive chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for early stage, HR-positive, HER-negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
In the ground-breaking TAILORx (Trial Assigning Individualized Options for Treatment) study which enrolled 10,273 patients with HR-positive, HER2-negative, axillary node-negative breast cancer, patients were divided into three groups based on their Recurrence Score. Patient with Intermediate Recurrence Score of 11-25 were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. There was no benefit noted from adding chemotherapy to endocrine therapy, for women older than 50 years in this Intermediate RS group, suggesting that a significant percentage of women with node-negative breast cancer do not achieve substantial benefit from chemotherapy. For women 50 years old or younger who received chemotherapy and had a Recurrence Score of 16 to 25, there was a lower rate of distant recurrence and the risk of recurrence and benefit of chemotherapy was further influenced by the tumor size and grade. Whether the results of TAILORx can be extrapolated to women with node-positive breast cancer has remained unclear. It is estimated that approximately 85% of women with node-positive disease have Recurrence Score results of 0-25.
The RxPONDER trial was designed to determine the benefit of chemotherapy, in patients who had a Recurrence Score of 0-25. This trial did not include pre and postmenopausal women with Recurrence Score results 26-100 based on previously published studies suggesting that this patient group benefited from chemotherapy. SWOG S1007 (RxPONDER) is an multicenter, international, prospective, randomized, Phase III trial, in which patients with HR-positive, HER2-negative breast cancer with 1-3 positive axillary lymph nodes were included, to determine which patients would benefit from chemotherapy and which patients could safely avoid it. In this study, a total of 5083 HR-positive, HER2-negative breast cancer patients with 1-3 positive lymph nodes and Oncotype DX Recurrence Score of less than 25 were randomly assigned 1:1 to receive chemotherapy plus endocrine therapy or endocrine therapy alone. Approximately two-thirds of patients were postmenopausal and one-third were premenopausal and had no contraindications to taxane and/or anthracycline based chemotherapy. Patients were stratified by Recurrence Score (0-13 versus 14-25), menopausal status, and axillary nodal dissection versus sentinel node biopsy. The Primary endpoint was Invasive Disease Free Survival (IDFS), defined as local, regional, or distant recurrence, any second invasive cancer, or death from any cause, and whether the effect depended on the Recurrence Score. Secondary endpoints included Overall Survival (OS).
At a median follow up of 5.1 years, there was no association noted between Recurrence Score (RS) values and chemotherapy benefit for the entire study population (P=0.30). However, a prespecified analysis did show a significant association between chemotherapy benefit and menopausal status. Premenopausal women (N=1665) with an RS between 0 and 25 had an IDFS benefit with the addition of chemotherapy to endocrine therapy compared with endocrine therapy alone (94.2% versus 89%, HR=0.54; P=0.0004). This absolute 5.2% benefit in the premenopausal subset was highly significant. The relative risk reduction with the addition of chemotherapy to endocrine therapy for the two RS risk groups 0-13 and 14-25 was consistent in the premenopausal population, with an overall Hazard Ratio of 0.54. The absolute benefit was numerically higher in those with RS 14-25. Consistent benefit was again noted regardless of number of involved lymph nodes, although there was slight variation in the absolute benefit. Postmenopausal women (N=3350) did not benefit with the addition of chemotherapy to endocrine therapy when compared endocrine therapy alone, regardless of Recurrence Score (91.9% versus 91.6%, HR=0.97; P=0.82). Chemotherapy also improved Overall Survival in the premenopausal cohort, although the follow up is limited.
It was concluded from this practice-changing outcomes that postmenopausal women with HR-positive, HER2-negative breast cancer with 1-3 positive nodes and Oncotype DX Recurrence Score of 25 or less can safely avoid receiving adjuvant chemotherapy, whereas premenopausal patients with 1-3 positive nodes and a Recurrence Score of 25 or less should consider adjuvant chemotherapy. The authors added that these finding demonstrate that the great majority of postmenopausal women can be spared unnecessary chemotherapy and receive only endocrine therapy.
First results from a phase III randomized clinical trial of standard adjuvant endocrine therapy ± chemotherapy in patients with 1-3 positive nodes, hormone receptor-positive and HER2-negative breast cancer with recurrence scores ≤ 25: SWOG S1007 (RxPONDER). Kalinsky K, Barlow WE, Meric-Bernstam F, et al. 2020 San Antonio Breast Cancer Symposium. Presented December 10, 2020. Abstract GS3-00.
FDA Grants Regular Approval to TRODELVY® for Advanced Triple Negative Breast Cancer
SUMMARY: The FDA on April 7, 2021, granted regular approval to TRODELVY® (Sacituzumab govitecan) for patients with unresectable locally advanced or metastatic Triple Negative Breast Cancer (mTNBC), who have received two or more prior systemic therapies, at least one of them for metastatic disease. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence.
Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients and African American females. It is usually aggressive, and tumors tend to be high grade, and patients with TNBC are at a higher risk of both local and distant recurrence and often develop visceral metastases. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Overall survival among patients with pretreated metastatic TNBC has not changed over the past 2 decades and standard chemotherapy is associated with low response rates of 10-15% and a Progression Free Survival of only 2-3 months.
TRODELVY® is an Antibody-Drug Conjugate (ADC) in which SN-38, an active metabolite of Irinotecan, a Topoisomerase I inhibitor, is coupled to the humanized Anti-Trophoblast cell-surface antigen 2 (Trop-2) monoclonal antibody (hRS7 IgG1κ), through the cleavable CL2A linker. SN-38 cannot be given directly to patients because of its toxicity and poor solubility. Trop-2, a transmembrane calcium signal transducer, stimulates cancer-cell growth, and this cell surface receptor is overexpressed in several epithelial cancers including cancers of the breast, colon and lung, and has limited expression in normal human tissues. Trop-2 is expressed in more than 85% of breast tumors including Triple Negative Breast Cancer. Upon binding to Trop-2, the anti-TROP-2 monoclonal antibody is internalized and delivers SN-38 directly into the tumor cell, making it a suitable transporter for the delivery of cytotoxic drugs. Further, the cleavable linker enables SN-38 to be released both intracellularly into the tumor cells, as well as the tumor microenvironment, thereby allowing for the delivery of therapeutic concentrations of the active drug in bystander cells to which the conjugate has not bound. Thus, TRODELVY®-bound tumor cells are killed by intracellular uptake of SN-38, whereas the adjacent tumor cells are killed by the extracellular release of SN-38.
The FDA granted accelerated approval to TRODELVY® in April 2020 based on Objective Response Rate of 33.3% and Duration of Response of 7.7 months in a Phase I/II study. The ASCENT trial served as a confirmatory analysis, expanding the previous TRODELVY® indication to include treatment in adult patients with unresectable locally advanced or metastatic TNBC who have received two or more prior systemic therapies, at least one of them for metastatic disease.
The ASCENT study is an open-label, multicenter, active-controlled, randomized, confirmatory Phase III trial in which 529 patients with unresectable locally advanced or metastatic TNBC patients were enrolled. Eligible patients had relapsed/refractory disease and had received two or more prior systemic therapies (including a taxane), one of which could be in the neoadjuvant or adjuvant setting, if disease progression occurred within 12 months. Patients were randomly assigned 1:1 to receive TRODELVY® 10 mg/kg IV on days 1 and 8 of a 21-day cycle (N=267) or physician’s choice of single-agent chemotherapy (N= 262). The Primary endpoint was Progression Free Survival (PFS) in patients without brain metastases at baseline (N=468), as measured by a blinded Independent Centralized Review. Secondary endpoints included PFS for the total population (with and without brain metastases), Overall Survival (OS), Objective Response Rates (ORR) and Safety.
Among all randomly assigned patients (with and without brain metastases), the median PFS for patients receiving TRODELVY® was 4.8 months, compared with 1.7 months in those receiving chemotherapy (HR=0.43; P <0.0001). This represented a statistically significant and clinically meaningful 57% reduction in the risk of disease progression or death. The median OS was 11.8 months and 6.9 months respectively, in favor of TRODELVY® (HR= 0.51; P<0.0001), representing a 49% reduction in the risk of death. The most common adverse reactions in patients receiving TRODELVY® were fatigue, rash, decreased appetite, nausea, vomiting diarrhea, constipation, alopecia, anemia and abdominal pain.
It was concluded that ASCENT is the first Phase III study of an Antibody Drug Conjugate, with significant PFS and OS improvement over Standard-of-Care chemotherapy, in pretreated patients with metastatic Triple Negative Breast Cancer, fulfilling an unmet medical need.
ASCENT: A randomized phase III study of sacituzumab govitecan (SG) vs treatment of physician’s choice (TPC) in patients (pts) with previously treated metastatic triple-negative breast cancer (mTNBC). Bardia A, Tolaney SM, Loirat D, et al. ESMO Virtual Congress 2020. Abstract LBA17. Presented September 19, 2020.
Advances in Triple Negative Breast Cancer
Written by: Debra Patt, MD, PhD, MBA
Content Sponsored by: Bristol Myers Squibb
Dr. Patt is a paid consultant for BMS and was compensated for her contribution in drafting this article.
Metastatic triple negative breast cancer (TNBC) is a devastating disease, making up 15% of all cancers, and having a limited outcome with an overall survival average of around a year.1,2 It is a diagnosis of exclusion, as tumor cells do not express the targetable hormone receptors (estrogen or progesterone receptors) or HER2, thus treatment options have historically relied on systemic chemotherapy rather than targeted treatment.3 This aggressive subtype is often associated with an earlier age of onset and an aggressive clinical course. Ethnic disparities have been identified for triple negative disease, with the incidence highest among patients who have a non-Hispanic black ethnic background compared to other ethnic groups.1 Furthermore, African American women are more likely to develop metastases compared to women of other races. Metastatic progression for triple-negative disease is generally characterized by early relapse and predominantly visceral (including liver, pulmonary and central nervous system) metastases.3
Historically, advances in the treatment of triple negative breast cancer have been rare. Multiple immunotherapy options in combination with chemotherapy are now approved in metastatic TNBC for patients with PD-L1 positive, first-line disease, and today there is much excitement about further evidence supporting its use in the metastatic and early stage settings.4 However, no head-to-head data exists to identify the optimal chemotherapy partner for checkpoint inhibition and not all chemotherapy agents appear to provide similar efficacy based on current data, hence more investigations are needed.5,6 Furthermore, while the incidence of immune-related adverse events such as endocrinopathies are low, the permanence of these side effects, particularly in the early stage setting, is concerning to some and should be closely monitored.
Germline BRCA mutations occur in approximately 10–30% of TNBC cases.7 In previously treated metastatic disease, the use of poly (ADP-ribose) polymerase (PARP) inhibitors in germline BRCA mutation positive patients has also shown improvements in survival, with the main reported side effects being hematologic, fatigue and diarrhea.3,8 In heavily pretreated metastatic TNBC patients, the use of antibody-drug conjugates has also resulted in anti-cancer effects.9
While advances in the aggressive and difficult-to-treat triple negative breast cancer subset are promising, all of these recent advances leave us with new treatment options but also unanswered questions. Our knowledge is limited and certainly will improve over time as we understand better predictors of outcome like PD-L1 expression, tumor infiltrating lymphocytes, and other factors as well as the importance of chemotherapy backbone choice. Other agents are now available for previously treated metastatic TNBC patients and further studies will be needed to assess the efficacy of these agents in earlier lines of therapy. Additionally, long-term follow up of studies will also be important to truly understand the impact of these new targeted approaches and the impact of drug tolerability on efficacy and patient quality of life.
References
1. DeSantis CE, Fedewa SA, Sauer AG, Kramer JL, Smith RA, Jemal A. CA Cancer J Clin. 2016;66:31-42.
2. Marra A, Viale G, Curigliano G. BMC Medicine. 2019;17:90-99.
3. Bergin ART, and Loi S. F1000Research. 2019;8: F1000 Faculty Rev-1342. Published online 2019 Aug 2.
4. Simmons CE, Brezden-Masley C, McCarthy J, McLeod D, Joy AA. Ther Adv Med Oncol. 2020;12:1-15.
5. Cortes J, Cescon DW, Rugo HS, Nowecki Z, Im SA, et al. DOI: 10.1200/JCO.2020.38.15_suppl.1000 Journal of Clinical Oncology 38, no. 15_suppl (May 20, 2020) 1000-1000.
6. Miles D, Gligorov J, Andre F, Cameron D, Schneeweiss A, et al. Annals of Oncology. 2020;31 (suppl 4):S1142-S1215. 10.1016/annonc/annonc325.
7. Vagia E, Mahalingam D, Cristofanilli M. Cancers (Basel). 2020 Apr;12:916-941.
8. Madariaga A, Bowering V, Ahrari S, Oza AM, and Lheureux S. Int J Gynecol Cancer. 2020; 30:903-915.
9. Bardia A, Mayer IA, Vahdat LT, Tolaney SM, Isakoff SJ, et al. N Engl J Med. 2019;380:741-751.
Sugar-Sweetened Beverages May Increase Breast Cancer Mortality
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. It is estimated that more than 3.5 million breast cancer survivors are alive in the US. Understanding risk factors and modifying lifestyle behaviors can impact outcomes in this patient group.
Consumption of sugar-sweetened beverages has significantly increased over the past 3 decades worldwide, increasing the risk of obesity, hypertension and Type 2 diabetes, and in turn impacting cardiometabolic health. It is estimated that among all worldwide yearly deaths from diabetes and cardiovascular diseases, over 178,000 were attributable to sugar-sweetened beverages consumption.
More recently published large prospective French study (BMJ 2019;366:l2408) concluded that consumption of sugary drinks was positively associated with the risk of overall cancer and breast cancer. It has been hypothesized that sugar-sweetened beverages contain large quantities of sucrose and fructose which promote visceral adiposity, which can boost tumorigenesis through alterations in adipokine secretion and cell signaling pathways, independent of body weight. Further, long term consumption of sugary drinks result in high glycemic index or glycemic load, with chronically high blood glucose, and therefore chronically elevated insulin concentration. Insulin increases bioactive IGF-1, as well as proinflammatory markers, such as C reactive protein, and systemic inflammation is thought to promote cancer development by inhibiting apoptosis and stimulating cell proliferation, thereby increasing the risk of several cancers, including breast, liver, pancreas, endometrium, colorectal and bladder. Even though there is growing evidence of an association between sugar-sweetened beverages and increased risk of mortality in various populations, the effect of sugary drinks on mortality among breast cancer patients is unknown.
The researchers in this Western New York Exposures and Breast Cancer (WEB) Study assessed the relationship between sugar-sweetened soda and both all-cause and breast cancer mortality among 927 women between ages 35 to 79, who had been diagnosed with breast cancer. In this study, a food frequency questionnaire was used to assess frequency of sugar-sweetened soda consumption in the 12 to 24 months prior to diagnosis of breast cancer. Breast cancer cases were followed for a median of 18.7 years, with ascertainment of vital status via the National Death Index (NDI). This study focused on sugar-sweetened soda, as they are sugar loaded and add extra calories to the diet with no nutritional benefit.
Consumption of sugar-sweetened soda five times or more per week was noted to result in a 62% higher likelihood of dying from any cause and 85% more likely to die from breast cancer specifically, compared to women who never or rarely consumed sugar-sweetened soda. The risk of mortality was similarly increased among ER-positive, but not ER-negative patients, among women with BMI above the median, but not below the median. The risk of total mortality was higher in premenopausal women but not post-menopausal women.
It was concluded from this study that a higher frequency of sugar-sweetened soda consumption was associated with increased risk of total and breast cancer mortality among breast cancer patients, supporting existing guidelines on reducing consumption of sugar-sweetened beverages, including for women with a diagnosis of breast cancer. The authors added that this study provides evidence that diet may impact longevity of women after breast cancer.
Study Finds Regularly Drinking Sugar-Sweetened Soda May Increase Total and Breast Cancer Mortality. Koyratty N, McCann SE, Millen AE, et al. Cancer Epidemiol Biomarkers Prev March 2 2021 DOI: https://doi.org/10.1158/1055-9965.EPI-20-1242.
MARGENZA® Superior to Trastuzumab in Heavily Pretreated HER2-Positive Breast cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease, largely due to metastatic recurrence. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2 oncoprotein is also expressed by tumor cells in GastroEsophageal and other solid tumors.
HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (Ado-Trastuzumab Emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.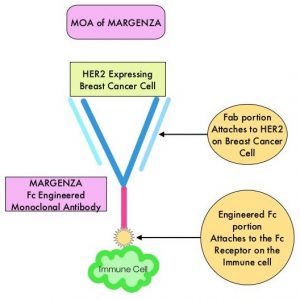
MARGENZA® (Margetuximab-cmkb) is an Fc-engineered, monoclonal antibody that binds to the HER2 oncoprotein with high specificity and affinity and inhibits tumor cell proliferation and survival, by mediating Antibody-Dependent Cellular Cytotoxicity (ADCC). It is postulated that the Fab portion of MARGENZA® has the same specificity and affinity to HER2 oncoprotein as Trastuzumab, with similar ability to disrupt signaling. However, the modified Fc region of MARGENZA®, which binds to Fc receptor expressing cells such as immune cells, has increased affinity for activating Fc receptor FCGR3A (CD16A) and decreased affinity for inhibitory Fc receptor FCGR2B (CD32B). These changes lead to greater ADCC and Natural Killer cell activation. Approximately 85% of individuals are CD16A-158F allele carriers.
The SOPHIA study is a randomized, multicenter, open-label, Phase III clinical trial, in which MARGENZA® plus chemotherapy was compared to Trastuzumab plus chemotherapy in patients with HER2-positive metastatic breast cancer, who have previously been treated with anti-HER2-targeted therapies. This study enrolled 536 patients who were randomized 1:1 to receive either MARGENZA® 15 mg/kg IV every three weeks (N=266) or Trastuzumab 6 mg/kg (8 mg/kg loading dose) IV every three weeks (N=270), in combination with Capecitabine, Eribulin, Gemcitabine or Vinorelbine, given at the standard doses. The median age was 56 years, all study patients had previously received Trastuzumab, all but one patient had previously received PERJETA® (Pertuzumab), and 91% of patients had previously received KADCYLA®. Patients were stratified by choice of chemotherapy, number of lines of therapy in the metastatic setting and number of metastatic sites. The dual Primary endpoints of the study were Progression Free Survival (PFS) by Blinded Independent Central Review (BICR) and Overall Survival (OS). Additional efficacy outcome measures included Objective Response Rate (ORR) and Duration of Response (DOR) assessed by BICR.
This study demonstrated a statistically significant 24% reduction in the risk of disease progression or death with MARGENZA® plus chemotherapy compared with Trastuzumab plus chemotherapy (HR= 0.76; P=0.03), with a median PFS of 5.8 months versus 4.9 months respectively. Treatment benefit was more pronounced in patients with CD16A genotypes containing a 158F allele (median PFS 6.9 versus 5.1 months, HR=0.68; P=0.005). The ORR for MARGENZA® plus chemotherapy was 22%, with a median Duration of Response of 6.1 months, compared to an ORR of 16% and median Duration of Response of 6.0 months for Trastuzumab plus chemotherapy. After the second planned interim analysis, the median OS was 21.6 months with MARGENZA® versus 19.8 months with Trastuzumab (HR= 0.89; P=0.33) and the ORR was 25% versus 14% respectively (P<0.001). The final Overall Survival (OS) analysis is expected in the second half of 2021. Safety was comparable in treatment groups, although the incidence of infusion-related reactions, mostly in cycle 1, was higher with MARGENZA® (13.3% versus 3.4%).
It was concluded that MARGENZA® in combination with chemotherapy significantly improved PFS, compared to Trastuzumab plus chemotherapy, in pretreated patients with HER2 positive metastatic breast cancer. MARGENZA® along with chemotherapy represents the newest treatment option for patients who have progressed on available HER2-directed therapies.
Efficacy of Margetuximab vs Trastuzumab in Patients With Pretreated ERBB2-Positive Advanced Breast Cancer: A Phase 3 Randomized Clinical Trial. Rugo HS, Im SA, Cardoso F, et al. for the SOPHIA Study Group. JAMA Oncol. Published online January 22, 2021. doi:10.1001/jamaoncol.2020.7932
Key Breast Cancer Risk Genes Identified from Two Large Studies
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease largely due to metastatic recurrence.
Genetic testing for cancer susceptibility with multigene testing panels is now becoming widely available and affordable. Identification of pathogenic variants in predisposition genes such as BRCA1 and BRCA2 among carriers has provided benefit through early intervention. However, the evidence of an association with cancer is often weak for many other genes on multigene testing panels, and estimates of the cancer risks associated with such variants are often not available. Further, estimates of the prevalence of pathogenic variants in predisposition genes in the general population are lacking.
Two large breast cancer case-control studies analyzed the associations between a number of commonly accepted cancer susceptibility genes and breast cancer risk.
The multinational study by Dorling et al. used a panel of 34 commonly accepted cancer susceptibility genes to perform sequencing on samples from 60,466 women with breast cancer and 53,461 controls (unaffected woman) from 25 countries participating in the Breast Cancer Association Consortium. The authors estimated odds ratios for breast cancer overall and tumor subtypes and evaluated missense-variant associations and classification of pathogenicity. The researchers found strong evidence of an association with breast cancer risk for Protein-Truncating Variants (genetic variants) caused by frameshift mutations in 9 genes, with a significant risk for breast cancer and P value of less than 0.0001 for 5 genes (ATM, BRCA1, BRCA2, CHEK2, and PALB2 - Odds Ratios ranging from 2.1 for ATM to 10.6 for BRCA1), and a P value of less than 0.05 for the other 4 genes (BARD1, RAD51C, RAD51D, and TP53 - Odds Ratio ranging from 1.8 for RAD51D to 3.06 for TP53). Further, it was noted that for the genetic variants in most of these genes, the Odds Ratio differed according to breast cancer subtype. Protein-Truncating Variants in ATM and CHEK2 were more strongly associated with ER-positive disease than with ER-negative disease, whereas genetic variants in BARD1, BRCA1, BRCA2, PALB2, RAD51C, and RAD51D were more strongly associated with ER-negative disease than with ER-positive disease. It was also found that rare missense variants in CHEK2 overall, as well as variants in specific domains in ATM, were associated with moderate breast cancer risk. The researchers also noted that none of the other 25 genes in the panel were informative for the prediction of breast cancer risk. This study places Protein-Truncating Variants in BRCA1, BRCA2, and PALB2 in the high-risk category and Protein-Truncating Variants in ATM, BARD1, CHEK2, RAD51C, and RAD51D in the moderate-risk category.
The US study by Hu et al. used a panel of 28 cancer predisposition genes to perform sequencing on samples from 32,247 women with breast cancer and 32,544 controls (unaffected women) from population-based studies in the Cancer Risk Estimates Related to Susceptibility (CARRIERS) consortium. The researchers assessed the associations between pathogenic variants in each gene and the risk of breast cancer.
The researchers noted that pathogenic variants in 12 established breast cancer predisposition genes were detected in 5% of breast cancer patients and in 1.63% of controls. Pathogenic variants in BRCA1 and BRCA2 were associated with a high risk of breast cancer, with Odds Ratios of 7.62 and 5.23 respectively and pathogenic variants in PALB2 were associated with a moderate risk (Odds Ratio 3.83). Pathogenic variants in BARD1, RAD51C, and RAD51D were associated with increased risks of ER-negative breast cancer and triple-negative breast cancer, whereas pathogenic variants in ATM, CDH1, and CHEK2 were associated with an increased risk of ER-positive breast cancer. Pathogenic variants in the other 16 candidate breast cancer predisposition genes were not associated with an increased risk of breast cancer.
Taken together, the results from these two large case-control studies suggested that variants in 8 genes - BRCA1, BRCA2, PALB2, BARD1, RAD51C, RAD51D, ATM, and CHEK2, had a significant association with breast cancer risk and majority of the other genes tested did not have a significant association with disease. Further, the distribution of mutations among women with breast cancer was different from the distribution among controls (unaffected women). Among breast cancer patients, the majority of mutations were in BRCA1, BRCA2, and PALB2, and among controls, the majority of mutations were in CHEK2 and ATM.
It can be concluded that, these two studies define the genes that are of utmost clinical value for inclusion on sequencing panels, for the prediction of breast cancer risk, and provides estimates of the prevalence of the pathogenic variants in the unaffected population. The authors added that these estimates can inform cancer testing and screening and improve clinical management strategies for women in the general population with inherited pathogenic variants in these genes.
Breast Cancer Risk Genes - Association Analysis in More than 113,000 Women. Breast Cancer Association Consortium; Dorling L, Carvalho S, Allen J, et al. N Engl J Med 2021;384:428-439.
A Population-Based Study of Genes Previously Implicated in Breast Cancer. Hu C, Hart SN, Gnanaolivu R, et al. N Engl J Med 2021;384:440-451.
Real-World Data: Surgery Improves Survival in Treatment Responsive Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women 12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease largely due to metastatic recurrence. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene and about 50% of HER2-positive breast cancers are Hormone Receptor positive. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. Not all HER2-positive, Hormone Receptor positive metastatic breast cancer patients, are candidates for chemotherapy. These patients however may benefit from anti-HER2 targeted therapy given along with endocrine therapy.
Approximately 6% of newly diagnosed breast cancer patients present with Stage IV disease. Systemic therapy has been the cornerstone of treatment for patients with metastatic breast cancer. Breast surgery is often not a consideration for patients with metastatic breast cancer. However, breast surgery can be offered for palliation of symptoms, taking into consideration the risks and benefits of such intervention, in a patient with an ulcerated, bleeding, or a fungating tumor mass, that cannot be controlled with systemic therapy.
Surgical resection of metastatic disease is not a new concept. Previously published results from randomized controlled trials among patients with metastatic breast cancer concluded that there was no survival advantage with surgical intervention. However these results have been questioned because of the small number of participants, and did not take into account either the Hormone Receptor, HER-2 status or the sequence of chemotherapy in relation to the surgical intervention. It therefore remains unclear whether surgery, in addition to systemic treatments and radiation therapy, improves survival for certain patients with metastatic breast cancer.
The authors in this real-world study identified 12,838 patients with HER-2 overexpressing and Hormone Receptor positive, Stage IV breast cancer, from the NCI database. They then studied patients who had either systemic therapy alone, systemic therapy and surgery, or had systemic therapy, surgery and radiation, and evaluated whether certain biologic subtypes and timing of chemotherapy were associated with survival advantages. Specifically, they evaluated whether the Hormone Receptor status had an influence on surgical benefit, in these treatment-responsive breast cancer patients, understanding that triple negative breast cancers are not very responsive to treatment. The researchers excluded patients who died within six months of their diagnoses, in order to ensure that only treatment-responsive cancers were being studied. The goal of this study was to understand if surgery made a difference in metastatic breast cancers that were responsive to treatment.
The researchers noted that patients with a surgical intervention tended to have a longer survival, compared to patients with other treatment plans. Patients whose cancers were HER2-positive saw prolonged survival, especially when their treatment plan included surgery. Further, in addition to the benefit of surgery among treatment-responsive metastatic breast cancer patients, the authors noted that systemic therapy before surgery (preoperative systemic therapy which included chemotherapy and targeted therapies) had the greatest survival advantage in patients with positive HER-2 and Estrogen and Progesterone Receptor status.
It was concluded from this study that patients with Stage IV breast cancer responsive to systemic therapy may be able to benefit from the addition of surgery, regardless of their biologic subtype. The authors added that clinicians should evaluate real-world evidence, including this study, when choosing the optimal treatment for their patients with metastatic breast cancer, as it may be difficult to conduct randomized clinical trials in this patient population.
ASO Author Reflections: Surgery Offers Survival Advantage in Treatment-Responsive Metastatic Breast Cancer. Stahl K, Dodge D, and Shen C. Annals of Surgical Oncology, 2020; DOI: 10.1245/s10434-020-09286-9
Adjuvant VERZENIO® with Endocrine Therapy in High Risk Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women 12%) will develop invasive breast cancer during their lifetime. Approximately 284,200 new cases of breast cancer will be diagnosed in 2021 and about 44,130 individuals will die of the disease largely due to metastatic recurrence. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors, and Hormone Receptor (HR)-positive/HER2-negative breast cancer is the most frequently diagnosed molecular subtype. Majority of these patients are diagnosed with early stage disease and are often cured with a combination of surgery, radiotherapy, chemotherapy, and hormone therapy. However approximately 20% of patients will experience local recurrence or distant relapse during the first 10 years of treatment. This may be more relevant for those with high risk disease, among whom the risk of recurrence is even greater during the first 2 years while on adjuvant endocrine therapy, due to primary endocrine resistance. More than 75% of the early recurrences are seen at distant sites.
Cyclin Dependent Kinases (CDKs) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. CDK 4 and 6 phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.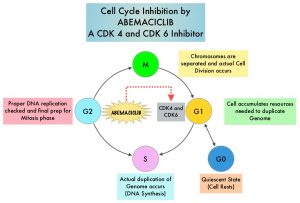
VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against cyclin D1/CDK 4 and cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
VERZENIO® is presently approved by the FDA as monotherapy as well as in combination with endocrine therapy for patients with HR-positive, HER2- negative advanced breast cancer. The addition of VERZENIO® to FASLODEX® resulted in a statistically significant improvement in Overall Survival among patients with HR-positive, HER2-negative advanced breast cancer, who had progressed on prior endocrine therapy. The goal of monarchE was to evaluate the additional benefit of adding a CDK4/6 inhibitor to endocrine therapy in the adjuvant setting, for patients with HR-positive, HER2-negative, high risk early breast cancer.
The International monarchE trial, is an open-label, randomized, Phase III study, which included 5637 patients, who were pre- and postmenopausal, with HR-positive, HER2-negative early breast cancer, and with clinical and/or pathologic risk factors that rendered them at high risk for relapse. The researchers defined high risk as the presence of four or more positive axillary lymph nodes, or 1-3 three positive axillary lymph nodes, with either a tumor size of 5 cm or more, histologic Grade 3, or centrally tested high proliferation rate (Ki-67 of 20% or more). Following completion of primary therapy which included both adjuvant and neoadjuvant chemotherapy and radiotherapy, patients were randomly assigned (1:1) to VERZENIO® 150 mg orally twice daily for 2 years plus 5 to 10 years of physicians choice of endocrine therapy as clinically indicated (N=2808), or endocrine therapy alone (N=2829). The median patient age was 51 years, about 43% of the patients were premenopausal, and 95% of patients had prior chemotherapy. Approximately 60% of patients had 4 or more positive lymph nodes. The Primary endpoint was Invasive Disease Free Survival (IDFS), and Secondary end points included distant Relapse Free Survival, Overall Survival, and safety. At a preplanned interim analysis, the addition of VERZENIO® to endocrine therapy resulted in a 25% reduction in the risk of developing a Invasive Disease Free Survival (IDFS) event, relative to endocrine therapy alone. Following the positive interim analysis, patients continued to be followed for IDFS, distant recurrence, and Overall Survival. The current study describes outcomes following an extended follow up of this trial, with a median follow up time of 19 months.
At the time of this primary outcome analysis, 1,437 patients (25.5%) had completed the two-year treatment period and 3,281 patients (58.2%) were in the two-year treatment period. The combination of VERZENIO® plus endocrine therapy continued to demonstrate superior Invasive Disease Free Survival (IDFS) compared to endocrine therapy alone, with a 28.7% reduction in the risk of developing invasive disease (P=0.0009; HR=0.713). The 2-year IDFS in the combination group was 92.3% and 89.3% in the endocrine therapy alone treatment group. This IDFS benefit with VERZENIO® was consistently noted in all prespecified subgroups. Further, there was an improvement in the 2-year distant Relapse Free Survival rate among patients who received the combination treatment compared with those who received endocrine therapy alone (93.8% versus 90.8%, respectively). Overall Survival data was immature at the time of analysis.
The researchers also evaluated outcomes among 2,498 patients with centrally assessed high tumor Ki-67 status. Among patients in this cohort, those who received the combination treatment had a 30.9% decreased risk of invasive disease compared with those who received endocrine therapy alone (P=0.01; HR=0.691) and the 2-year IDFS rates in the combination group and the endocrine therapy alone group were 91.6% and 87.1%, respectively. There were no new safety signals observed with VERZENIO®.
It was concluded that at the time of this primary outcome analysis, VERZENIO® combined with endocrine therapy continued to demonstrate a clinically meaningful improvement in Invasive Disease Free Survival, among patients with HR-positive, HER2-negative, node-positive, high risk, early breast cancer.
Primary outcome analysis of invasive disease-free survival for monarchE: abemaciclib combined with adjuvant endocrine therapy for high risk early breast cancer. O’Shaughnessy JA, Johnston S, Harbeck N, et al. Presented at the 2020 San Antonio Breast Cancer Symposium, December 8-11. Abstract. GS1-01.
Chemotherapy Can Be Spared in Majority of Postmenopausal Women with Node Positive Early Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer were diagnosed in 2020 and about 42,170 women died of the disease. Approximately 25% of patients with Hormone Receptor (HR)-positive, HER2-negative early breast cancer have metastatic lymph node involvement and two third of these patients are postmenopausal. Majority of these patients currently receive chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for early stage, HR-positive, HER-negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
In the ground-breaking TAILORx (Trial Assigning Individualized Options for Treatment) study which enrolled 10,273 patients with HR-positive, HER2-negative, axillary node-negative breast cancer, patients were divided into three groups based on their Recurrence Score. Patient with Intermediate Recurrence Score of 11-25 were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. There was no benefit noted from adding chemotherapy to endocrine therapy, for women older than 50 years in this Intermediate RS group, suggesting that a significant percentage of women with node-negative breast cancer do not achieve substantial benefit from chemotherapy. Whether the results of TAILORx can be extrapolated to women with node-positive breast cancer has remained unclear. It is estimated that approximately 85% of women with node-positive disease have Recurrence Score results of 0-25.
The RxPONDER trial was designed to determine the benefit of chemotherapy, in patients who had a Recurrence Score of 0-25. This trial did not include pre and postmenopausal women with Recurrence Score results 26-100 based on previously published studies suggesting that this patient group benefited from chemotherapy. SWOG S1007 (RxPONDER) is an multicenter, international, prospective, randomized, Phase III trial, in which patients with HR-positive, HER2-negative breast cancer with 1-3 positive axillary lymph nodes were included, to determine which patients would benefit from chemotherapy and which patients could safely avoid it. In this study, a total of 5083 HR-positive, HER2-negative breast cancer patients with 1-3 positive lymph nodes and Oncotype DX Recurrence Score of less than 25 were randomly assigned 1:1 to receive chemotherapy plus endocrine therapy or endocrine therapy alone. Approximately two-thirds of patients were postmenopausal and one-third were premenopausal and had no contraindications to taxane and/or anthracycline based chemotherapy. Patients were stratified by Recurrence Score (0-13 versus 14-25), menopausal status, and axillary nodal dissection versus sentinel node biopsy. The Primary endpoint was Invasive Disease Free Survival (IDFS), defined as local, regional, or distant recurrence, any second invasive cancer, or death from any cause, and whether the effect depended on the Recurrence Score. Secondary endpoints included Overall Survival (OS).
At a median follow up of 5.1 years, there was no association noted between Recurrence Score (RS) values and chemotherapy benefit for the entire study population (P=0.30). However, a prespecified analysis did show a significant association between chemotherapy benefit and menopausal status. Premenopausal women (N=1665) with an RS between 0 and 25 had an IDFS benefit with the addition of chemotherapy to endocrine therapy compared with endocrine therapy alone (94.2% versus 89%, HR=0.54; P=0.0004). This absolute 5.2% benefit in the premenopausal subset was highly significant. The relative risk reduction with the addition of chemotherapy to endocrine therapy for the two RS risk groups 0-13 and 14-25 was consistent in the premenopausal population, with an overall Hazard Ratio of 0.54. The absolute benefit was numerically higher in those with RS 14-25. Consistent benefit was again noted regardless of number of involved lymph nodes, although there was slight variation in the absolute benefit. Postmenopausal women (N=3350) did not benefit with the addition of chemotherapy to endocrine therapy when compared endocrine therapy alone, regardless of Recurrence Score (91.9% versus 91.6%, HR=0.97; P=0.82). Chemotherapy also improved Overall Survival in the premenopausal cohort, although the follow up is limited.
It was concluded from this practice-changing outcomes that postmenopausal women with HR-positive, HER2-negative breast cancer with 1-3 positive nodes and Oncotype DX Recurrence Score of 25 or less can safely avoid receiving adjuvant chemotherapy, whereas premenopausal patients with 1-3 positive nodes and a Recurrence Score of 25 or less should consider adjuvant chemotherapy. The authors added that these finding demonstrate that the great majority of postmenopausal women can be spared unnecessary chemotherapy and receive only endocrine therapy.
First results from a phase III randomized clinical trial of standard adjuvant endocrine therapy ± chemotherapy in patients with 1-3 positive nodes, hormone receptor-positive and HER2-negative breast cancer with recurrence scores ≤ 25: SWOG S1007 (RxPONDER). Kalinsky K, Barlow WE, Meric-Bernstam F, et al. 2020 San Antonio Breast Cancer Symposium. Presented December 10, 2020. Abstract GS3-00.
FDA Approves MARGENZA® for HER2 Positive Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2 oncoprotein is also expressed by tumor cells in gastroesophageal and other solid tumors.
HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (Ado-Trastuzumab Emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.
MARGENZA® (Margetuximab-cmkb) is an Fc-engineered, monoclonal antibody that binds to the HER2 oncoprotein with high specificity and affinity and inhibits tumor cell proliferation, and survival by mediating Antibody-Dependent Cellular Cytotoxicity (ADCC). It is postulated that the Fab portion of MARGENZA® has the same specificity and affinity to HER2 oncoprotein as Trastuzumab, with similar ability to disrupt signaling. However, the modified Fc region of MARGENZA® which binds Fc receptor expressing cells such as immune cells, has increased affinity for activating Fc receptor FCGR3A (CD16A) and decreases affinity for inhibitory Fc receptor FCGR2B (CD32B). These changes lead to greater ADCC and Natural Killer cell activation.
The SOPHIA study is a randomized, multicenter, open-label Phase III clinical trial, in which MARGENZA® plus chemotherapy was compared to Trastuzumab plus chemotherapy in patients with HER2-positive metastatic breast cancer, who have previously been treated with anti-HER2-targeted therapies. This study enrolled 536 patients who were randomized 1:1 to receive either MARGENZA® 15 mg/kg IV every three weeks (N=266) or Trastuzumab 6 mg/kg (or 8 mg/kg for loading dose) IV every three weeks (N=270), in combination with either Capecitabine, Eribulin, Gemcitabine or Vinorelbine, given at the standard doses. All study patients had previously received Trastuzumab, all but one patient had previously received PERJETA® (Pertuzumab), and 91% of patients had previously received KADCYLA®. Patients were stratified by choice of chemotherapy, number of lines of therapy in the metastatic setting and number of metastatic sites. The dual Primary endpoints of the study were Progression Free Survival (PFS) by Blinded Independent Central Review (BICR) and Overall Survival (OS). Additional efficacy outcome measures included Objective Response Rate (ORR) and Duration of Response (DOR) assessed by BICR.
This study demonstrated a statistically significant 24% reduction in the risk of disease progression or death with MARGENZA® plus chemotherapy compared with Trastuzumab plus chemotherapy (HR= 0.76; P=0.033), with a median PFS of 5.8 months versus 4.9 months respectively. Treatment benefit was more pronounced in patients with CD16A genotypes containing a 158F allele (median PFS 6.9 versus 5.1 months, HR=0.68; P=0.005). The ORR for MARGENZA® plus chemotherapy was 22%, with a median Duration of Response of 6.1 months, compared to an ORR of 16% and median Duration of Response of 6.0 months for Trastuzumab plus chemotherapy. The final Overall Survival (OS) analysis is expected in the second half of 2021. The most common adverse drug reactions occurring in more than 10% of patients receiving MARGENZA® plus chemotherapy included fatigue/asthenia, nausea, diarrhea, vomiting, headache, pyrexia, alopecia, abdominal pain, peripheral neuropathy, arthralgia/myalgia, cough, dyspnea, infusion-related reactions, palmar-plantar erythrodysesthesia, and extremity pain.
It was concluded that MARGENZA® in combination with chemotherapy significantly improved PFS, compared to Trastuzumab plus chemotherapy, in pretreated patients with HER2 positive metastatic breast cancer. MARGENZA® along with chemotherapy represents the newest treatment option for patients who have progressed on available HER2-directed therapies.
SOPHIA primary analysis: A phase 3 study of margetuximab + chemotherapy (C) versus trastuzumab + C in patients with HER2+ metastatic breast cancer after prior anti-HER2 therapies. Rugo HS, Im SA, Shaw Wright GL, et al. J Clin Oncol 37, 2019 (suppl; abstr 1000)
SABCS 2020: Ongoing Benefit with VERZENIO® in High Risk Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women 12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease largely due to metastatic recurrence. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors, and Hormone Receptor (HR)-positive/HER2-negative breast cancer is the most frequently diagnosed molecular subtype. Majority of these patients are diagnosed with early stage disease and are often cured with a combination of surgery, radiotherapy, chemotherapy, and hormone therapy. However approximately 20% of patients will experience local recurrence or distant relapse during the first 10 years of treatment. This may be more relevant for those with high risk disease, among whom the risk of recurrence is even greater during the first 2 years while on adjuvant endocrine therapy, due to primary endocrine resistance. More than 75% of the early recurrences are seen at distant sites.
Cyclin Dependent Kinases (CDKs) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. CDK 4 and 6 phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against cyclin D1/CDK 4 and cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
VERZENIO® is presently approved by the FDA as monotherapy as well as in combination with endocrine therapy for patients with HR-positive, HER2- negative advanced breast cancer. The addition of VERZENIO® to FASLODEX® resulted in a statistically significant improvement in Overall Survival among patients with HR-positive, HER2-negative advanced breast cancer, who had progressed on prior endocrine therapy. The goal of monarchE was to evaluate the additional benefit of adding a CDK4/6 inhibitor to endocrine therapy in the adjuvant setting, for patients with HR-positive, HER2-negative, high risk early breast cancer.
The International monarchE trial, is an open-label, randomized, Phase III study, which included 5637 patients, who were pre- and postmenopausal, with HR-positive, HER2-negative early breast cancer, and with clinical and/or pathologic risk factors that rendered them at high risk for relapse. The researchers defined high risk as the presence of four or more positive axillary lymph nodes, or 1-3 three positive axillary lymph nodes, with either a tumor size of 5 cm or more, histologic Grade 3, or centrally tested high proliferation rate (Ki-67 of 20% or more). Following completion of primary therapy which included both adjuvant and neoadjuvant chemotherapy and radiotherapy, patients were randomly assigned (1:1) to VERZENIO® 150 mg orally twice daily for 2 years plus 5 to 10 years of physicians choice of endocrine therapy as clinically indicated (N=2808), or endocrine therapy alone (N=2829). The median patient age was 51 years, about 43% of the patients were premenopausal, and 95% of patients had prior chemotherapy. Approximately 60% of patients had 4 or more positive lymph nodes. The Primary endpoint was Invasive Disease Free Survival (IDFS), and Secondary end points included distant Relapse Free Survival, Overall Survival, and safety. At a preplanned interim analysis, the addition of VERZENIO® to endocrine therapy resulted in a 25% reduction in the risk of developing a Invasive Disease Free Survival (IDFS) event, relative to endocrine therapy alone. Following the positive interim analysis, patients continued to be followed for IDFS, distant recurrence, and Overall Survival. The current study describes outcomes following an extended follow up of this trial, with a median follow up time of 19 months.
At the time of this primary outcome analysis, 1,437 patients (25.5%) had completed the two-year treatment period and 3,281 patients (58.2%) were in the two-year treatment period. The combination of VERZENIO® plus endocrine therapy continued to demonstrate superior Invasive Disease Free Survival (IDFS) compared to endocrine therapy alone, with a 28.7% reduction in the risk of developing invasive disease (P=0.0009; HR=0.713). The 2-year IDFS in the combination group was 92.3% and 89.3% in the endocrine therapy alone treatment group. This IDFS benefit with VERZENIO® was consistently noted in all prespecified subgroups. Further, there was an improvement in the 2-year distant Relapse Free Survival rate among patients who received the combination treatment compared with those who received endocrine therapy alone (93.8% versus 90.8%, respectively). Overall Survival data was immature at the time of analysis.
The researchers also evaluated outcomes among 2,498 patients with centrally assessed high tumor Ki-67 status. Among patients in this cohort, those who received the combination treatment had a 30.9% decreased risk of invasive disease compared with those who received endocrine therapy alone (P=0.01; HR=0.691) and the 2-year IDFS rates in the combination group and the endocrine therapy alone group were 91.6% and 87.1%, respectively. There were no new safety signals observed with VERZENIO®.
It was concluded that at the time of this primary outcome analysis, VERZENIO® combined with endocrine therapy continued to demonstrate a clinically meaningful improvement in Invasive Disease Free Survival, among patients with HR-positive, HER2-negative, node-positive, high risk, early breast cancer.
Primary outcome analysis of invasive disease-free survival for monarchE: abemaciclib combined with adjuvant endocrine therapy for high risk early breast cancer. O'Shaughnessy JA, Johnston S, Harbeck N, et al. Presented at the 2020 San Antonio Breast Cancer Symposium, December 8-11. Abstract. GS1-01.
Adjuvant Trastuzumab Monotherapy for Older Patients with HER-2 Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Trastuzumab is a humanized monoclonal antibody targeting HER2. Adjuvant and neoadjuvant chemotherapy given along with Trastuzumab reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of Trastuzumab, several other HER2-targeted therapies have become available. The duration of adjuvant Trastuzumab therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials.
Elderly patients with HER-2 positive breast cancer may not be candidates for adjuvant chemotherapy. Single agent Trastuzumab used as adjuvant treatment without chemotherapy could be of potential benefit, avoiding chemotherapy-induced toxicities. However, the benefit of single agent Trastuzumab has not been investigated in patients older than 70 years. The present study was designed to investigate the efficacy of Trastuzumab monotherapy, compared with Trastuzumab in combination with chemotherapy, incidence of Adverse Events, as well as Quality of Life, in terms of the noninferiority criterion.
RESPECT Study is a multicenter, open-label, randomized controlled, prospective, adjuvant, noninferiority trial, in which Trastuzumab monotherapy was compared with Trastuzumab plus chemotherapy, among patients older than 70 years, with HER-2 positive breast cancer. A total of 275 patients, aged 70-80 years with surgically treated HER-2 positive invasive breast cancer, were randomly assigned in a 1:1 ratio to receive either Trastuzumab monotherapy (N=137) or Trastuzumab plus chemotherapy (N=138). Trastuzumab plus chemotherapy treatment consisted of a loading dose of Trastuzumab at 8 mg/kg and a maintenance dose of 6 mg/kg every 3 weeks for 1 year. Chemotherapy regimens consisted of either Paclitaxel 80 mg/m2 IV weekly for 12 weeks, Docetaxel 75 mg/m2 IV every 3 weeks for 4 cycles, Doxorubicin 60 mg/m2 IV and Cyclophosphamide 600 mg/m2 IV (AC) every 3 weeks for 4 cycles, Epirubicin 90 mg/m2 IV and Cyclophosphamide 600 mg/m2 IV (EC) every 3 weeks for 4 cycles, Cyclophosphamide 75-100 mg orally, Methotrexate 40 mg/m2, and 5-fluorouracil 500-600 mg/m2 IV (CMF) for 6 cycles, Docetaxel 75 mg/m2 IV and Cyclophosphamide 600 mg/m2 IV (TC) every 3 weeks for 4 cycles or Docetaxel 60-75 mg/m2 IV, Carboplatin AUC 5-6 mg/ml/min IV along with Trastuzumab IV (TCH) every 3 weeks for 6 cycles. Patients treated with Trastuzumab monotherapy received similar doses of loading and maintenance Trastuzumab. Patients were stratified based on Performance Status, Hormone Receptor status and pathologic nodal status. Approximately 44% of patients had Stage I disease, 42% had Stage IIA, 13% had IIB, and 1% had IIIA disease. Approximately 14% of patients received Selective Estrogen Receptor Modulators such as Tamoxifen, and about 69% of patients received Aromatase Inhibitors. The Primary endpoint was Disease Free Survival (DFS) with assessment of prespecified Hazard Ratio (HR) and Restricted Mean Survival Time (RMST) for each treatment group. (RMST has been advocated as an alternative or a supplement to the Hazard Ratio for reporting the effect of an intervention in a randomized clinical trial, and is a measure of average survival from time 0 to a specified time point, and may be estimated as the area under the KM curve up to that point. RMST measure is especially informative for older patient populations in which Quality of Life issues are more important). Secondary endpoints included Overall Survival (OS), Relapse-Free Survival (RFS), Adverse Events (AEs) and Health-Related Quality of Life (HRQoL). The median follow up time was 4.1 years.
The 3-year DFS was 89.5% with Trastuzumab monotherapy versus 93.8% with Trastuzumab plus chemotherapy (HR=1.36; P=0.51) and this study failed to meet the prespecified criterion for noninferiority. However, a preplanned analysis of DFS according to RMST was -0.39 months, suggesting that only 0.39 months of DFS were lost within 3 years, by avoiding chemotherapy. The 3-year RFS was 92.4% with Trastuzumab monotherapy versus 95.3% with Trastuzumab plus chemotherapy (HR=1.33) and the difference in RMST for RFS between treatment groups at 3 years was −0.41 months (P=0.53). There were significant differences noted in clinically meaningful HRQoL deterioration rate at 2 months (31% for Trastuzumab monotherapy versus 48% for Trastuzumab plus chemotherapy; P=.016) and at 1 year (19% versus 38%; P=0.009). Breast cancer-specific survival at 3 years was 99.2% with Trastuzumab monotherapy versus 99.2% with Trastuzumab plus chemotherapy (HR=0.20; P=0.14).
The authors concluded that even though the Primary endpoint of noninferiority for Trastuzumab monotherapy was not met, the Restricted Mean Survival Time revealed that the observed loss of survival without chemotherapy was less than 1 month at 3 years, and Health-Related Quality of Life was better, with lower toxicities. Therefore, Trastuzumab monotherapy can be considered as a reasonable adjuvant therapy option for a select group of elderly patients with favorable outcomes.
Randomized Controlled Trial of Trastuzumab With or Without Chemotherapy for HER2-Positive Early Breast Cancer in Older Patients. Sawaki M, Taira N, Uemura Y, et al. J Clin Oncol. 2020;38:3743-3752.
FDA Approves KEYTRUDA® Plus Chemotherapy for Triple Negative Breast Cancer
SUMMARY: The FDA on November 13, 2020, granted accelerated approval to KEYTRUDA® (Pembrolizumab) in combination with chemotherapy for the treatment of patients with locally recurrent, unresectable or metastatic, Triple Negative Breast Cancer (TNBC), whose tumors express PD-L1 (Combined Positive Score-CPS 10 or more) as determined by an FDA approved test. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients and African American females. It is usually aggressive, and tumors tend to be high grade, and patients with TNBC are at a higher risk of both local and distant recurrence and often develop visceral metastases. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates, similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Overall survival among patients with pretreated metastatic TNBC has not changed over the past 2 decades and standard chemotherapy is associated with low response rates of 10-15% and a Progression Free Survival (PFS) of only 2-3 months.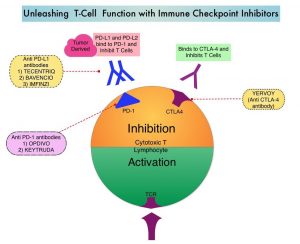
KEYTRUDA® is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response, and unleashes the tumor-specific effector T cells. The rationale for combining chemotherapy with immunotherapy is that cytotoxic chemotherapy releases tumor-specific antigens, and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity. Single agent KEYTRUDA® in metastatic TNBC demonstrated durable antitumor activity in several studies, with Objective Response Rates (ORRs) ranging from 10% to 21% and improved clinical responses in patients with higher PD-L1 expression. When given along with chemotherapy as a neoadjuvant treatment for patients with high-risk, early-stage TNBC, KEYTRUDA® combination achieved Pathological Complete Response rate of 65%, regardless of PD-L1 expression. Based on this data, KEYTRUDA® in combination with chemotherapy was studied, for first-line treatment of TNBC.
KEYNOTE-355 is a randomized, double-blind, Phase III study, which evaluated the benefit of KEYTRUDA® in combination with one of the three different chemotherapy regimens, nab-Paclitaxel, Paclitaxel, or the non-taxane containing Gemzar/Carboplatin, versus placebo plus one of the three chemotherapy regimens, in patients with previously untreated or locally recurrent inoperable metastatic TNBC. In this study, 847 patients were randomized 2:1 to receive either KEYTRUDA® 200 mg IV on day 1 of each 21-day cycle along with either nab-paclitaxel 100 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, Paclitaxel 90 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, or Gemcitabine 1000 mg/m2 IV plus Carboplatin AUC 2, IV on days 1 and 8 of each 21-day cycle (N= 566) or placebo along with one of the three chemotherapy regimens (N= 281). This study was not designed to compare the efficacy of the different chemotherapy regimens. Treatment was continued until disease progression. Patients were stratified by chemotherapy, PD-L1 tumor expression (CPS of 1 or higher versus CPS of less than 1), and prior treatment with the same class of neoadjuvant/adjuvant chemotherapy (Yes versus No). The baseline characteristics of treatment groups were well-balanced. The co-Primary end points of the trial were Progression Free Survival (PFS) and Overall Survival (OS) in patients with PD-L1-positive tumors, and in all patients. Secondary end points were Objective Response Rate (ORR), Duration of Response, Disease Control Rate, and Safety. The median follow up for patients assigned to receive KEYTRUDA® was 17.5 months and 15.5 months for the placebo group. The authors reported the results from an interim analysis conducted by an Independent Data Monitoring Committee (IDMC).
KEYTRUDA® in combination with chemotherapy, significantly improved PFS in patients with CPS (Combined Positive Score) of 10 or greater. The median PFS was 9.7 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for placebo plus chemotherapy (HR=0.65, P=0.0012). This represented a 35% reduction in the risk of disease progression. Among patients with CPS of 1 or greater, the median PFS was 7.6 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for the placebo plus chemotherapy arm (HR= 0.74; P=0.0014). This however based on prespecified statistical criteria, was not considered statistically significant. Among the entire Intention-To-Treat (ITT) population, the median PFS was 7.5 months in the KEYTRUDA® plus chemotherapy group, compared with 5.6 months for chemotherapy plus placebo group (HR=0.82). Formal statistical significance was not tested in the ITT population. Overall Survival data are pending. Adverse Events (AEs) were similar in both treatment groups, although immune-related AEs occurred at a higher incidence in the KEYTRUDA® arm.
It was concluded that KEYTRUDA® in combination with several chemotherapy regimens, showed a statistically significant and clinically meaningful improvement in PFS, compared with chemotherapy alone, in patients with previously untreated locally recurrent, inoperable or metastatic TNBC, whose tumors expressed PD-L1 with a Combined Positive Score (CPS) of 10 or more. This data may be particularly relevant for patients who may have received a taxane in the adjuvant setting within a year, and could be more appropriately treated with a non-taxane regimen, in combination with KEYTRUDA®.
KEYNOTE-355: Randomized, double-blind, phase III study of pembrolizumab + chemotherapy versus placebo + chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer. Cortes J, Cescon DW, Rugo HS. et al. J Clin Oncol 38: 2020 (suppl; abstr 1000)
NERLYNX® Combination Superior to TYKERB® Combination in Advanced HER2-Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes.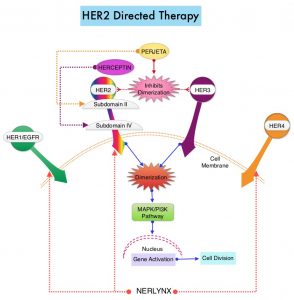
NERLYNX® (Neratinib) is a potent, irreversible, oral Tyrosine Kinase Inhibitor, of HER1, HER2 and HER4 (pan-HER inhibitor). NERLYNX® interacts with the catalytic domain of HER1, HER2, and HER4 and blocks their downstream signaling pathways, resulting in decreased cell proliferation and increased cell death. Clinical data has suggested that NERLYNX® has significant activity in suppressing HER-mediated tumor growth and is able to overcome tumor escape mechanisms experienced with current HER2-targeted and chemotherapeutic agents. It has been well known that hormone receptor positive breast cancer patients, who are also HER2-positive, have relative resistance to hormone therapy. Preclinical models had suggested that the addition of NERLYNX® could improve responses in ER positive, HER2-positive breast cancer patients. Further, NERLYNX® has clinical activity in patients with HER2-positive metastatic breast cancer. NERLYNX® is the first TKI approved by the FDA, shown to reduce the risk for disease recurrence, in patients with early stage HER2-positive breast cancer. NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly reduced the proportion of clinically relevant breast cancer relapses that might lead to death, such as distant and locoregional recurrences outside the preserved breast.
TYKERB® (Lapatinib) is a Tyrosine Kinase Inhibitor of HER2 and EGFR, and in a previously published Phase III study, a combination of TYKERB® plus XELODA® (Capecitabine) was found to be superior to XELODA® alone in women with HER2-positive advanced breast cancer, that has progressed after treatment with regimens that included an Anthracycline, a Taxane, and HERCEPTIN®. (N Engl J Med 2006; 355:2733-2743)
The NALA trial was designed to compare NERLYNX® plus XELODA® versus TYKERB® plus XELODA® in patients with heavily pretreated Stage IV HER2-positive metastatic breast cancer, including those with asymptomatic or stable (treated or untreated) CNS metastases. In this multinational, randomized, active-controlled, Phase III study, 621 patients (N = 621) with metastatic HER2-positive breast cancer who received two or more prior anti-HER2 based regimens in the metastatic setting were randomly assigned in a 1:1 to receive NERLYNX® 240 mg given orally once daily on days 1-21 along with XELODA® 750 mg/m2 given orally twice daily on days 1-14 for each 21-day cycle (N=307) or TYKERB® 1250 mg given orally once daily on days 1-21 along with XELODA® 1000 mg/m2 given orally twice daily on days 1-14 for each 21-day cycle (N=314). Approximately 85% of patients had visceral metastases, and about 30% had received at least three anti-HER2 therapies. Patients in the NERLYNX® group also received antidiarrheal prophylaxis with Loperamide. Patients were treated until disease progression or unacceptable toxicity. The Co-Primary endpoints were Progression Free Survival (PFS) and Overall Survival (OS). Secondary endpoints included Objective Response Rate (ORR) and Duration of Response, Clinical Benefit Rate (CBR), time to intervention for symptomatic metastatic Central Nervous System (CNS) disease and Safety.
At a median follow up of 29.9 months, treatment with NERLYNX® with XELODA® significantly improved the median PFS, compared to those receiving TYKERB® with XELODA® (HR=0.76; P=0.006). This represented a 24% reduction in the risk of disease progression or death for those receiving a combination of NERLYNX® and XELODA®. The PFS rate at 12 months was 29% versus 15% respectively. The median OS was 21 months for patients receiving NERLYNX® and XELODA® compared to 18.7 months for those receiving TYKERB® and XELODA® (HR=0.88; P=0.20) and this was not statistically significant. The ORR was numerically higher with NERLYNX® and XELODA® combination in patients with measurable disease (32.8% versus 26.7%), and there was a statistically significant improvement in the Clinical Benefit Rate (45% versus 36%; P=0.03). The median Duration of Response was 8.5 versus 5.6 months respectively (HR=0.50; P=0.0004), favoring the NERLYNX® combination. The time to intervention for symptomatic CNS disease was significantly delayed with NERLYNX® combination versus TYKERB® combination, with an overall cumulative incidence of 22.8% versus 29.2% respectively (P= 0.043). The most common toxicities of any grade in the study population were diarrhea, nausea, palmar-plantar erythrodysesthesia syndrome, and vomiting. Treatment related toxicities were similar between arms, but there was a higher rate of Grade 3 diarrhea with the NERLYNX® combination (24% versus 13% respectively).
It was concluded from this study that a combination of NERLYNX® and XELODA® significantly improved Progression Free Survival, with a trend towards improved Overall Survival, and also resulted in a delayed time to intervention for symptomatic CNS disease, among patients with heavily pretreated advanced HER2-positive breast cancer. This is the first study to demonstrate superiority of one HER2-directed Tyrosine Kinase Inhibitor over another, in HER2-positive metastatic breast cancer.
Neratinib Plus Capecitabine Versus Lapatinib Plus Capecitabine in HER2-Positive Metastatic Breast Cancer Previously Treated With 2 or More HER2-Directed Regimens: Phase III NALA Trial. Saura C, Oliveira M, Y Feng Y-H, et al. for the NALA Investigators. J Clin Oncol. 2020;38:3138-3149.
DCIS and Risk of Death from Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease largely due to metastatic recurrence. Carcinoma in situ of the breast also known as Ductal Carcinoma In Situ (DCIS) is defined as a malignant proliferation of ductal epithelial cells that are confined to the milk ducts without invasion of the basement membrane, and is a precursor lesion to invasive carcinoma. DCIS accounts for approximately 25% of all newly diagnosed breast cancers. Patients with small, screening-detected lesions, are often treated with breast-conserving surgery (to prevent the development of invasive breast cancer), followed by adjuvant radiation and hormonal therapy, although neither of the latter two interventions have been shown to improve survival outcomes. As such, a significant number of patients are over treated. DCIS in itself is not life-threatening but can potentially progress to invasive breast cancer. The two important goals of DCIS treatment therefore are, to prevent invasive ipsilateral cancer recurrence and to prevent death from breast cancer. There remains a large unmet need, to distinguish relatively benign DCIS from DCIS that will develop into invasive breast cancer.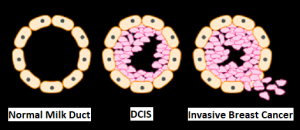
In a previously published meta-analysis (Cancer Epidemiol Biomarkers Prev. 2019;28:835-845), researchers identified six prognostic factors that were statistically significant and were associated with a 36% to 84% increase in the relative risk of recurrence of invasive disease after a DCIS diagnosis. These six factors included-
1) African American race (43% higher risk)
2) Premenopausal status (59% higher risk)bre
3) Detection by palpation (84% higher risk)
4) Positive margins (63% higher risk)
5) High histologic grade (36% higher risk)
6) High p16 expression (51% higher risk).
This present large cohort study was conducted to determine the risk of death from breast cancer, following diagnosis and treatment of DCIS, compared with the mortality risk among cancer-free women, in the general population. This study included a total of 144,524 women diagnosed with first primary DCIS between 1995 and 2014, from the Surveillance, Epidemiology and End Results (SEER) registries database. Patients with DCIS with microinvasion, Lobular Carcinoma In Situ (LCIS), nonepithelial histological presentations, Paget disease of the nipple, diffuse DCIS, unknown laterality, no surgical intervention on the primary tumor, DCIS diagnosis in women younger than 25 years or aged 80 years or older, were all excluded. Patients with DCIS underwent surgical treatment, and approximately half of these patients also received radiotherapy. These patients were followed from the date of DCIS diagnosis until death from breast cancer, or date of last follow up. These patients were compared with women in the general population without a diagnosis of breast cancer (control group). The mean age at diagnosis was 57.4 years. The Primary outcome was death from breast cancer. Standardized Mortality Ratios (SMR) were estimated by comparing deaths from breast cancer among women diagnosed with DCIS, with expected deaths from breast cancer among women in the general population who did not have cancer.
At a mean follow up period of 9.2 years, the incidence of ipsilateral invasive recurrence events was 3.1%, resulting in a 20-year actuarial risk of 13.9%. There was a 3.8% incidence of contralateral invasive breast cancer events during this follow up period, resulting in a 20-year actuarial risk of 11.3%. The 20-year actuarial risk of breast cancer death among women with DCIS was 3.3%.
The Standardized Mortality Ratio (SMR) for death from breast cancer given a diagnosis of DCIS was 3.36, but varied based on age and race. The SMR for women younger than 40 years was much higher at 11.95, whereas the SMR for women aged 40 to 49 years was 4.15. The SMR for White women was 3.03, for Black women was 7.56, and for East Asian women was 1.89. The SMR for Black women diagnosed with DCIS before age 50 years was 12.10, and the SMR for White women diagnosed with DCIS before age 50 years was 4.21, suggesting that Black women did worse than White woman.
All women with DCIS underwent surgical treatment, and 47.1% also received radiotherapy. Among those patients who were not treated with radiotherapy, the SMR was 4.12, for those treated with unilateral mastectomy and 4.14 for those treated with bilateral mastectomies. Among women who underwent lumpectomy, the SMR was 2.81 for women treated with radiotherapy and 3.42 for those who underwent surgical treatment alone. There were 1540 women who died of breast cancer in the cohort, of whom 45.7% experienced an ipsilateral invasive recurrence or contralateral invasive breast cancer in the interval between DCIS and death from breast cancer. Among the patients who died, 27.8% were known to have undergone a mastectomy.
The annual mortality rate from breast cancer over the entire period of follow up was, 0.12% per year. The mortality rate increased for the first 10 years of the follow-up period and remained constant through years 15 thru 20. The cumulative 20-year risk of breast cancer-specific mortality following DCIS was 3.3% overall, but for Black women diagnosed before age 50 years, the 20-year risk of breast cancer-specific mortality was 8.1%. It has been postulated that the highest risk for recurrence among women who underwent mastectomy may be related to them having more extensive disease with close margins or may have genetic mutations that increase the likelihood of recurrence. Further, patients with DCIS undergoing bilateral mastectomies generally are not treated with endocrine therapy.
It was concluded from this cohort study that women with DCIS had a 3-fold increased risk of death from breast cancer after surgical treatment. The Standardized Mortality Ratio was lower among women who received lumpectomy plus radiation compared with women who received lumpectomy alone. The rate of breast cancer death was nearly 12-fold higher among women diagnosed with DCIS before age 40 years and 7-fold higher in Black women diagnosed with DCIS, compared with the general population.
Association of a Diagnosis of Ductal Carcinoma In Situ With Death From Breast Cancer. Giannakeas V, Sopik V and Narod SA. JAMA Netw Open. 2020;3(9):e2017124. doi:10.1001/jamanetworkopen.2020.17124
Late Breaking Abstract - ESMO 2020. VERZENIO® Plus Endocrine Therapy Improves Disease Free Survival in Early Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women 12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease largely due to metastatic recurrence. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors, and Hormone Receptor (HR)-positive/HER2-negative breast cancer is the most frequently diagnosed molecular subtype. Majority of these patients are diagnosed with early stage disease and are often cured with a combination of surgery, radiotherapy, chemotherapy, and hormone therapy. However approximately 20% of patients will experience local recurrence or distant relapse during the first 10 years of treatment. This may be more relevant for those with high risk disease, among whom the risk of recurrence is even greater during the first 2 years while on adjuvant endocrine therapy, due to primary endocrine resistance. More than 75% of the early recurrences are seen at distant sites.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. CDK 4 and 6 phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against cyclin D1/CDK 4 and cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
VERZENIO® is presently approved by the FDA as monotherapy as well as in combination with endocrine therapy for patients with HR-positive, HER2- negative advanced breast cancer. The addition of VERZENIO® to FASLODEX® resulted in a statistically significant improvement in Overall Survival among patients with HR-positive, HER2-negative advanced breast cancer, who had progressed on prior endocrine therapy. The goal of monarchE was to evaluate the additional benefit of adding a CDK4/6 inhibitor to endocrine therapy in the adjuvant setting, for patients with HR-positive, HER2-negative early breast cancer.
The international monarchE trial, is an open-label, randomized, Phase III study, which included 5637 patients, who were pre- and postmenopausal, with HR-positive, HER2-negative early breast cancer, and with clinical and/or pathologic risk factors that rendered them at high risk for relapse. The researchers defined high risk as the presence of four or more positive axillary lymph nodes, or 1-3 three positive axillary lymph nodes, with either a tumor size of 5 cm or more, histologic Grade 3, or centrally tested high proliferation rate (Ki-67 of 20% or more). Following completion of primary therapy which included both adjuvant and neoadjuvant chemotherapy and radiotherapy, patients were randomly assigned (1:1) to VERZENIO® 150 mg orally twice daily for 2 years plus 5 to 10 years of physicians choice of endocrine therapy as clinically indicated (N=2808), or endocrine therapy alone (N=2829). The median patient age was 51 years, about 43% of the patients were premenopausal, and 95% of patients had prior chemotherapy. Approximately 60% of patients had 4 or more positive lymph nodes. The Primary endpoint was Invasive Disease Free Survival (IDFS), and Secondary end points included distant Relapse Free Survival, Overall Survival, and safety. The authors in this publication reported the first results, following a preplanned interim analysis.
The addition of VERZENIO® to endocrine therapy resulted in an IDFS of 92.2% at 2 years compared with 88.7% with endocrine therapy alone, and this was statistically significant (HR=0.75; P=0.01). This suggested a 25% reduction in the risk of developing an IDFS event, relative to endocrine therapy alone, and a 3.5% absolute improvement in 2-year IDFS rates. VERZENIO® plus endocrine therapy combination also reduced the risk of metastatic recurrence especially in bone and liver (distant recurrences or Distant Relapse Free Survival) by a clinically meaningful 28% compared to endocrine therapy alone. This clinical benefit was observed in all prespecified subgroups, and among the 43% of patients who were premenopausal at diagnosis, there was a significant 37% reduction in the risk of recurrence compared to endocrine therapy alone. The safety was consistent with the known profile of VERZENIO® and included diarrhea, neutropenia, and fatigue. Diarrhea was well managed with antidiarrheal medications and dose adjustments.
It was concluded that VERZENIO® when combined with endocrine therapy demonstrated a significant improvement in Invasive Disease Free Survival, compared to endocrine therapy alone, in patients with high risk HR-positive, HER2-negative early breast cancer. The researchers also plan to look at genomic signatures in the tissue and plasma samples of enrolled patients and response to VERZENIO®.
Abemaciclib Combined With Endocrine Therapy for the Adjuvant Treatment of HR+, HER2−, Node-Positive, High-Risk, Early Breast Cancer (monarchE). Johnston SRD, Harbeck N, Hegg R, et al. DOI: 10.1200/JCO.20.02514 Journal of Clinical Oncology - published online before print September 20, 2020
Novel Ultrasensitive Liquid Biopsy Detects Minimal Residual Disease in Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women 12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease largely due to metastatic recurrence. Recurrent disease can occur early, but majority of patients with hormone receptor-positive breast cancer may develop recurrent disease decades, following their initial diagnosis. Once a diagnosis of metastatic breast cancer is established, it is generally incurable.
Systemic recurrence likely arises from micrometastatic disease present at initial diagnosis, which is undetectable by imaging or conventional blood tests. Adjuvant systemic therapy is recommended to eradicate micrometastatic disease and reduce the risk of cancer recurrence. However, current clinical tools are not accurate in identifying which patients would benefit from adjuvant systemic therapy and further are unable to, in real-time, predict whether the recommended therapies have achieved their therapeutic objective. Therefore, more sensitive techniques to detect micrometastatic disease are needed, so that patients receive the most appropriate and optimal therapy, with improved outcomes.
Recently published studies have shown that detection of circulating tumor DNA (ctDNA) in the peripheral blood may identify patients at risk of relapse following definitive therapy using digital droplet Polymerase Chain Reaction (ddPCR) assays. ctDNA refers to DNA fragments that are shed into the bloodstream by cancer cells after apoptosis or necrosis. The clinical sensitivity of this technique however is limited at the early postoperative time points, at which treatment decisions are usually made, and the lead time prior to clinical manifestation of overt metastatic disease has been relatively short. This is because presently available techniques track one or few mutations and are unable to detect MRD when the fraction of cancerous cell free DNA (cfDNA) in the bloodstream is low.
The authors developed an ultrasensitive blood test for tracking hundreds of patient-specific mutations, to detect Minimal Residual Disease (MRD), with a 1,000-fold lower error rate than conventional sequencing, to identify patients who might benefit from additional systemic treatment or de-escalation of therapy. The authors performed Whole-Exome Sequencing (WES) to define several hundred mutations from each patient's tumor, and to limit potential errors, selected somatic SNVs (Single Nucleotide Variants) to track, using duplex sequencing in cfDNA and employing strict criteria. The detection of 2 or more mutations in a cfDNA sample was considered MRD-positive and any mutations found in a patient's own genomic DNA was excluded.
For this study, the authors identified 142 patients who had been treated for Stage 0-III breast cancer with curative intent surgery, had postoperative blood and plasma samples available. Overall, 92% of patients received either neoadjuvant, or adjuvant chemotherapy, 76% received adjuvant endocrine therapy and 73% received adjuvant radiation treatment. Approximately 2% of patients had Stage 0 disease, 23% had Stage I, 48% had Stage II, and 27% had Stage III breast cancer at diagnosis. The MRD levels were tracked post-op (median 3.5 months) and 1 year out (median 14.2 months). The patients were monitored for distant recurrences for up to 13 years. A median of 57 mutations were targeted in each patient, identified via Whole-Exome Sequencing of primary tumor tissue and genomic DNA from whole blood. About 78% of patients had post-op samples available, while 86% had 1-year samples. The Primary objective of this study was to determine the predictive power of MRD testing and associated lead time to recurrence, in patients treated for early-stage breast cancer.
The median lead time (the time from a positive test to diagnosis of metastatic disease) between the first MRD-positive result and disease recurrence was 18.9 months in the patients with the most mutations tracked. This is significantly longer than what has been seen in prior studies. Distant disease recurrence was shown to be more likely if MRD was detected at the 1-year mark (HR=20.8; P<0.0001) compared with the post-op setting. Among these patients, the positive and negative predictive values for distant recurrence, was 0.70 and 0.77, respectively. Overall, the clinical sensitivities were 81% in patients with newly diagnosed metastatic breast cancer, 23% in the post-op setting, and 19% at the one year in early stage disease, and highest among patients with the most tumor mutations available to track. The authors noted that their testing methodology was 100-fold more sensitive than ddPCR, when tracking 488 mutations.
It was concluded that the ultrasensitive blood test developed by investigators for Minimal Residual Disease (MRD) could identify survivors who might benefit from additional systemic treatment versus de-escalation. MRD detection was strongly associated with distant recurrence and provided significant lead time to recurrence, enabling early therapeutic intervention in patients who may otherwise develop metastatic recurrence. The authors recommended that future blood-based Whole-Genome Sequencing assays should aim for extra sensitivity, to identify enough mutations to track in all patients.
Sensitive Detection of Minimal Residual Disease in Patients Treated for Early-Stage Breast Cancer. Parsons HA, Rhoades J, Reed SC, et al. Clin Can Res. DOI: 10.1158/1078-0432.CCR-19-3005. Published June 2020.
Favorable Outcomes with KADCYLA® in HER2+ Breast Cancer Irrespective of Mutational Status
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of HERCEPTIN®, several other HER2-targeted therapies have become available. The duration of adjuvant HERCEPTIN® therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials.
KADCYLA® is an Antibody-Drug Conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release. It is well established that patients with HER2-positive early breast cancer following HERCEPTIN® based neoadjuvant therapies have a pathological Complete Response (pCR) rate of 40-60%. Those without a pCR tend to have significantly less favorable outcomes. These patients irrespective of pathological response status complete their standard adjuvant therapy which includes 12 months of HER2-targeted therapy. KATHERINE trial was conducted to address an unmet need, and evaluate the benefit of switching from standard HER2-directed therapy to single-agent KADCYLA®, after neoadjuvant chemotherapy, in patients with residual invasive cancer at surgery.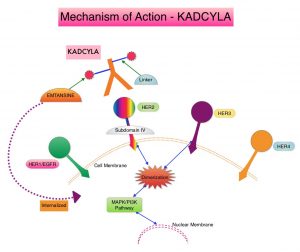
The KATHERINE trial is an open-label, Phase III global study, which compared KADCYLA® with HERCEPTIN®, as an adjuvant treatment for patients with HER2-positive early breast cancer, who had residual invasive disease following neoadjuvant chemotherapy and HERCEPTIN®. This study included 1,486 patients with HER2-positive early stage breast cancer, who were found to have residual invasive disease in the breast or axillary lymph nodes at surgery, following at least six cycles (16 weeks) of neoadjuvant chemotherapy with a Taxane (with or without Anthracycline) and HERCEPTIN®. Within 12 weeks of surgery, patients (N=1486) were randomly assigned in a 1:1 ratio to KADCYLA® 3.6 mg/kg IV every 3 weeks or HERCEPTIN® 6 mg/kg IV every 3 weeks, for 14 cycles (743 patients in each group). Both treatment groups were well balanced and Hormone Receptor positive disease was present in 72% of the patients. The Primary end point was invasive Disease Free Survival (iDFS-defined as freedom from ipsilateral invasive breast tumor recurrence, ipsilateral locoregional invasive breast cancer recurrence, contralateral invasive breast cancer, distant recurrence, or death from any cause). At the prespecified interim analysis, invasive disease occurred in 12.2% of patients who received KADCYLA® and 22.2% of patients who received HERCEPTIN®. The estimated percentage of patients who were free of invasive disease at 3 years was 88.3% in the KADCYLA® group and 77.0% in the HERCEPTIN® group, which translated to an absolute improvement of 11.3%. Invasive Disease Free Survival (iDFS), which was the Primary end point of the study, was significantly higher in the KADCYLA® group than in the HERCEPTIN® group (HR=0.50; P<0.001).This suggested that KADCYLA® reduced the risk of developing an invasive breast cancer recurrence or death by 50%.
The authors in this publication reported the exploratory analyses of the relationship between iDFS, and biomarkers potentially related to response. The authors focused on pathways that have been implicated in resistance to HER2 treatment such as pathways associated with PIK3CA mutations, as well as HER2 and PD-L1 expression in the post-neoadjuvant residual surgical samples.
In the first part of this biomarker analysis, a total of 1,363 available post-neoadjuvant surgery samples were analyzed through DNA sequencing for PIK3CA mutations. In the second part of this analysis, mRNA expression through RNA sequencing was determined on 1,059 tissue samples of which 244 were pre-neoadjuvant samples and 815 were post-neoadjuvant surgical samples. Because the post-neoadjuvant surgical samples were representative of the entire Intent-To-Treat (ITT) patient population, biomarker analysis for markers such as HER2, PD-L1, CD8, and predefined immune signatures including 3-gene, 5-gene, Teffector, chemokine signaling, and checkpoint inhibitor signatures, were performed by using post-neoadjuvant surgical samples.
The authors noted that in the ITT population (N=743), PIK3CA mutation status had no impact on outcomes when treated with KADCYLA®. Among those patients with mutated tumors who received KADCYLA® and HERCEPTIN®, the iDFS rates were 88.9% versus 77.9%, respectively (HR=0.54) and among those with non-mutated tumors the Invasive Disease Free Survival rates were 88.3% versus 77.0%, respectively (HR=0.48). There was no prognostic impact of PIK3CA mutations in this cohort of patients and the 3 year iDFS rates were almost identical between the mutated and non-mutated tumors.
The authors next looked at HER2 gene expression in the post-neoadjuvant surgical samples and noted that patients who had a tumors with high HER2 expression in the post-neoadjuvant residual surgical samples, and received subsequent treatment with HERCEPTIN®, had the worst outcomes with worse iDFS. This detrimental effect was not seen in the KADCYLA® group, suggesting that residual tumors that have a high HER2 expression in this setting are resistant to HERCEPTIN® but not to KADCYLA®.
When patients were evaluated based on their tumor PD-L1 expression, low PD-L1 expression in post-neoadjuvant residual tumors was associated with a worse outcome for those who received treatment with HERCEPTIN®, whereas treatment with KADCYLA® did not impact outcomes. These findings suggested that PD-L1 may be involved in some resistance mechanisms.
It was concluded that in the KATHERINE trial biomarker analysis, PIK3CA mutation status did not influence outcomes in either treatment groups. However, in the post-neoadjuvant HERCEPTIN® group, high HER2 expression and low PD-L1 expression was associated with less favorable outcomes. The benefit with KADCYLA® in this patient population was independent of all biomarkers assessed.
Biomarker data from KATHERINE: A phase III study of adjuvant trastuzumab emtansine (T-DM1) versus trastuzumab (H) in patients with residual invasive disease after neoadjuvant therapy for HER2-positive breast cancer. Denkert C, Lambertini C, Fasching PA, et al. J Clin Oncol. 2020;38(suppl 15):502. doi: 10.1200/JCO.2020.38.15_suppl.502
First Line KEYTRUDA® plus Chemotherapy Significantly Improves PFS in PD-L1-High Triple Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients and African American females. It is usually aggressive, and tumors tend to be high grade, and patients with TNBC are at a higher risk of both local and distant recurrence and often develop visceral metastases. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Overall survival among patients with pretreated metastatic TNBC has not changed over the past 2 decades and standard chemotherapy is associated with low response rates of 10-15% and a Progression Free Survival (PFS) of only 2-3 months.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response and unleashes the tumor-specific effector T cells. The rationale for combining chemotherapy with immunotherapy is that cytotoxic chemotherapy releases tumor-specific antigens, and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity.
Single agent KEYTRUDA® in metastatic TNBC demonstrated durable antitumor activity in several studies, with Objective Response Rates (ORRs) ranging from 10% to 21% and improved clinical responses in patients with higher PD-L1 expression. When given along with chemotherapy as a neoadjuvant treatment for patients with high-risk, early-stage TNBC, KEYTRUDA® combination achieved Pathological Complete Response rate of 65%, regardless of PD-L1 expression. Based on this data, KEYTRUDA® in combination with chemotherapy was studied, for first-line treatment of triple-negative metastatic breast cancer.
KEYNOTE-355 is a randomized, double-blind, phase III study, which evaluated the benefit of KEYTRUDA® in combination with one of the three different chemotherapy regimens, nab-Paclitaxel, Paclitaxel, or the non-taxane containing Gemzar/Carboplatin, versus placebo plus one of the three chemotherapy regimens, in patients with previously untreated or locally recurrent inoperable metastatic TNBC. In this study, 847 patients were randomized 2:1 to receive either KEYTRUDA® 200 mg IV on day 1 of each 21-day cycle along with either nab-paclitaxel 100 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, Paclitaxel 90 mg/m2 IV on days 1, 8 and 15 of each 28-day cycle, or Gemcitabine 1000 mg/m2 IV plus Carboplatin AUC 2 IV on days 1 and 8 of each 21-day cycle (N= 566) or placebo along with one of the three chemotherapy regimens (N= 281). This study was not designed to compare the efficacy of the different chemotherapy regimens. Treatment was continued until disease progression. Patients were stratified by chemotherapy, PD-L1 tumor expression (CPS of 1 or higher versus CPS of less than 1), and prior treatment with the same class of neoadjuvant/adjuvant chemotherapy (yes vs no). The baseline characteristics of treatment groups were well-balanced. The co-Primary end points of the trial were Progression Free Survival (PFS) and Overall Survival (OS) in patients with PD-L1-positive tumors, and in all patients. Secondary end points were Objective Response Rate (ORR), Duration of Response, Disease Control Rate, and safety. The median follow up for patients assigned to receive KEYTRUDA® was 17.5 months and 15.5 months for the placebo group. The authors reported the results from an interim analysis conducted by an Independent Data Monitoring Committee (IDMC).
KEYTRUDA® in combination with chemotherapy, significantly improved PFS in patients with CPS (Combined Positive Score) of 10 or greater. The median PFS was 9.7 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for placebo plus chemotherapy (HR=0.65, P=0.0012). This represented a 35% reduction in the risk of disease progression. Among patients with CPS of 1 or greater, the median PFS was 7.6 months for KEYTRUDA® plus chemotherapy, compared with 5.6 months for the placebo plus chemotherapy arm (HR= 0.74; P=0.0014). This however based on prespecified statistical criteria, was not considered statistically significant. Among the entire Intention-To-Treat (ITT) population, the median PFS was 7.5 months in the KEYTRUDA® plus chemotherapy group, compared with 5.6 months for chemotherapy plus placebo group (HR=0.82). Formal statistical significance was not tested in the ITT population. Overall Survival data are pending. Adverse Events (AEs) were similar in both treatment groups, although immune-related AEs occurred at a higher incidence in the KEYTRUDA® arm.
It was concluded that KEYTRUDA® in combination with several chemotherapy regimens, showed a statistically significant and clinically meaningful improvement in PFS, compared with chemotherapy alone, in patients with previously untreated locally recurrent, inoperable or metastatic TNBC, whose tumors expressed PD-L1 with a Combined Positive Score (CPS) of 10 or more. This data may be particularly relevant for patients who may have received a taxane in the adjuvant setting within a year, and could be more appropriately treated with a non-taxane regimen, in combination with KEYTRUDA®.
KEYNOTE-355: Randomized, double-blind, phase III study of pembrolizumab + chemotherapy versus placebo + chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer. Cortes J, Cescon DW, Rugo HS. et al. J Clin Oncol 38: 2020 (suppl; abstr 1000)
PIQRAY® Effective after Progression on CDK Inhibition in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Approximately 6% of newly diagnosed breast cancer patients present with Stage IV disease and about half of patients with primary breast cancer will progress later to the metastatic stage. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and Hormone Receptor (HR)-positive/HER2-negative breast cancer is the most frequently diagnosed molecular subtype. Most of these patients with advanced disease in the current era are treated with a combination of CDK4/6 inhibitor and endocrine therapy (often an oral Aromatase Inhibitor), based on survival data. However, resistance to these regimens typically develops in a majority of the patients.
The PhosphoInositide 3-Kinase (PI3K) pathway is an intracellular signaling pathway important in the regulation of cancer cell proliferation and metastasis. PI3K is a lipid kinase and has four distinct isoforms – alpha, beta, gamma and delta, which play a unique role in the survival of different tumor types and establishment of supportive tumor microenvironments. The alpha and beta isoforms are expressed in a wide variety of tissues whereas the gamma and delta isoforms are primarily expressed in hematopoietic cells such as B and T cells. The PI3K alpha isoform is particularly important in breast cancer and plays an important role in tumorigenesis, supporting tumor angiogenesis and stromal interactions, making this a viable target. PIK3CA is an oncogene that codes for the alpha isoform of PI3K, (PI3Kα), more specifically for the alpha isoform of p110. The PI3k pathway is the most frequently altered pathway in human cancers including breast cancer, and has been implicated in disease progression in a significant number of patients with breast cancer. Activation of the PI3K pathway in breast cancer has been associated with resistance to endocrine therapy and disease progression. Approximately 40% of patients with Hormone Receptor positive (HR+), HER2-negative breast cancers, harbor activating mutations in the PIK3CA isoform of PI3K, which is the most common mutation in HR+ breast cancer. Patients with advanced breast cancer harboring PIK3CA mutations typically have a poor prognosis. This provides a strong rationale for targeting the PI3K pathway in breast cancer.
PIQRAY® is an oral, alpha-specific PI3K inhibitor that specifically inhibits PIK3 in the PI3K/AKT kinase signaling pathway. Further, it was shown in preclinical studies that cancer cells with PIK3CA mutations are more sensitive to PIQRAY® than those without the mutation, across a broad range of tumor types. In the SOLAR-1 Phase III trial, there was a 35% improvement in Progression Free Survival (PFS) in patients randomized to PIQRAY® plus FASLODEX®, compared to the placebo plus FASLODEX® group, among postmenopausal patients with PIK3CA-mutated, HR+/HER2- negative, advanced breast cancer, who had progressed on or following prior Aromatase Inhibitor (AI) treatment with or without a CDK 4/6 inhibitor. However in this study, only 6% had received prior CDK4/6 inhibitor therapy and there is presently limited data available, to inform treatment decisions in patients who progress on AI and CDK 4/6 inhibitor combination.
BYLieve is an ongoing, prospective, open-label, Phase II, non-comparative trial, which evaluated the benefit of PIQRAY® in combination with endocrine therapy in patients with HR+, HER-negative, PIK3CA-mutated, advanced breast cancer, who progressed on or after a prior therapy including CDK inhibitor. This study included 3 patient cohorts- Cohort A included patients who received a CDK4/6 inhibitor plus an AI as immediate prior therapy, Cohort B included patients who received a CDK4/6 inhibitor plus FASLODEX® (Fulvestrant) as immediate prior therapy, and Cohort C included patients who progressed on/after an AI and received chemotherapy or endocrine therapy as immediate prior treatment.
The authors in this publication shared findings from Cohort A group of patients, who had received CDK4/6 inhibitor plus an AI as their immediate prior therapy. Cohort A enrolled 127 patients of whom 121 patients had centrally confirmed PIK3CA mutation. Patients in Cohort A received PIQRAY® 300 mg orally once daily along with FASLODEX® 500 mg IM on Day 1 and 15 of cycle 1 followed by Day 1 treatment, of each 28 day cycle thereafter. The median patient age was 58 years. Seventy percent (70%) of patients had received one prior metastatic regimen, none of the patients had received FASLODEX® as a first-line metastatic agent, and 60% of patients had secondary endocrine resistance. The median follow up was 11.7 months. The Primary endpoint was proportion of patients alive without disease progression at 6 months. Secondary end points included Progression Free Survival (PFS), Overall Response Rate (ORR), Overall Survival (OS), and safety.
The Primary endpoint was met and the proportion of patients with confirmed PIK3CA mutation and without disease progression at 6 months was 50.4%. The median PFS was 7.3 months. Among the 121 patients in Cohort A with a confirmed PIK3CA mutation, the response rate, which was all partial responses was 17.4%, and 45.5% achieved stable disease.
Although the BYLieve trial did not have a control group to allow comparing patients in Cohort A to patients receiving other standard therapies, the authors conducted a weighted/matched analysis between the patients in Cohort A of the BYLieve trial and a Real-World similar group of 95 patients with HR+, HER2-negative, PIK3CA-mutated advanced breast cancer, who were treated with standard therapies. The Real-World patient data was obtained from the de-identified clinic-genomic database of Flatiron Health and Foundation Medicine. These 95 patients had received a wide range of regimens, with the most frequent being XELODA® (Capecitabine) monotherapy, FASLODEX® monotherapy, FASLODEX® plus IBRANCE® (Palbociclib), AFINITOR® (Everolimus) plus AROMASIN® (Exemestane), FASLODEX® plus FEMARA® (Letrozole), and IBRANCE® monotherapy.
Unadjusted results showed a median PFS of 7.3 months in BYLieve Cohort A versus 3.6 months in the Real-World cohort. Similar outcomes were noted when data were weighted by odds, propensity score matching, and exact matching.
It was concluded that the BYLieve trial is continuing to show clinically meaningful efficacy with a combination of PIQRAY® and FASLODEX® in HR+, HER2-negative, PIK3CA-mutated advanced breast cancer, post CDK inhibitor treatment, building further on the findings of SOLAR-1 trial. The matched analysis comparing BYLieve with Real-World Data in the post-CDK4/6 inhibitor setting, further supports use of PIQRAY® plus FASLODEX® for this patient group.
Alpelisib (ALP) + fulvestrant (FUL) in patients (pts) with PIK3CA-mutated (mut) hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2–) advanced breast cancer (ABC) previously treated with cyclin-dependent kinase 4/6 inhibitor (CDKi) + aromatase inhibitor (AI): BYLieve study results. Rugo HS, Lerebours F, Ciruelos E, et al. J Clin Oncol 38: 2020 (suppl; abstr 1006).
Late Breaking Abstract - ASCO 2020: Local Therapy Does Not Extend Survival in Newly Diagnosed Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. Approximately 6% of newly diagnosed breast cancer patients present with Stage IV disease. Breast surgery is often not a consideration for patients with metastatic breast cancer. However, breast surgery can be offered for palliation of symptoms, taking into consideration the risks and benefits of such intervention, in a patient with an ulcerated, bleeding, or a fungating tumor mass, that cannot be controlled with systemic therapy. It has been hypothesized based on retrospective analyses, that the addition of surgical resection of the primary tumor in the breast, to systemic therapy, in patients presenting with Stage IV disease, improved survival. Randomized clinical trials however have provided conflicting results.
E2108 is a randomized, Phase III trial which evaluated the benefit of locoregional treatment for the intact primary breast tumor, following initial systemic therapy, in newly diagnosed breast cancer patients presenting with Stage IV disease. In this study, 256 eligible patients with de novo metastatic disease, who did not progress during a 4-8 months period, while on optimal systemic therapy based on patient and tumor characteristics, were randomized to either continue systemic therapy alone (N=131) or combine it with locoregional therapy such as surgery and radiation for the intact primary breast tumor (N = 125). Of the 125 patients who received early locoregional therapy, 109 patients underwent surgery of whom 87 had free margins and 74 patients received locoregional radiation therapy. The Primary endpoint was Overall Survival (OS), and Secondary endpoint was locoregional disease control.
At a median follow up of 59 months, there was no significant difference in Overall Survival (OS) between the optimal systemic therapy plus locoregional therapy compared with optimal systemic therapy alone (3-year OS rate was 68.4% versus 67.9%; HR=1.09; P=0.63). Further, the addition of locoregional therapy to systemic therapy, also failed to improve 3-year Progression Free Survival (P=0.40). There was however significantly higher locoregional recurrence or progression in the systemic therapy alone group compared with the systemic therapy plus locoregional therapy group (3-year rate 25.6% versus 10.2%, P=0.003). Health-related Quality of Life measures such as depression, anxiety and well-being were significantly worse in patients who underwent systemic therapy plus locoregional therapy, compared with systemic therapy alone.
The authors concluded that for patients with a new diagnosis of breast cancer presenting with Stage IV disease, surgery and radiation for the primary breast tumor should not be offered, with the expectation of a survival benefit.
A randomized phase III trial of systemic therapy plus early local therapy versus systemic therapy alone in women with de novo stage IV breast cancer: a trial of the ECOG-ACRIN Research Group (E2108). Khan SA, Zhao F, Solin LJ, et al. J Clin Oncol 38: 2020 (suppl; abstr LBA2)
Dietary Supplement Use during Adjuvant Chemotherapy May Increase Risk for Breast Cancer Recurrence
SUMMARY: The Council for Responsible Nutrition reported that 77% of Americans consume dietary supplements. With the growing awareness regarding health, fitness and nutrition, the market size for dietary supplements is projected to hit a valuation of $349.4 billion by 2026.
Patients often use dietary supplements following a diagnosis of cancer, even though clinical recommendations discourage the use of antioxidant supplements during chemotherapy. One of the mechanisms of action of cytotoxic chemotherapeutic agents is through the generation of Reactive Oxygen Species (ROS). The use of dietary supplements during treatment, particularly antioxidants, could reduce the efficacy of cytotoxic agents. DELCaP study was conducted to address this concern.
DELCaP (Diet, Exercise, Lifestyle and Cancer Prognosis) trial is a prospective observational study, ancillary to an intergroup therapeutic clinical trial for high-risk breast cancer, conducted to evaluate associations between supplement use, particularly antioxidants during chemotherapy treatment, and breast cancer survival outcomes.
The Phase III SWOG S0221 trial evaluated the optimal dose and schedule of Anthracycline/Taxane adjuvant chemotherapy in women with high-risk early breast cancer. The current analysis involved a cohort of 1,134 of 2,014 patients enrolled in this study, who answered a baseline and follow-up questionnaires that included their use of dietary supplement at enrollment and during treatment. The authors then analyzed associations of dietary supplement use with clinical outcomes, after adjusting for clinical and lifestyle factors. Approximately 18% of patients used antioxidants such as Vitamins C, A, and E, Carotenoids or Coenzyme Q10 during treatment, whereas 44% of patients took multivitamins during chemotherapy.
It was noted from this analysis that the use of any antioxidant supplement (Vitamins A, C, and E, Carotenoids and Coenzyme Q10), both before and during adjuvant treatment was associated with an increased risk of recurrence versus no such use of supplements (HR=1.41; P=0.06). There was also a nonsignificant increased risk of overall mortality with the use of any antioxidant supplement (HR=1.40; P=0.14). There was a weaker relationship of outcomes with individual antioxidants and this may perhaps be due to the small numbers of patients. With regards to nonoxidants, Vitamin B12 use both before and during chemotherapy was significantly associated with poorer Disease Free Survival (HR=1.83; P<0.01) and Overall Survival (HR= 2.04; P<0.01). Use of iron during chemotherapy was also significantly associated with recurrence (HR=1.79; P<0.01), as was use both before and during treatment (HR=1.91; P=0.06). Results were similar for Overall Survival. Multivitamin use however was not associated with survival outcomes.
The researchers based on this analysis concluded that the use of antioxidant and nonantioxidant dietary supplements, but not multivitamins, before and during adjuvant chemotherapy may be associated with inferior treatment outcomes, in patients with early stage high risk breast cancer. They added that caution should be exercised by patients, when considering the use of supplements, other than a multivitamin, during chemotherapy, and patients should try to get their vitamins and minerals including antioxidants through food products..
Dietary Supplement Use During Chemotherapy and Survival Outcomes of Patients With Breast Cancer Enrolled in a Cooperative Group Clinical Trial (SWOG S0221). Ambrosone CB, Zirpoli GR, Hutson AD, et al. J Clin Oncol. 2019;38:804-814
FDA Approves TRODELVY® for Advanced Triple Negative Breast Cancer
SUMMARY: The FDA on April 22, 2020, granted accelerated approval to TRODELVY® (Sacituzumab govitecan-hziy), for adult patients with metastatic Triple-Negative Breast Cancer (TNBC), who received at least two prior therapies for metastatic disease. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease.
Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients and African American females. It is usually aggressive, and tumors tend to be high grade, and patients with TNBC are at a higher risk of both local and distant recurrence and often develop visceral metastases. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Overall survival among patients with pretreated metastatic TNBC has not changed over the past 2 decades and standard chemotherapy is associated with low response rates of 10-15% and a Progression Free Survival of only 2-3 months.
TRODELVY® is an Antibody-Drug Conjugate (ADC) in which SN-38, an active metabolite of Irinotecan, a Topoisomerase I inhibitor, is coupled to the humanized Anti-Trophoblast cell-surface antigen 2 (Trop-2) monoclonal antibody (hRS7 IgG1κ), through the cleavable CL2A linker. SN-38 cannot be given directly to patients because of its toxicity and poor solubility. Trop-2, a transmembrane calcium signal transducer, stimulates cancer-cell growth, and this cell surface receptor is overexpressed in several epithelial cancers including cancers of the breast, colon and lung, and has limited expression in normal human tissues. Trop-2 is expressed in more than 85% of breast tumors including Triple Negative Breast Cancer. Upon binding to Trop-2, the anti-TROP-2 monoclonal antibody is internalized and delivers SN-38 directly into the tumor cell, making it a suitable transporter for the delivery of cytotoxic drugs. Further, the cleavable linker enables SN-38 to be released both intracellularly into the tumor cells as well as the tumor microenvironment, thereby allowing for the delivery of therapeutic concentrations of the active drug in bystander cells to which the conjugate has not bound. Thus, TRODELVY®-bound tumor cells are killed by intracellular uptake of SN-38, whereas the adjacent tumor cells are killed by the extracellular release of SN-38.
IMMU-132-01 is a Phase I/II, basket design, open-label, single-group, multicenter trial involving patients with various types of advanced epithelial cancers, who have received at least one previous therapy for metastatic disease. (One example of a basket design is a single drug evaluated in multiple baskets, with each basket representing a different malignancy or tumor site with the same target). A total of 108 patients with metastatic Triple-Negative Breast Cancer (TNBC) were enrolled between June 2013 and February 2017. Patients received TRODELVY® 10 mg/kg IV on days 1 and 8 every 21 days. Tumor imaging was obtained every 8 weeks, and patients were treated until disease progression or intolerance to therapy. The median patient age was 55 years. Enrolled patients had a median of 3 prior anticancer regimens and 98% had received taxanes and 86% had received anthracyclines. The Primary efficacy end point was the Objective Response Rate (ORR). Other efficacy end points included Time to Response and Duration of Response in patients who had a response, the Clinical Benefit Rate (defined as a Complete or Partial Response or stable disease for at least 6 months), Progression Free and Overall Survival. The median duration of follow up for this basket of 108 patients with metastatic TNBC was 9.7 months.
The Objective Response Rate was 33.3% including a Complete Response Rate of 2.8%. The median Time to Response was 2.0 months and the median Duration of Response was 7.7 months. The Clinical Benefit Rate was 45.4%. There was no meaningful difference in response rates in the various patient subgroups including patient age, onset of metastatic disease, number of previous therapies and the presence or absence of visceral metastases. The median PFS was 5.5 months and median OS was 13.0 months. The most common adverse reactions were, possibly severe neutropenia and diarrhea, fatigue, nausea, vomiting, alopecia and abdominal discomfort.
It was concluded that TRODELVY® was associated with durable Objective Responses in patients with heavily pretreated metastatic Triple Negative Breast Cancer. This unique Antibody Drug Conjugate may be of potential benefit for other Trop-2 expressing advanced epithelial solid tumors.
Sacituzumab Govitecan-hziy in Refractory Metastatic Triple-Negative Breast Cancer. Bardia A, Mayer IA, Vahdat LT, et al. N Engl J Med. 2019;380:741-751.
FDA Approves TUKYSA® for HER2+ Breast Cancer
SUMMARY: The FDA on April 17, 2020, approved TUKYSA® (Tucatinib) in combination with Trastuzumab and XELODA® (Capecitabine), for adult patients with advanced unresectable or metastatic HER2-positive breast cancer, including patients with brain metastases, who have received one or more prior anti-HER2-based regimens in the metastatic setting. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease.
The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.
It is estimated that close to 50% of patients with HER2-positive metastatic breast cancer develop brain metastases. Systemic HER2-targeted agents, including Tyrosine Kinase Inhibitors, as well as chemotherapy have limited antitumor activity in the brain. Local therapeutic interventions for brain metastases include neurosurgical resection and Stereotactic or Whole-Brain Radiation Therapy.
TUKYSA® (Tucatinib) is an oral Tyrosine Kinase Inhibitor that is highly selective for the kinase domain of HER2 with minimal inhibition of Epidermal Growth Factor Receptor. In a Phase 1b dose-escalation trial, TUKYSA® in combination with HERCEPTIN® and XELODA® (Capecitabine) showed encouraging antitumor activity in patients with HER2-positive metastatic breast cancer, including those with brain metastases.
HER2CLIMB is an international, randomized, double-blind trial in which the combination of TUKYSA® plus HERCEPTIN® and XELODA® was compared with placebo plus HERCEPTIN® and XELODA®. A total of 612 patients with unresectable locally advanced or metastatic HER2-positive breast cancer, who were previously treated with HERCEPTIN®, PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine) were enrolled. Patients were randomly assigned in a 2:1 ratio to receive either TUKYSA® 300 mg orally twice daily throughout the treatment period (N=410) or placebo orally twice daily (N=201), in combination with HERCEPTIN® 6 mg/kg IV once every 21 days, following an initial loading dose of 8 mg/kg, and XELODA® 1000 mg/m2 orally twice daily on days 1 to 14 of each 21-day cycle. Stratification factors included presence or absence of brain metastases, ECOG Performance Status and geographic region. The median patient age was 54 years and patient demographic as well as disease characteristics at baseline were well balanced between the two treatment groups. In the total treatment population, 47.5% had brain metastases at baseline, 48.3% in the TUKYSA® combination group and 46% in the placebo combination group. The median duration of follow up in the total treatment population was 14 months. The Primary endpoint was Progression Free Survival (PFS) among the first 480 patients who underwent randomization. Secondary end points assessed in the total treatment population (612 patients) included, Overall Survival (OS), PFS among patients with brain metastases, confirmed Objective Response Rate (ORR), and safety.
The Primary endpoint of PFS at 1 year was 33.1% in the TUKYSA®-combination group and 12.3% in the placebo-combination group (HR for disease progression or death=0.54; P<0.001), and the median duration of PFS was 7.8 months and 5.6 months, respectively. This represented a 46% reduction in the risk of cancer progression or death in the TUKYSA®-combination group compared to patients who received HERCEPTIN® and XELODA® alone. The Overall Survival at 2 years was 44.9% in the TUKYSA®-combination group and 26.6% in the placebo-combination group (HR for death=0.66; P=0.005), and the median Overall Survival was 21.9 months and 17.4 months, respectively. This represented a 44% reduction in the risk of death in the TUKYSA®-combination group compared to the placebo-combination group. Among the patients with brain metastases, PFS at 1 year was 24.9% in the TUKYSA®-combination group and 0% in the placebo-combination group (HR=0.48; P<0.001), and the median PFS was 7.6 months and 5.4 months, respectively. This represented a 52% reduction in the risk of cancer progression or death in the TUKYSA®-combination group compared to the placebo-combination group. Among the patients with measurable disease at baseline, the confirmed Objective Response Rate was 40.6% in the TUKYSA®-combination group and 22.8% in the placebo-combination group (P<0.001). Common adverse events in the TUKYSA® group included diarrhea, Palmar-Plantar Erythrodysesthesia syndrome, nausea, vomiting and fatigue. Diarrhea and abnormal liver function tests were more common in the TUKYSA®-combination group than in the placebo-combination group.
It was concluded that in heavily pretreated patients with HER2-positive metastatic breast cancer, including those with brain metastases, the addition of TUKYSA® to HERCEPTIN® and XELODA® resulted in clinically significant improvement in PFS and OS, compared to the placebo-combination group. This trial is unique in that it included patients with active brain metastases, either untreated or progressing.
Tucatinib, Trastuzumab, and Capecitabine for HER2-Positive Metastatic Breast Cancer. Murthy RK, Loi S, Okines A, et al. N Engl J Med 2020;382:597-609.
ENHERTU® Highly Effective in Heavily Pretreated HER2-Positive Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.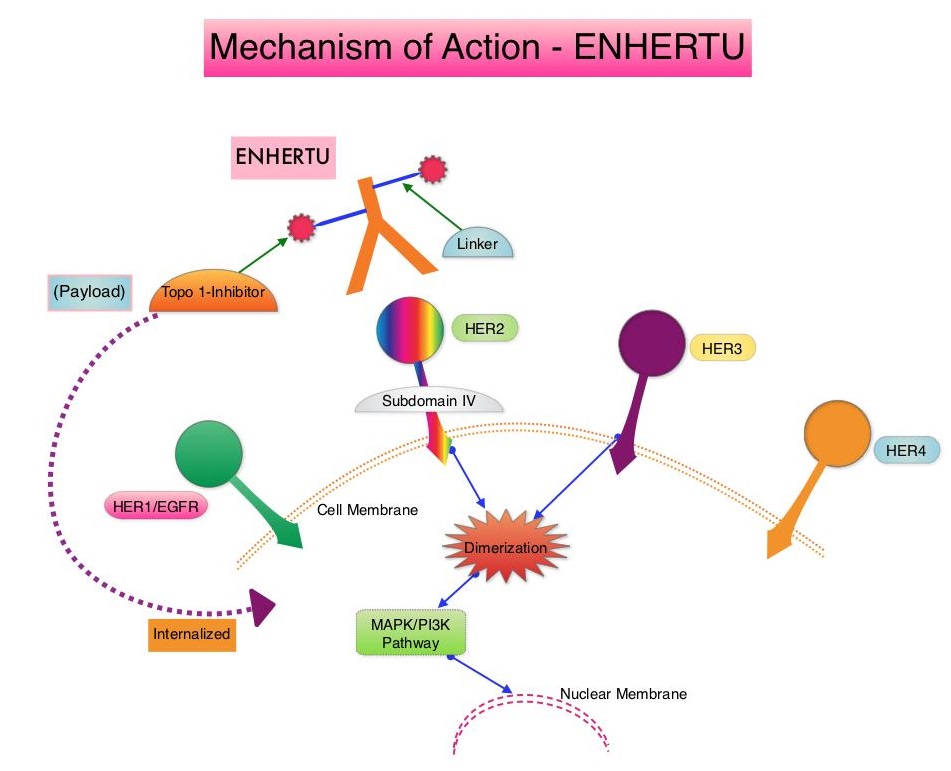
ENHERTU® is an Antibody-Drug Conjugate (ADC) composed of a humanized monoclonal antibody specifically targeting HER2, with the amino acid sequence similar to Trastuzumab, a cleavable tetrapeptide-based linker, and a potent cytotoxic Topoisomerase I inhibitor as the cytotoxic drug (payload). ENHERTU® has a favorable pharmacokinetic profile and the tetrapeptide-based linker is stable in the plasma and is selectively cleaved by cathepsins that are up-regulated in tumor cells. Unlike KADCYLA®, ENHERTU® has a higher drug-to-antibody ratio (8 versus 4), released payload easily crosses the cell membrane with resulting potent cytotoxic effect on neighboring tumor cells regardless of target expression, and the released cytotoxic agent (payload) has a short half-life, minimizing systemic exposure. In a Phase 1 dose-finding study involving patients with advanced HER2-positive breast cancer, treatment with ENHERTU® resulted in a confirmed response rate was 59.5%, and the median response duration was 20.7 months. However, the efficacy of ENHERTU® in patients with HER2-positive metastatic breast cancer, previously treated with KADCYLA® remained unclear.
DESTINY-Breast 01 study is a multicenter, single-arm, Phase II registration trial, in which 184 patients with HER2-positive, metastatic breast cancer, who had received two or more prior HER2 targeted therapies including KADCYLA®, were enrolled. Patients received ENHERTU® 5.4 mg/kg IV every 3 weeks until disease progression or unacceptable toxicity. The median age was 55 years, 53% had Hormone Receptor-positive tumors and the median number of previous lines of therapy for metastatic disease was SIX and included KADCYLA® (100%), Trastuzumab (100%), Pertuzumab (66%), and other anti-HER2 therapies (54%). The Primary end point was Objective Response Rate (ORR) assessed by Independent Central Review and Secondary endpoints included Duration of Response, Progression Free Survival (PFS) and Overall Survival (OS). The median follow up was 11.1 months.
The ORR was 60.9%, with 6% Complete Responses and 54.9% Partial Responses. The median time to response was 1.6 months and the median response duration was 14.8 months. The median PFS was 16.4 months the median OS was not reached at the time of this publication. The efficacy results were consistent across all key subgroups, including patients who had received previous PERJETA® (Pertuzumab) therapy. The most Grade 3 or higher adverse events were cytopenias, nausea, diarrhea and Interstitial Lung Disease.
It was concluded that ENHERTU® has a high level of clinical efficacy with a durable antitumor activity in a heavily pretreated patient population with HER2-positive metastatic breast cancer. The FDA in December 2019, granted accelerated approval to ENHERTU® (Trastuzumab deruxtecan) for patients with unresectable or metastatic HER2-positive breast cancer, who have received two or more prior anti-HER2-based regimens in the metastatic setting. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Breast Cancer. Modi S, Saura C, Yamashita T, et al. for the DESTINY-Breast01 Investigators. N Engl J Med 2020;382:610-621.
FDA Approves NERLYNX® for Advanced HER2-Positive Breast Cancer
SUMMARY: The FDA on February 25, 2020, approved NERLYNX® (Neratinib) in combination with XELODA® (Capecitabine) for adult patients with advanced or metastatic HER2-positive breast cancer, who have received two or more prior anti-HER2 based regimens in the metastatic setting. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes.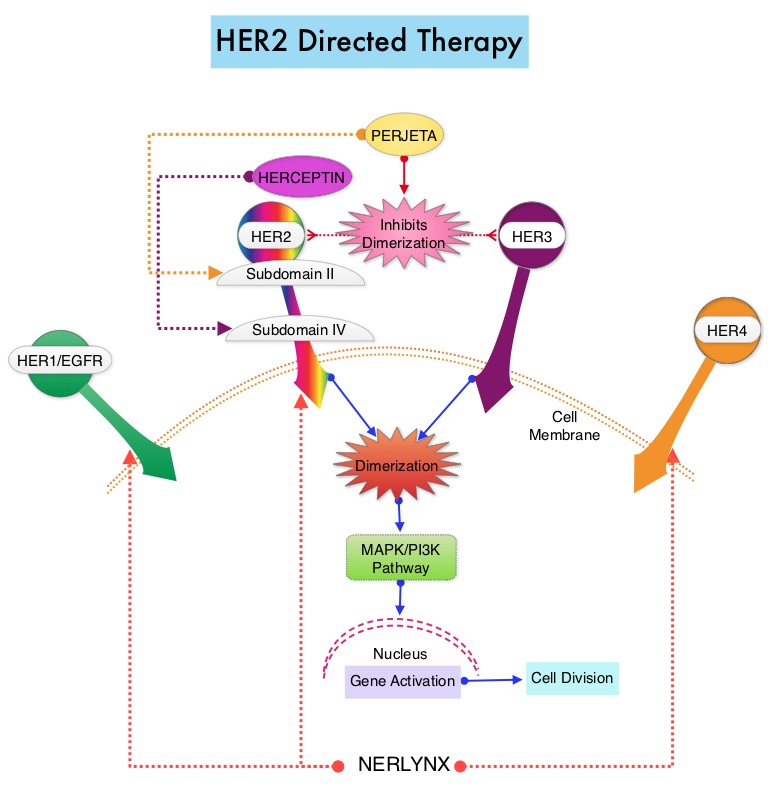
NERLYNX® (Neratinib) is a potent, irreversible, oral Tyrosine Kinase Inhibitor, of HER1, HER2 and HER4 (pan-HER inhibitor). NERLYNX® interacts with the catalytic domain of HER1, HER2, and HER4 and blocks their downstream signaling pathways, resulting in decreased cell proliferation and increased cell death. Clinical data has suggested that NERLYNX® has significant activity in suppressing HER-mediated tumor growth and is able to overcome tumor escape mechanisms experienced with current HER2-targeted and chemotherapeutic agents. It has been well known that hormone receptor positive breast cancer patients, who are also HER2-positive, have relative resistance to hormone therapy. Preclinical models had suggested that the addition of NERLYNX® could improve responses in ER positive, HER2-positive breast cancer patients. Further, NERLYNX® has clinical activity in patients with HER2-positive metastatic breast cancer. NERLYNX® is the first TKI approved by the FDA, shown to reduce the risk for disease recurrence, in patients with early stage HER2-positive breast cancer. NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly reduced the proportion of clinically relevant breast cancer relapses that might lead to death, such as distant and locoregional recurrences outside the preserved breast.
TYKERB® (Lapatinib) is a Tyrosine Kinase Inhibitor of HER2 and EGFR, and in a previously published Phase III study, a combination of TYKERB® plus XELODA® (Capecitabine) was found to be superior to XELODA® alone in women with HER2-positive advanced breast cancer, that has progressed after treatment with regimens that included an Anthracycline, a Taxane, and HERCEPTIN®. (N Engl J Med 2006; 355:2733-2743)
NALA is a multinational, randomized, open-label, Phase III trial of NERLYNX® plus XELODA® in patients with heavily pretreated Stage IV HER2-positive metastatic breast cancer. In this study, 621 patients with metastatic HER2-positive breast cancer who received two or more prior anti-HER2 based regimens in the metastatic setting were randomly assigned in a 1:1 to receive NERLYNX® 240 mg given orally once daily on days 1-21 along with XELODA® 750 mg/m2 given orally twice daily on days 1-14 for each 21-day cycle (N=307) or TYKERB® 1250 mg given orally once daily on days 1-21 along with XELODA® 1000 mg/m2 given orally twice daily on days 1-14 for each 21-day cycle (N=314). Approximately 80% had visceral metastases, and about 30% had received at least three anti-HER2 therapies. Patients in the NERLYNX® group also received antidiarrheal prophylaxis with Loperamide. Patients were treated until disease progression or unacceptable toxicity. The Co-Primary endpoints were Progression Free Survival (PFS) and Overall Survival (OS). Secondary endpoints included Objective Response Rate (ORR) and Duration of Response, Clinical Benefit Rate (CBR), time to intervention for symptomatic metastatic Central Nervous System (CNS) disease and Safety.
It was noted that the median PFS was 5.6 months for patients who received NERLYNX® with XELODA® and 5.5 months for those receiving TYKERB® with XELODA® (HR=0.76; P=0.006). This represented a 24% reduction in the risk of disease progression or death for those receiving a combination of NERLYNX® and XELODA®. The PFS rate at 12 months was 29% versus 15% respectively. The median OS was 21 months for patients receiving NERLYNX® and XELODA® compared to 18.7 months for those receiving TYKERB® and XELODA® (HR=0.88; P=0.20). The ORR was numerically higher with NERLYNX® and XELODA® combination in patients with measurable disease (32.8% versus 26.7%), and there was a statistically significant improvement in the Clinical Benefit Rate (45% versus 36%; P=0.03). The median Duration of Response was 8.5 versus 5.6 months respectively (HR=0.50; P=0.0004), favoring the NERLYNX® combination. The time to intervention for symptomatic CNS disease was significantly delayed with NERLYNX® combination versus TYKERB® combination, with an overall cumulative incidence of 22.8% versus 29.2% respectively (P= 0.043). Treatment related toxicities were similar between arms, but there was a higher rate of Grade 3 diarrhea with the NERLYNX® combination (24.4% versus 12.5% respectively). Discontinuation due to treatment related toxicities were lower with NERLYNX® combination versus TYKERB® combination (10.9% versus 14.5%).
It was concluded from this study that a combination of NERLYNX® and XELODA® significantly improved Progression Free Survival, with a trend towards improved Overall Survival, and also resulted in a delayed time to intervention for symptomatic CNS disease, among patients with heavily pretreated advanced HER2-positive breast cancer. Neratinib + capecitabine versus lapatinib + capecitabine in patients with HER2+ metastatic breast cancer previously treated with ≥ 2 HER2-directed regimens: Findings from the multinational, randomized, phase III NALA trial. Saura C, Oliveira M, Feng Y-H, et al. J Clin Oncol. 2019;37 (suppl; abstr 1002).
Neoadjuvant KEYTRUDA® Plus Chemotherapy Improves Pathological Complete Response in Triple Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (13%) will develop invasive breast cancer during their lifetime. Approximately 276,480 new cases of invasive female breast cancer will be diagnosed in 2020 and about 42,170 women will die of the disease.
Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. In addition to increasing the likelihood of tumor resectability and breast preservation, patients achieving a pCR following neoadjuvant chemotherapy have a longer Event Free Survival (EFS) and Overall Survival (OS). Those who do not achieve a pathological Complete Response tend to have a poor prognosis. For all these reasons, pCR is considered a valid endpoint for clinical testing of neoadjuvant therapy in patients with early stage TNBC. It appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway. Previously published studies have shown that presence of tumor-infiltrating lymphocytes was associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC.
KEYTRUDA® (Pembrolizumab) is a fully humanized, Immunoglobulin G4, anti-PD-1, monoclonal antibody, that binds to the PD-1 receptor and blocks its interaction with ligands PD-L1 and PD-L2. It thereby reverses the PD-1 pathway-mediated inhibition of the immune response and unleashes the tumor-specific effector T cells. Cytotoxic chemotherapy releases tumor-specific antigens and immune checkpoint inhibitors such as KEYTRUDA® when given along with chemotherapy can enhance endogenous anticancer immunity. Preliminary results from Phase I and II trials have shown that in patients with TNBC, KEYTRUDA® given along with chemotherapy in a neoadjuvant setting resulted in a high rate of pCR.
KEYNOTE-522 is an international, placebo controlled Phase III trial, conducted to evaluate the safety and efficacy of neoadjuvant KEYTRUDA® plus chemotherapy followed by adjuvant KEYTRUDA® or placebo, in patients with early stage TNBC. In this study, 1,174 patients were randomly assigned in a 2:1 ratio to receive neoadjuvant KEYTRUDA® 200 mg IV every 3 weeks (N=784) or placebo (N=390). All patients received 4 cycles of Carboplatin plus Paclitaxel, followed by 4 cycles of Doxorubicin or Epirubicin plus Cyclophosphamide, in the neoadjuvant setting. Following definitive surgery, adjuvant KEYTRUDA® or placebo was continued every 3 weeks for 9 cycles or until disease recurrence or unacceptable toxicity. Enrolled TNBC patients were newly diagnosed, treatment naïve, and included both node-negative and node-positive patients with nonmetastatic disease (Tumor Stage T1c, Nodal Stage N1-N2 or Tumor Stage T2-T4, Nodal Stage N0-N2, per AJCC criteria). Treatment groups were well balanced and patients were stratified according to nodal status, tumor size, and Carboplatin schedule (weekly versus every 3 weeks). The two Primary endpoints were pathological Complete Response (pCR) at the time of definitive surgery and Event Free Survival (EFS). The median follow up was 15.5 months.
At the first interim analysis, the pCR among the first 602 patients who underwent randomization was 64.8% in the KEYTRUDA® plus chemotherapy group, compared with 51.2% in the placebo plus chemotherapy group (P<0.001). This pCR benefit was consistent across subgroups including PD-L1 expresssion subgroups. In the PD-L1-positive population, the pCR was 68.9% in the KEYTRUDA® plus chemotherapy group compared with 54.9% in the placebo plus chemotherapy group. In the PD-L1 negative group, the pCR in the KEYTRUDA® plus chemotherapy group was 45.3% and 30.3% in the placebo plus chemotherapy group. Neoadjuvant KEYTRUDA® plus chemotherapy followed by adjuvant KEYTRUDA® showed a favorable trend for Event Free Survival compared with chemotherapy alone, although these data are still premature. Across all treatment phases, Grade 3 or higher treatment-related toxicities were 78.0% in the KEYTRUDA® plus chemotherapy group and 73.0% in the placebo plus chemotherapy group.
It was concluded that among patients with early stage Triple Negative Breast Cancer, the addition of KEYTRUDA® to neoadjuvant chemotherapy significantly increased the pathological Complete Response rate, compared to those who received placebo plus neoadjuvant chemotherapy, with a favorable trend in Event Free Survival. Pembrolizumab for Early Triple-Negative Breast Cancer. Schmid P, Cortés J, Pusztai L, et al. for the KEYNOTE-522 Investigators. N Engl J Med 2020;382:810-821
KISQALI® plus FASLODEX® Improve Overall Survival in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors. The most common subtype of metastatic breast cancer is Hormone Receptor-positive (HR-positive), HER2-negative breast cancer (65% of all metastatic breast tumors), and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients, with a median Overall Survival (OS) of 36 months. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies.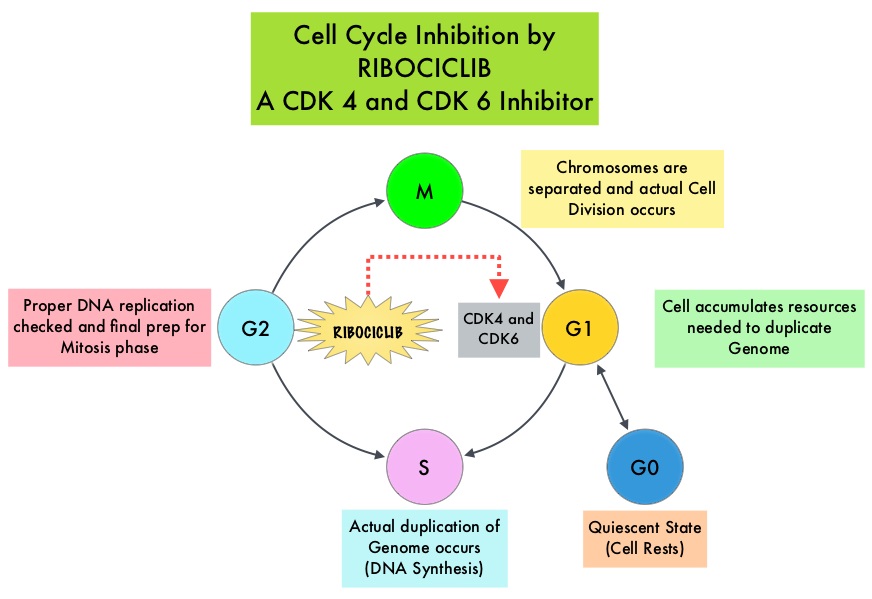
Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6, that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest. KISQALI® in combination with an Aromatase Inhibitor has been approved by the FDA for pre and perimenopausal women with HR (Hormone Receptor)-positive, HER2-negative advanced or metastatic breast cancer, as initial endocrine-based therapy. The efficacy of KISQALI® was evaluated in two prior randomized Phase III studies. In the MONALEESA-2 trial which evaluated KISQALI® in combination with FEMARA® (Letrozole) compared to FEMARA® alone, in postmenopausal women with HR-positive, HER2-negative advanced breast cancer, who received no prior therapy for their advanced breast cancer, the addition of KISQALI® to FEMARA® significantly prolonged Progression Free Survival (PFS) compared to FEMARA® alone. In the MONALEESA-7 study, KISQALI® in combination with Tamoxifen or a Non-Steroidal Aromatase Inhibitor plus ZOLADEX® (Goserelin) was compared with Tamoxifen or an Aromatase Inhibitor plus ZOLADEX®, in premenopausal or perimenopausal women with HR-positive, HER2- negative advanced breast cancer, who had not previously received endocrine therapy for advanced disease. In this study of premenopausal women, KISQALI® plus endocrine therapy significantly improved PFS and OS, compared with placebo plus endocrine therapy.
MONALEESA-3 is a randomized, double-blind, placebo-controlled Phase III study which compared the efficacy of KISQALI® in combination with FASLODEX® with FASLODEX® alone, among postmenopausal women with HR-positive, HER2-negative advanced breast cancer, who received no prior or only one line of prior endocrine therapy for advanced disease. In this trial, 726 women were randomized, of whom 367 were treatment-naïve and 345 patients had received up to one line of prior endocrine therapy for advanced disease. . Patients were randomized 2:1 to receive KISQALI® plus FASLODEX® (N=484) or placebo plus FASLODEX® (N=242). Treatment consisted of KISQALI® 600 mg orally daily 3 weeks on and 1 week off and FASLODEX® 500 mg IM on day 1 of each 28-day cycle, with an additional dose given on day 15 of cycle 1. Patients were stratified by the presence or absence of lung or liver metastases and prior endocrine therapy (first-line versus second-line). The median age in both groups was 63 years. The Primary endpoint was Progression Free Survival (PFS). Secondary end points included Overall Survival (OS), Overall Response Rate (ORR), and Safety.
In the primary analysis of the trial, the median PFS in the KISQALI® plus FASLODEX® group was 20.5 months compared to 12.8 months in the FASLODEX® plus placebo group (HR= 0.59; P<0.001). This represented a 41% reduction in the risk of disease progression. The authors now report the results of the second interim analysis of Overall Survival and an updated analysis of Progression Free Survival. The median duration of follow up for all patients was 39.4 months, and the median duration of treatment was 15.8 months in the KISQALI® group and 12.0 months in the placebo group.
There was a significant Overall Survival benefit with KISQALI® plus FASLODEX® compared to placebo plus FASLODEX®. The estimated Overall Survival at 42 months was 57.8% in the KISQALI® group compared to 45.9% in the placebo group, and this represented a 28% reduction in the relative risk of death with the KISQALI® combination (HR=0.72; P=0.00455). The survival benefit was consistent across most subgroups. The median PFS among patients receiving first line treatment was 33.6 months (95% CI, 27.1 to 41.3) in the KISQALI® group and 19.2 months in the placebo group. No new safety signals were observed during this longer follow up period.
It was concluded that KISQALI® plus FASLODEX® significantly improved Overall Survival compared to placebo plus FASLODEX® in patients with HR-positive, HER2-negative advanced breast cancer, regardless of whether they received their treatment in the frontline setting or subsequently. Overall Survival with Ribociclib plus Fulvestrant in Advanced Breast Cancer. Slamon DJ, Neven P, Chia S, et al. N Engl J Med 2020;382:514-524
Assessment of Molecular Relapse Detection in Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease. Majority of the breast cancer patients present with early stage disease without macroscopic metastatic disease. Several small proof of principle studies have shown that detection of circulating tumor DNA (ctDNA) in the peripheral blood may identify patients at risk of relapse following definitive therapy. ctDNA refers to DNA fragments that are shed into the bloodstream by cancer cells after apoptosis or necrosis. ctDNA can detect almost all molecular alterations present in cancer cells and the proportion of ctDNA that originates from a tumor depends on the anatomic location, tumor burden and cell turnover. ctDNA allows real-time monitoring for treatment response and resistance.
To assess the clinical validity of molecular relapse detection with circulating tumor DNA (ctDNA) analysis in early-stage breast cancer, the authors conducted a multicenter, prospective study in the UK over a 5 year period, and included patients, irrespective of their hormone receptor or HER2 status, who were receiving neoadjuvant chemotherapy followed by surgery or surgery before adjuvant chemotherapy. All patients had primary breast cancer without evidence of distant metastatic disease and patients scheduled to receive standard neoadjuvant chemotherapy followed by surgery consented for plasma sample collection before chemotherapy, and patients scheduled to receive adjuvant chemotherapy consented for plasma sample collection after surgery and before chemotherapy. This study enrolled 170 women, and somatic mutations were identified in 101 patients, and this formed the primary analysis cohort and secondary analyses were conducted on a combined cohort of 144 patients, including 43 patients previously analyzed in a proof of principle study (N=101+43). Tumor DNA extracted from the primary tumor was sequenced to identify somatic mutations to track in plasma, and personalized tumor-specific digital Polymerase Chain Reaction (dPCR) assays were designed to monitor these somatic mutations in serial plasma samples collected from patients every 3 months during the first year of follow up, and subsequently every 6 months for 5 years. ctDNA testing can identify genetic alterations unrelated to the lesion of interest (Clonal Hematopoiesis of Indeterminate Potential- CHIP). The authors analyzed buffy coat DNA to control for CHIP, which is common with increasing age and can potentially cause false-positive results in ctDNA analysis. The mean patient age was 54 years and the median follow up was 35.5 months. The Primary objective of the study was to assess whether patients with ctDNA detected in follow-up blood samples had worse Relapse Free Survival than patients without detectable ctDNA. Secondary end points included lead time between ctDNA detection and relapse, and association between detection of ctDNA in the diagnosis sample before neoadjuvant chemotherapy and relapse.
Results from the combined cohort of 144 patients showed that detection of ctDNA during follow up was associated with relapse (88.4% of patients who experienced relapse had antecedent ctDNA detection- P<0.001). Detection of ctDNA at diagnosis, before any treatment, was associated with larger tumor size and higher tumor grade, and patients with Triple Negative Breast Cancer (TNBC) had the highest level of ctDNA, likely due to high proliferative rates and cell turnover. This patient group was at a higher risk to relapse. Further, ctDNA detection had a median lead time of 10.7 months before clinical relapse and was associated with relapse in all breast cancer subtypes. Distant extracranial metastatic relapse was detected by ctDNA in 96% of patients. Brain-only metastasis however was less commonly detected by ctDNA, suggesting that cranial metastases are less readily detectable by ctDNA analysis.
It was concluded that detection of ctDNA during follow up of early-stage breast cancer is associated with a high risk of future relapse. A clinical trial is underway evaluating the benefit of treatment initiation at molecular relapse, before clinical development of metastatic disease. Assessment of Molecular Relapse Detection in Early-Stage Breast Cancer. Garcia-Murillas I, Chopra N, Comino-Méndez I, et al. JAMA Oncol.2019;5:1473-1478.
Multigene Testing for All Patients with Breast Cancer Could Identify Many More Mutation Carriers
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease. DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. These mutations can be inherited from either of the parents in an Autosomal Dominant pattern and a child has a 50% chance of inheriting this mutation, and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal.
It is estimated that BRCA1/2 gene mutations occur in approximately 1 in 400 women in the general population and account for 20-25% of hereditary breast cancers, about 5-10% of all breast cancers and 15% of ovarian cancers. Mutations in the BRCA1/2 genes increase breast cancer risk 45-65% by age 70 years. The risk of ovarian, fallopian tube, or peritoneal cancer, increases to 39% for BRCA1 mutations, and 10-17% for BRCA2 mutations. PALB2 (Partner And Localizer of BRCA2) gene provides instructions to make a protein that works along with the BRCA2 protein, to repair damaged DNA. PALB2 mutation is rare in sporadic breast cancer, and is considered a high-penetrance breast cancer predisposing gene associated with 14% risk of developing breast cancer by age 50 and a 35% risk of developing breast cancer by age 70. Women with a PALB2 mutation face an increased risk of triple negative breast cancer and higher risk of death from breast cancer. PALB2 gene mutations have also been implicated in ovarian, pancreatic and other malignancies.
Current guidelines recommend genetic testing in women with breast cancer who fulfill recognized or established family history or clinical criteria. However, patients with breast cancer and genetic pathogenic variants do not always have a positive family history, and these criteria miss approximately 50% of pathogenic variant carriers. Further, genetic testing based on family history or clinical criteria depends on the awareness and understanding both by the health care providers and patients, and appropriate referrals to genetic counselors. Because of limited awareness and restricted access to genetic testing and counseling services, only 20-30% of eligible patients undergo genetic testing, and 97% of estimated carriers in the general population remain unidentified, thereby missing substantial opportunities for primary prevention.
Knowledge of a patient’s genetic pathogenic variant status has important therapeutic and prognostic implications. Identifying unaffected relatives carrying pathogenic variants enables early diagnosis and cancer prevention by offering risk management options such as enhanced MRI imaging and mammography screening, risk-reducing surgeries such as prophylactic mastectomy, salpingo-oopherectomy and chemoprevention with Selective Estrogen Receptor Modulators.
The authors in this study estimated the downstream health effects, costs and cost-effectiveness of multigene testing for all patients with breast cancer, compared with current practice of BRCA testing based on clinical criteria or family history alone. In this modeling study, data was incorporated from four large breast cancer clinical trials and/or research cohorts in the United States, United Kingdom, and Australia. This analysis included 11,836 women with invasive breast cancer, regardless of the family history, and compared lifetime costs and effects of high-risk BRCA1/BRCA2/PALB2 (multigene) testing of all unselected patients with breast cancer (Strategy A) with BRCA1/BRCA2 testing based on family history or clinical criteria (Strategy B), in UK and US populations, from January 1, 2018, through June 8, 2019.
Affected patients with BRCA/PALB2 mutations could undertake contralateral preventive mastectomy and BRCA carriers could choose Risk-Reducing Salpingo-Oophorectomy (RRSO). If patients had a BRCA1/BRCA2/PALB2 pathogenic variant, their first-degree relatives undergo testing for the familial pathogenic variant. If the first-degree relative had a BRCA1/BRCA2/PALB2 pathogenic variant, second-degree relatives undergo testing. Unaffected relative carriers could undergo MRI or mammography screening, chemoprevention, or risk-reducing mastectomy for breast cancer risk and RRSO for ovarian cancer risk. This analysis incorporated lifetime risks and long-term consequences to provide a lifetime horizon. Incidence of ovarian cancer, breast cancer, excess deaths due to heart disease, and the overall population effects were estimated.
Multigene testing was restricted to BRCA1/BRCA2/PALB2, to comply with the ACCE framework for genetic testing, which was advocated for clinical applicability of genetic testing. The ACCE framework includes Analytic validity which is technical test performance, Clinical validity which is the ability of a genetic test to identify or predict accurately and reliably the clinically defined disorder or phenotype of interest, Clinical utility which is evidence that a genetic test improves clinical outcomes measurably and that it adds value for patient management decision making compared with current management without genetic testing, and ELSI which are the complex Ethical, Legal, and Social Implications associated with genetic tests.
This study showed that unselected BRCA1/BRCA2/PALB2 testing for all patients at breast cancer diagnosis was extremely cost-effective compared with BRCA1/BRCA2 testing based on clinical criteria or family history for both UK and US health systems, with incremental cost-effectiveness ratios of £10,464 or £7,216 and $65,661 or $61,618 per Quality-Adjusted Life-Year, respectively. Quality-Adjusted Life-Year (QALY) is a measurement of health outcomes in economic evaluations recommended by NICE (National Institute of Health and Clinical Excellence). This is well below UK and US cost-effectiveness thresholds. The authors estimated that one year’s unselected panel genetic testing could prevent 1142 cases of breast cancer, 959 cases of ovarian cancer, and 633 deaths related to breast or ovarian cancer in the UK. In the US, one year’s unselected panel genetic testing could prevent 5478 cases of breast cancer, 4255 cases of ovarian cancer, and 2406 deaths related to breast or ovarian cancer.
It was concluded from this analysis that unselected, high-risk multigene testing for all women with breast cancer is extremely cost-effective, compared with testing based on family history or clinical criteria, and could identify many more mutation carriers who can benefit from precision prevention. The authors added that these findings support changing current policy to expand genetic testing to all women with breast cancer. A Cost-effectiveness Analysis of Multigene Testing for All Patients with Breast Cancer. Sun L, Brentnall A, Patel S, et al. JAMA Oncol. 2019;5:1718-1730
CDK4/6 Inhibitors May Replace Chemotherapy in HR-Positive, HER2-Negative Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 279,100 new cases of invasive breast cancer will be diagnosed in 2020 and about 42,690 individuals will die of the disease. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and the most common subtype of metastatic breast cancer is Hormone Receptor-positive (HR-positive), HER2-negative breast cancer (65% of all metastatic breast tumors), and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients with a median Overall Survival (OS) of 36 months. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies.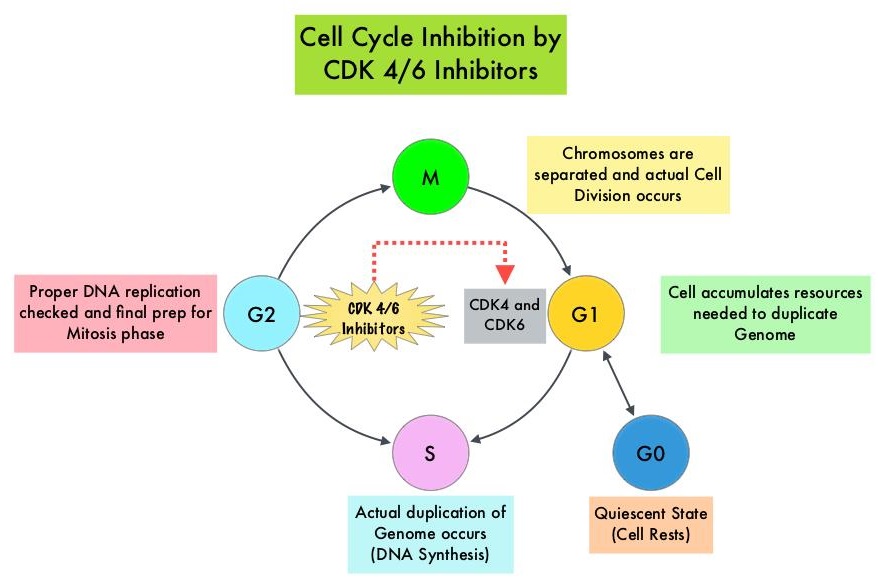
Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
Even though major international oncology treatment guidelines recommend a sequence of endocrine based therapies with or without targeted therapies in postmenopausal women with HR-positive, HER2-negative metastatic breast cancer, Real-World Data suggests that upfront use of chemotherapy remains common even in the absence of visceral crisis. This treatment approach may partly be due to paucity of data directly comparing hormonal therapies with chemotherapy regimens, in this patient group. To provide guidance with additional evidence, the authors conducted a comprehensive systematic review and network meta-analysis to evaluate the efficacy and activity of several first or second line hormonal therapy and chemotherapy regimens that have been investigated in randomized controlled trials, and the researchers aimed to compare these two different approaches.
This analysis included all Phase II and III randomized controlled trials investigating chemotherapy with or without targeted therapies and hormone therapies with or without targeted therapies as first-line or second-line treatments, or both, in postmenopausal women with HR-positive, HER2-negative metastatic breast cancer. Relevant examples of new targeted therapies are mTOR inhibitor Everolimus (AFINITOR®), CDK4/6 inhibitors Palbociclib (IBRANCE®), Ribociclib (KISQALI®) and Abemaciclib (VERZENIO®), and PI3K inhibitor Alpelisib (PIQRAY®), which are used in combination with endocrine therapy. Following a literature search on PubMed, Embase, Cochrane Central Register of Clinical Trials, Web of Science, and online archives of the most relevant international oncology conferences published between Jan 1, 2000 and Dec 31, 2017, 140 studies were selected, comprising of 50,029 patients. Studies exclusively enrolling premenopausal patients and those with HER2-positive or triple-negative breast cancer were excluded from this analysis. The median age was 58 yrs and median follow up was 20 months. All treatments were compared to Anastrozole (ARIMIDEX®) and to CDK4/6 inhibitor Palbociclib (IBRANCE®) plus Letrozole (FEMARA®). The Primary outcome was Progression Free Survival (PFS) and the Secondary outcome was Overall Response Rate.
In this analysis, it was noted that CDK4/6 inhibitors and PIK3K inhibitor (in patients with PIK3CA mutation) along with endocrine therapy was superior to standard endocrine therapy such as Anastrozole alone or Fulvestrant (FASLODEX®) alone, with significantly better PFS. Chemotherapy regimens with or without targeted agents were not significantly better than CDK4/6 inhibitors plus endocrine therapy. Further, the combination of CDK4/6 inhibitors plus endocrine therapy was associated with a favorable toxicity profile compared to chemotherapy. There were no significant differences noted in PFS among the three CDK4/6 inhibitors in combination with an Aromatase Inhibitor or Fulvestrant.
The authors concluded that in the first and second line setting, CDK4/6 inhibitors plus endocrine therapies are superior to standard single agent endocrine therapies in terms of Progression Free Survival. Moreover, no chemotherapy regimen with or without targeted therapy is significantly better than CDK4/6 inhibitors plus endocrine therapies in terms of Progression Free Survival. The researchers added that this is the first study to directly compare all three CDK4/6 inhibitors combined with an Aromatase Inhibitor or Fulvestrant. Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2-negative, metastatic breast cancer: a systematic review and network meta-analysis. Giuliano M, Schettini F, Rognoni C, et al. Lancet Oncol. 2019;20:1360-1369.
FDA Approves ENHERTU® for Advanced HER2-Positive Breast Cancer
SUMMARY: The FDA on December 20, 2019, granted accelerated approval to ENHERTU® (Trastuzumab deruxtecan) for patients with unresectable or metastatic HER2-positive breast cancer, who have received two or more prior anti-HER2-based regimens in the metastatic setting. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival (OS). The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Patients progressing on Dual HER2 blockade often receive KADCYLA® which results in an Objective Response Rate (ORR) of 44% and a median PFS of 9.6 months, when administered after HERCEPTIN® and a taxane. There is however no standard treatment option for this patient population following progression on KADCYLA®.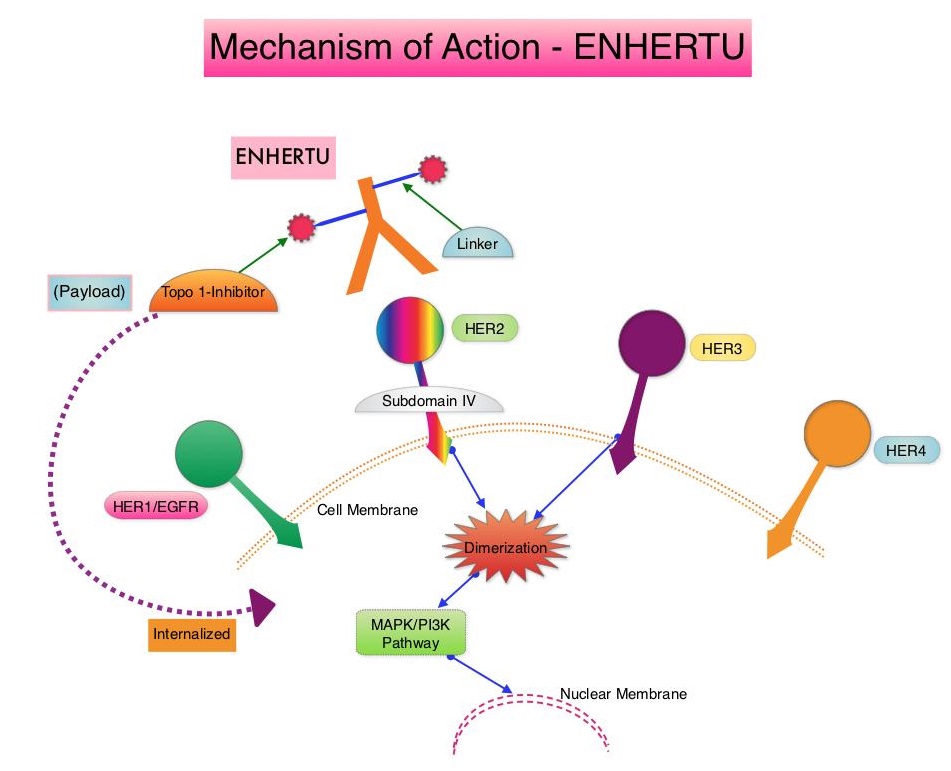
ENHERTU® is an Antibody-Drug Conjugate (ADC) composed of a humanized monoclonal antibody specifically targeting HER2, with the amino acid sequence similar to Trastuzumab, a cleavable tetrapeptide-based linker, and a potent cytotoxic Topoisomerase I inhibitor as the cytotoxic drug (payload). ENHERTU® has a favorable pharmacokinetic profile and the tetrapeptide-based linker is stable in the plasma and is selectively cleaved by cathepsins that are up-regulated in tumor cells. Unlike KADCYLA®, ENHERTU® has a higher drug-to-antibody ratio (8 versus 4), released payload easily crosses the cell membrane with resulting potent cytotoxic effect on neighboring tumor cells regardless of target expression, and the released cytotoxic agent (payload) has a short half-life, minimizing systemic exposure. In a Phase 1 dose-finding study involving patients with advanced HER2-positive breast cancer, treatment with ENHERTU® resulted in a confirmed response rate was 59.5%, and the median response duration was 20.7 months. However, the efficacy of ENHERTU® in patients with HER2-positive metastatic breast cancer, previously treated with KADCYLA® remained unclear.
The present FDA approval was based on DESTINY-Breast01 study, which is a multicenter, single-arm, Phase II trial, in which 184 patients with HER2-positive, metastatic breast cancer, who had received two or more prior HER2 targeted therapies including KADCYLA®, were enrolled. Patients received ENHERTU® 5.4 mg/kg IV every 3 weeks until disease progression or unacceptable toxicity. The median age was 55 years, 53% had Hormone Receptor-positive tumors and the median number of previous lines of therapy for metastatic disease was six and included KADCYLA® (100%), Trastuzumab (100%), Pertuzumab (66%), and other anti-HER2 therapies (54%). The Primary end point was Objective Response Rate (ORR) assessed by Independent Central Review and Secondary endpoints included Duration of Response, Progression Free Survival (PFS) and Overall Survival (OS). The median follow up was 11.1 months. The ORR was 60.9%, with 6% Complete Responses and 54.9% Partial Responses. The median time to response was 1.6 months and the median response duration was 14.8 months. The median PFS was 16.4 months the median OS was not reached at the time of this publication. The most Grade 3 or higher adverse events were cytopenias, nausea, diarrhea and Interstitial Lung Disease.
It was concluded that ENHERTU® has a high level of clinical efficacy with a durable antitumor activity in a pretreated patient population with HER2-positive metastatic breast cancer. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Breast Cancer. Modi S, Saura C, Yamashita T, et al. for the DESTINY-Breast01 Investigators. N Engl J Med. 2019 Dec 11. doi: 10.1056/NEJMoa1914510. [Epub ahead of print]
Long-Term Breast Cancer Preventive Benefit with ARIMIDEX®
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Some high risk factors for the development of breast cancer include first-degree relative with breast cancer at any age, first-degree relative with bilateral breast cancer who developed the first breast cancer at 50 years of age or less, Lobular Carcinoma In Situ (LCIS), Atypical ductal or lobular hyperplasia in a benign lesion, and Ductal Carcinoma In-Situ (DCIS).
Previously published studies have shown a 38% reduction in all breast cancers and 50% reduction of ER-positive tumors with the use of SERMs (Selective Estrogen Receptor Modulators) such as Tamoxifen and Raloxifene for breast cancer prevention. Further it has been shown that the effects of Tamoxifen continue with a constant 29% annual preventive effect for at least 15 years after completion of treatment. A further improvement in breast cancer incidence short-term was seen in two trials using, two Aromatase Inhibitors, ARIMIDEX® (Anastrozole) in the International Breast Cancer Intervention Study II (IBIS-II) and AROMASIN® (Exemestane) in the MAP.3 trial. However in the MAP.3 study, the study was unblinded after the initial publication and a post-treatment effect, as was seen with Tamoxifen, was not possible.
The International Breast Cancer Intervention Study II (IBIS-II) is an international, randomized, double-blind, placebo-controlled trial, which was initiated in 2003. In this trial, 3864 postmenopausal women aged 40-70 years, at increased risk of developing breast cancer were recruited and were randomly assigned (1:1) to either ARIMIDEX® 1 mg orally daily (N=1920) or matching placebo (N=1944) daily for 5 years. After treatment completion, women were followed on a yearly basis and data was collected on breast cancer incidence, death, incidence of other cancers, and major adverse events (cardiovascular events and fractures). The median age at study entry was 59 years. The exclusion criteria for this study included premenopausal status, prior breast cancer including Ductal Carcinoma In Situ (DCIS) diagnosed more than 6 months before trial entry, current or previous Tamoxifen, Raloxifene, or other SERM use for more than 6 months, or previous or planned prophylactic mastectomy. Unblinding was only permitted if the participant developed breast cancer, when a clinician considered there to be valid medical or safety reasons. The Primary outcome was the development of histologically confirmed breast cancer, either invasive or non-invasive (DCIS), particularly during the post-5-year time period. Secondary outcomes were ER-positive breast cancer, breast cancer mortality, incidence of other cancers, cardiovascular disease, fractures, and all-cause mortality. The decision to analyze the data was made without looking at the results before hand. The first analysis after a median follow-up of 60 months showed a significant reduction (53%) in incidence for all breast cancer (including DCIS). The authors now report the results on the extended duration of benefit of ARIMIDEX® in preventing breast cancer, for up to 12 years after trial entry.
After a median follow up of 10.9 years for this analysis, women assigned to the ARIMIDEX® group were 49% less likely to develop breast cancer than women assigned to the placebo arm of the study ((HR=0.51, P<0.0001). The reduction in incidence in the first 5 years of follow up was 61% (HR=0.39; P<0.0001), and a smaller but still significant 36% reduction (HR=0.64; P=0•014) was seen in subsequent years, which was still larger than that seen for Tamoxifen in previous trials, and the effects in the two time periods was not significantly different (P=0.08). Invasive ER-positive breast cancer was reduced by 54% with ARIMIDEX® treatment (HR=0.46; P<0.0001), with a continued significant effect observed in the post-treatment follow up period. A 59% reduction in DCIS overall was observed (HR=0.41; P=0.0081), with a very large reduction noted in those cases known to be ER-positive (HR = 0.22; P<0.0001). A significant decrease in non-breast cancers was observed in the ARIMIDEX® group, primarily contributed by non-melanoma skin cancer (P=0.0042), and no excess rates of fractures or cardiovascular disease was observed.
The authors concluded that this updated analysis shows a continuing long-term effect of 5 years of ARIMIDEX® treatment, in preventing breast cancer, in high-risk postmenopausal women. These new results strongly suggest that ARIMIDEX® should be preferred therapy for breast cancer prevention in postmenopausal women at increased risk for the disease, with Tamoxifen used for women who experience severe side effects from ARIMIDEX®. Use of anastrozole for breast cancer prevention (IBIS-II): long-term results of a randomised controlled trial. Cuzick J, Sestak I, Forbes JF, et al. The Lancet. Published:December 12, 2019. DOI:https://doi.org/10.1016/S0140-6736(19)32955-1
Supplemental MRI Screening for Women with Extremely Dense Breast Tissue
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Screening mammography complemented by breast self exam and clinical breast exam has resulted in early detection of breast cancer and successful outcomes. Even though mammography is a sensitive screening test, a small percentage of breast cancers may not show up on mammograms but may be palpable on examination by the patient or the clinician. Further, mammograms are less likely to find breast tumors in younger women with dense breast tissue. A breast Magnetic Resonance Imaging (MRI) is more sensitive than mammography although the specificity of a breast MRI is lower, resulting in a higher rate of false-positive findings and potentially unnecessary biopsies. Microcalcifications in the breast can be missed by a breast MRI. Taking these factors into consideration, appropriate utilization of breast MRI becomes relevant.
The American Cancer Society (ACS) recommends an annual MRI as an adjunct to screening mammogram and clinical breast exam in certain groups with increased risk of breast cancer. In a study published by Stout NK, et al., (JAMA Intern Med. 2014;174:114-121), it was noted that breast MRI was over utilized in those who did not fit the ACS criteria and was under utilized in those with documented genetic mutations. Routine breast MRI screening is not recommended for a new breast cancer diagnosis or for breast cancer surveillance and should only be considered for the group of individuals who have the most benefit. Breast MRI is performed preferably between days 7-15 of menstrual cycle for premenopausal women, using a dedicated breast coil, with the ability to perform a biopsy under MRI guidance by experienced radiologists, during the same visit.
DENSE trial is a multicenter, randomized, controlled study which evaluated the effect of supplemental Magnetic Resonance Imaging (MRI) on the incidence of interval cancers, in women with extremely dense breast tissue. In this trial, 40,373 women between the ages of 50 and 75 years with extremely dense breast tissue and normal results on screening mammography were randomly assigned in a 1:4 ratio to a group that was invited to undergo supplemental MRI (N=8061) or to a group that received mammography screening alone (N=32,312). Of the women who were invited to undergo MRI, 59% accepted the invitation (N=4783). All MRI examinations were performed with the use of a dedicated bilateral breast coil. The Primary endpoint was the difference in the incidence of interval cancers during a 2-year screening period, between the mammography screening-only group and MRI-invitation group. Secondary endpoints included the recall rate for additional examination, the cancer-detection rate on MRI, the false positive rate, the positive predictive value, and tumor characteristics.
The interval cancer rate was 2.5 per 1000 screenings in the MRI-invitation group and 5.0 per 1000 screenings in the mammography-only group (P<0.001). The MRI cancer-detection rate among the women who actually underwent MRI screening was 16.5 per 1000 screenings. The Positive Predictive Value of a positive MRI result was 17.4%, the Positive Predictive Value of an indication for biopsy was 23.9% and the Positive Predictive Value of a biopsy was 26.3%. The false positive rate was 79.8 per 1000 screenings. As a result of the MRI screening, 300 women underwent a breast biopsy and of these women, breast cancer was diagnosed in 79 women, of whom 64 had invasive breast cancer and 15 were diagnosed with DCIS.
The authors concluded that the use of supplemental MRI screening in women with extremely dense breast tissue and normal results on mammography resulted in the diagnosis of significantly fewer interval cancers than mammography alone, during a 2-year screening period. Whether a reduction in interval cancers is an appropriate surrogate for improved Overall Survival, remains unclear. Supplemental MRI Screening for Women with Extremely Dense Breast Tissue. Bakker MF, de Lange SV, Pijnappel RM, et al. for the DENSE Trial Study Group. N Engl J Med 2019; 381:2091-2102
Early Adjuvant Chemotherapy Dose Reductions Can Impact Breast Cancer Survival
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Patients with early stage breast cancer often receive adjuvant chemotherapy and this is even more so true for HER positive and triple negative (ER, PR and HER negative) breast cancer patients, who are at an increased risk to develop recurrent disease. Meta-analyses conducted by the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) has shown a 20-25% relative risk reduction in breast cancer mortality with first-generation adjuvant chemotherapy regimens such as CMF (Cyclophosphamide/Methotrexate/Fluorouracil) and additional survival benefit with the Anthracyclines and Taxane based regimens.
Chemotherapy dose reductions are often considered for patients with obesity, BSA of more than 2.0 m2, age over 65 years, comorbidities such as kidney disease or diabetes, and febrile neutropenia. ASCO guidelines recommend full weight-based chemotherapy doses in the treatment of obese patients. Dose reductions to less than 85% of the optimal (total cumulative) chemotherapy dose (mg/m2) and cycle delay have shown inferior survival outcomes in both prospective and some retrospective studies. However, it is unclear if dose reductions made for third-generation Anthracycline/Taxane-based regimens as well as sequential regimens such as Taxanes following Anthracycline based regimens, has an impact on survival.
The authors therefore performed a retrospective analysis to evaluate the effect of Total Cumulative Dose (TCD) of adjuvant chemotherapy, on breast cancer outcomes, in women diagnosed with Stage I-III, Hormone Receptor positive or negative, HER2-negative breast cancer, treated with adjuvant FEC (5-FU 500 mg/m2, Epirubicin 100 mg/m2, Cyclophosphamide 500 mg/m2) for 3 cycles followed by Docetaxel 100 mg/m2 for 3 cycles (FEC-D chemotherapy regimen) from 2007 through 2014. Using the historical cutoff of 85% as the optimal Total Cumulative Dose (TCD), this study focused on data from the Alberta Cancer Registry on 1,302 women with Stage I to III breast cancer.
The TCD for cycles 1-6 of less than 85% or 85% and more was calculated. The majority of patients (84%) received 85% or more of the TCD across all six cycles. Sixteen percent (16%) received reduced doses (less than 85% of TCD). Those receiving a TCD of 85% or more were more likely to be younger (median age of 54 yrs) and premenopausal, with a lower number of comorbidities, compared with those with a TCD of less than 85%. The average cumulative dose was also calculated for early (cycles 1-3) and late (cycles 4-6) chemotherapy, to explore the effects of early (FEC) versus late (Docetaxel only) dose reduction. The median follow up was 60 months.
It was noted that the amount of chemotherapy delivered had a significant impact on survival. Patients receiving a TCD of 85% or more had a 5-year Disease Free Survival (DFS) of 85.9% versus 79.2% for those receiving a lower TCD (P=0.025). The 5-year Overall Survival (OS) was also superior at 88.8% versus 80.7% (P<0.001), respectively. When the researchers split the lower TCD group into two cohorts based on dose reduction during cycles 1-3 versus cycles 4-6, they found that outcomes were not compromised when dose reduction occurred only during the later cycles (which were the only cycles to include Docetaxel), suggesting that late dose reductions in chemotherapy may not have as much of an impact on DFS and OS, compared with early dose reductions. The authors hypothesized that majority of cancer cells that are sensitive to chemotherapy are eradicated during the first few treatments, rather than in the later treatments. Further, the amount of Docetaxel that was prescribed in the last three cycles may be higher than needed for the FEC-D regimen. Menopausal status, use of G-CSF and dose delay of 14 days or more, were not shown to affect OS.
It was concluded that early dose reductions in adjuvant FEC-D chemotherapy results in inferior outcomes, with adverse affect on DFS and OS. Conversely, late reductions in chemotherapy dose (Docetaxel only) appear to have minimal impact on survival. Based on this data, the authors recommended that Medical Oncologists should strive to deliver full-dose FEC when prescribing adjuvant FEC-D chemotherapy for breast cancer. Impact of Cumulative Chemotherapy Dose on Survival With Adjuvant FEC-D Chemotherapy for Breast Cancer. Veitch Z, Khan OF, Tilley D, et al. J Natl Compr Canc Netw. 2019;17:957-967.
Late Breaking Abstract - ESMO 2019: VERZENIO® plus FASLODEX® Combination Improves Overall Survival in HR+ Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
VERZENIO® (Abemaciclib) is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against cyclin D1/CDK 4 and cyclin D3/CDK 6, in enzymatic assays, but potentially less toxic than earlier pan-CDK inhibitors. At higher doses, only VERZENIO® causes significant cancer cell death, compared with other CDK4/6 inhibitors, suggesting that this drug may be affecting proteins, other than CDK4/6. Additionally, preclinical studies have demonstrated that VERZENIO® may have additional therapeutic benefits for a subset of tumors that are unresponsive to treatment or have grown resistant to other CDK4/6 inhibitors. It has also been shown to cross the blood-brain barrier.
The FDA approved VERZENIO® in combination with FASLODEX® (Fulvestrant) in 2017, for women with Hormone Receptor positive (HR-positive), HER2-negative, advanced or metastatic breast cancer, with disease progression following endocrine therapy. This approval was based on the MONARCH-2 study. VERZENIO® was also approved as monotherapy for women and men with HR-positive, HER2-negative advanced or metastatic breast cancer, with disease progression following endocrine therapy and prior chemotherapy in the metastatic setting, based on improved Objective Response Rate (ORR) and PFS (MONARCH-1 study).
MONARCH-2 is an International, double-blind, Phase III study in which 669 patients were randomized in a 2:1 ratio to receive either VERZENIO® plus FASLODEX® (N=446) or placebo plus FASLODEX® (N=223). Enrolled patients had HR-positive, HER2-negative metastatic breast cancer, with disease progression while receiving neoadjuvant or adjuvant endocrine therapy, within 12 months of adjuvant endocrine therapy, or while receiving endocrine therapy for metastatic breast cancer. Patients must not have received more than one endocrine therapy or any prior chemotherapy for metastatic breast cancer. Randomized patients received either VERZENIO® 150 mg or placebo orally twice daily plus FASLODEX® 500 mg IM on Day 1 and Day 15 of cycle 1 and then on Day 1 of cycle 2 and beyond (28-day cycles). Treatment was continued until disease progression or unmanageable toxicities. The mean patient age was 60 years, 82% of patients were postmenopausal, 72% had measurable disease, 56% had visceral disease, and 25% had primary endocrine therapy resistance. About 60% of patients had received chemotherapy in the adjuvant or neoadjuvant setting and 69% of the patients had prior therapy with Aromatase Inhibitors (AI). The Primary end point was Progression Free Survival (PFS), and Secondary end points included Overall Survival (OS), Objective Response Rate (ORR), Duration of Response, Clinical Benefit Rate, Quality of Life, and safety. The authors had previously reported the PFS data and the median PFS for the group receiving VERZENIO® plus FASLODEX® was 16.4 months compared with 9.3 months for those taking placebo with FASLODEX® (HR= 0.55; P<0.0001).
The authors now reported the Overall Survival (OS) data. At a median follow up of 47.7 months, the median OS with the combination of VERZENIO® plus FASLODEX® was 46.7 months compared with 37.3 months for placebo plus FASLODEX® (HR=0.757; P=0.0137). The OS benefit was consistently seen across subgroups, including those with poor prognostic factors such as visceral metastasis (HR=0.675) and primary endocrine therapy resistance (HR=0.686). The updated median PFS with VERZENIO® plus FASLODEX® was 16.9 months compared to 9.3 months with placebo plus FASLODEX® (HR=0.53; P<0.001). The time to second disease progression (PFS2) was also improved with the addition of VERZENIO® at a median 23.1 months versus 20.6 months with placebo plus FASLODEX® (HR, 0.675). VERZENIO® combination also showed a statistically significant improvement in median chemotherapy-free survival compared with placebo/FASLODEX® combination at 25.5 months and 18.2 months, respectively (HR, 0.638). The most common adverse events in the VERZENIO® versus placebo groups were diarrhea neutropenia, nausea and fatigue.
It was concluded that the addition of VERZENIO® to FASLODEX® resulted in a statistically significant improvement in Overall Survival among patients with HR-positive, HER2-negative breast cancer, who had progressed on prior endocrine therapy, significantly delaying the receipt of subsequent chemotherapy. MONARCH 2: Overall survival of abemaciclib plus fulvestrant in patients with HR+, HER2 advanced breast cancer. Sledge Jr GW, Toi M, Neven P, et al. Presented at 2019 ESMO Congress; September 27 to October 1, Barcelona, Spain. Abstract LBA6_PR.
Primary Breast Tumor Surgery Improves Overall Survival in HER2 Positive Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Approximately 50% of HER2-positive breast cancers are Hormone Receptor positive. Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine). Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival. The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Not all HER2-positive, Hormone Receptor positive metastatic breast cancer patients, are candidates for chemotherapy. These patients however may benefit from anti-HER2 targeted therapy given along with endocrine therapy.
Breast surgery is often not a consideration for patients with metastatic breast cancer. However, breast surgery can be offered for palliation of symptoms, taking into consideration the risks and benefits of such intervention, in a patient with an ulcerated, bleeding, or a fungating tumor mass, that cannot be controlled with systemic therapy. A few previously published studies which evaluated the addition of surgical resection of the primary tumor to systemic therapy, among patients with metastatic HER2-positive breast cancer, have provided mixed results, with one study showing improved outcomes. The authors therefore conducted a retrospective study to examine the impact of primary breast tumor resection on survival, in HER2-positive Stage IV breast cancer patients, treated with HER2 targeted therapy.
This retrospective cohort study included records of 3,231 women with HER2 positive Stage IV breast cancer from the National Cancer Database, from 2010 to 2012. Of these women, 89.4% had received chemotherapy/anti-HER2 targeted therapies, 37.7% had received endocrine therapy, 31.8% had received radiation and 25% of the patients had bone only metastasis. Overall, 1,130 women (35%) underwent primary breast tumor resection. The mean age of those who had surgery was 56 years and the Primary endpoints were receipt of surgery and Overall Survival (OS).
At a median follow-up of 21.2 months, the median OS for patients who had surgery was 25 months compared with 18 months for those who did not undergo surgical resection (HR=0.56; P=0.0004). This suggested a 44% reduction in the risk of death with primary breast tumor resection. Patients having Medicare/other government or private insurance, as well as those who received radiation, chemotherapy/anti-HER2 targeted therapies and endocrine therapy were more likely to have surgery. Additionally, Caucasian women were also more likely than non-Hispanic black women, to have surgery. These findings suggested that women with Medicare or private insurance as well as white women were also more likely to have surgery and less likely to die of their disease, than non-Hispanic black women, and those with Medicaid or no insurance.
It was concluded that after controlling for independent variables, surgery of the primary site, in patients with metastatic HER2 positive breast cancer who are managed with present day contemporary treatment, is associated with improved Overall Survival. Breast surgery should therefore be discussed as a part of the treatment strategy, for qualified women, in this patient group. The impact of primary tumor surgery on survival in HER2 positive stage IV breast cancer patients in the current era of targeted therapy. Mudgway R, Chavez de Paz Villaneva C, Lin AC, et al. Presented at AACR Annual Meeting; March 29 to April 3, 2019; Atlanta, GA. Abstract 4873.
KISQALI® Improves Overall Survival in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. About 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Breast cancer is the second leading cause of cancer death in the US. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. The incidence of breast cancer among women under the age of 50 has been increasing by 0.2% per year. Premenopausal breast cancer may be biologically different than post menopausal breast cancer and diagnosis of breast cancer at a young age has been associated with adverse outcomes and less sensitivity to endocrine therapy. Further, premenopausal women are often excluded from hormone therapy trials. The incidence of metastatic disease at the time of diagnosis among patients with Hormone Receptor (HR)- positive breast cancer, has been increasing by about 2% per year.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in HR-positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
There are presently three CDK4/6 inhibitors approved by the FDA and they include KISQALI® (Ribociclib), IBRANCE® (Palbociclib) and VERZENIO® (Abemaciclib). All three agents have demonstrated similar, significantly prolonged Progression Free Survival (PFS) when administered in combination with endocrine therapy, as first-line treatment, in women with HR-positive metastatic breast cancer (MONALEESA-2 with KISQALI®, PALOMA-2 with IBRANCE® and MONARCH-3 with VERZENIO®). These trials for the first-line treatment of advanced breast cancer however excluded premenopausal women. The toxicities were slightly different with neutropenia more commonly encountered in the IBRANCE® and KISQALI® studies and diarrhea more often noted with VERZENIO®. KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6 that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest. It is four times more selective for CDK4 than for CDK6.
The MONALEESA-7 trial is an international, randomized, double-blind, placebo-controlled, Phase III trial in which KISQALI® in combination with endocrine therapy was compared with placebo in combination with endocrine therapy, in premenopausal or perimenopausal women with HR-positive, HER2- negative advanced breast cancer. Patients (N=672) were randomly assigned in a 1:1 ratio, to receive KISQALI® at 600 mg orally once daily for 21 days of each 28 day cycle (N=335), or matching placebo (N=337). Both groups received ZOLADEX® (Goserelin) 3.6 mg administered subcutaneously on day 1 of each 28 day cycle. Patients also received either a nonsteroidal Aromatase Inhibitor (Letrozole 2.5 mg or Anastrozole 1 mg) or Tamoxifen 20 mg, orally once daily continuously. The choice of endocrine therapy was made on the basis of the patient’s previous adjuvant or neoadjuvant therapy or investigator or patient preference. Crossover was not permitted between the two treatment groups. Patients were stratified according to the presence or absence of liver or lung metastases, previous chemotherapy for advanced disease and endocrine therapy. The Primary end point was Progression Free Survival (PFS) and Secondary endpoint included Overall Survival (OS). The superior PFS data with KISQALI® compared to endocrine therapy alone, was previously reported. The authors herein report the results on Overall Survival.
After a median follow up of 34.6 months, the addition of KISQALI® to endocrine therapy resulted in significantly longer Overall Survival, compared to endocrine therapy alone. The estimated OS at 42 months was 70.2% in the KISQALI® group and 46.0% in the placebo group (HR for death=0.71; P=0.00973), suggesting a 29% reduction in the risk of death. No new safety signals were observed and in the KISQALI® group, more instances of QT-interval prolongation were observed in patients who received Tamoxifen than in those who received an Aromatase Inhibitor, but without symptomatic arrhythmias or Torsades de pointes.
It was concluded that KISQALI® along with endocrine therapy significantly prolonged Overall Survival, compared to endocrine therapy alone, among pre and perimenopausal patients with advanced HR-positive, HER2-negative breast cancer and these findings represent a major treatment advance in this patient group. Overall Survival with Ribociclib plus Endocrine Therapy in Breast Cancer. Im S-A, Lu Y-S, Bardia A, et al. N Engl J Med 2019; 381:307-316.
US Preventive Services Task Force Recommendations on BRCA Mutation Testing
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. About 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Breast cancer is the second leading cause of cancer death in the US. DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. These mutations can be inherited from either of the parents in an autosomal dominant pattern and a child has a 50 percent chance of inheriting this mutation and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal.
It is estimated that BRCA1/2 gene mutations occur in approximately 1 in 400 women in the general population and account for 5-10% of breast cancer cases and 15% of ovarian cancer cases. The estimated prevalence of potentially harmful BRCA1/2 mutations is 6% in women with cancer onset before age 40 years, and 2.1% among Ashkenazi Jewish women. Among individuals with a family history of breast or ovarian cancer, BRCA1 mutation prevalence is approximately 13%, BRCA2 mutation prevalence is about 8%, and prevalence of either mutation is about 20%. Mutations in the BRCA1/2 genes increase breast cancer risk by 45-65% by age 70 years. The risk of ovarian, fallopian tube, or peritoneal cancer, increases to 39% for BRCA1 mutations, and 10-17% for BRCA2 mutations.
The US Preventive Services Task Force (USPSTF) in this publication updated the 2013 recommendations on risk assessment, genetic counseling, and genetic testing for BRCA-related cancer. These recommendations are based on the evidence of both the benefits and harms of the service and this assessment does not consider the costs of providing a service.
The USPSTF reviewed the evidence on risk assessment, genetic counseling, and genetic testing for potentially harmful BRCA1/2 mutations in asymptomatic women who have never been diagnosed with BRCA-related cancer, as well as those with a previous diagnosis of breast, ovarian, tubal, or peritoneal cancer who have completed treatment and are considered cancer free. In addition, the USPSTF reviewed interventions to reduce the risk for breast, ovarian, tubal, or peritoneal cancer in women with potentially harmful BRCA1/2 mutations, including intensive cancer screening, medications, and risk-reducing surgery.
Recommendations
1) The USPSTF recommended that primary care clinicians assess women with a personal or family history of breast, ovarian, tubal, or peritoneal cancer or who have an ancestry associated with BRCA1/2 gene mutations, with an appropriate brief familial risk assessment tool. Women with a positive result on the risk assessment tool should receive genetic counseling and, if indicated after counseling, genetic testing.
2) The USPSTF recommends against routine risk assessment, genetic counseling, or genetic testing for women whose personal or family history or ancestry is not associated with potentially harmful BRCA1/2 gene mutations.
Risk Assessment
Clinicians should obtain medical and family history specifically inquiring patients about specific types of cancer, primary cancer sites, which family members were affected, whether relatives had multiple types of primary cancer, the age at diagnosis, age at death, and sex of affected family members, both immediate (parents and siblings) as well as more distant (aunts, uncles, grandparents, and cousins). Women who have a family or personal history of breast, ovarian, tubal, or peritoneal cancer should be evaluated by clinicians, to determine the need for in-depth genetic counseling, using appropriate familial risk assessment tools, recognized by the USPSTF. Familial risk assessment factors include breast cancer diagnosis before age 50 years, bilateral breast cancer, presence of both breast and ovarian cancer in one individual, male family members with breast cancer, multiple cases of breast cancer in the family, one or more family members with 2 primary types of BRCA-related cancer (such as ovarian cancer), and Ashkenazi Jewish ancestry. Breast cancer risk assessment models such as the National Cancer Institute Breast Cancer Risk Assessment Tool, which is based on the Gail model are not designed to identify BRCA-related cancer risk and should not be used for this purpose.
Genetic Counseling
Genetic counseling includes detailed kindred analysis and risk assessment for potentially harmful BRCA1/2mutations, identification of individuals for testing, discussion of the benefits and harms of genetic testing, interpretation of results after testing, and discussion of management options. Genetic counseling should be performed by trained health professionals, including suitably trained primary care clinicians.
Genetic Testing
Testing for BRCA1/2 mutations should be performed only when an individual with history suggesting inherited cancer susceptibility, is willing to talk with a qualified health professional, trained to provide genetic counseling and interpret test results, and when test results will aid in decision-making. BRCA mutation testing should begin with a relative with known BRCA-related cancer, including male relatives, to determine if a clinically significant mutation is detected in the family, before testing individuals without cancer. If an affected family member with a BRCA-related cancer is not available, then the relative with the highest probability of mutation should be tested. Reporting BRCA1/2 mutations identified by genetic tests should include a 5-tier terminology system, using the terms “pathogenic,” “likely pathogenic,” “uncertain significance,” “likely benign,” and “benign.
Treatment and Interventions for women with harmful BRCA1/2 mutations
The USPSTF recommends that clinicians offer intensive screening and risk-reducing medications such as Tamoxifen, Raloxifene, or Aromatase Inhibitors to women at increased risk for breast cancer and at low risk for adverse medication effects. General care for these individuals may include risk-reducing mastectomy and salpingo-oophorectomy.
US Preventive Services Task Force. Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2019;322:652-665.
Adjuvant Treatment with PROLIA® Improves Disease Free Survival in Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. About 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. In postmenopausal women with hormone receptor-positive, early-stage breast cancer, treatment with adjuvant Aromatase Inhibitors is the standard of care.
ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors that bind reversibly to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. Aromatase Inhibitors however are associated with accelerated bone loss, leading to a decrease in Bone Mineral Density (BMD) and can thus cause osteopenia and osteoporosis, thereby increasing fracture risk.
PROLIA® (Denosumab) is a monoclonal antibody that inhibits osteoclast formation, function and survival, by selectively targeting the RANK ligand. The RANK-RANK ligand system is an important mediator of signaling in the genesis of osteoclasts and bone resorption. Additionally, the RANK-RANK ligand system has been implicated in antitumor immunity and may have a role in suppressing tumor agenesis. It has been hypothesized that targeting RANK with anti-RANK inhibitor might reverse the immunosuppressive effect of the RANK-RANK ligand signaling pathway.
ABCSG-18 is a randomized, double-blind, placebo controlled, Phase III trial in which the authors evaluated the benefits of the anti-RANK ligand antibody PROLIA® on bone health, in postmenopausal patients, with early stage hormone receptor-positive breast cancer, treated with Aromatase Inhibitors. Of the 3425 patients enrolled in this study, 3420 patients were randomly assigned 1:1 to receive PROLIA® 60 mg (N=1711) or matching placebo (N=1709), subcutaneously every 6 months, during Aromatase Inhibitor therapy. Majority of the patients participating in this study had breast cancer with good prognosis and only 25% of the patients required adjuvant chemotherapy. Patient received a median of 7 doses of PROLIA®. The Primary endpoint was the time to first clinical fracture after randomization. The Secondary endpoint was Disease Free Survival (defined as time from randomization to first evidence of local or distant metastasis, contralateral breast cancer, secondary carcinoma, or death from any cause) in the intention-to-treat population. The Primary endpoint of the ABCSG-18 trial was met (The Lancet 2015;386:433-443) and the use of PROLIA® as an adjuvant to Aromatase Inhibitor therapy significantly delayed time to first clinical fracture, compared to Placebo (HR=0.50; P<0.0001). The authors in publication reported the Secondary endpoint of Disease Free Survival (DFS) outcomes from this study.
After a median follow-up of 73 months, there was a significant improvement in the Disease Free Survival among patients in the PROLIA® group compared to the placebo group (HR=0•82; P=0.026). In the PROLIA® group, the DFS was 89.2% at 5 years and 80.6% at 8 years of follow-up, compared with 87.3% at 5 years and 77.5% at 8 years in the placebo group. The total number of adverse events was similar in the PROLIA® and placebo groups and there were no reported cases of osteonecrosis of the jaw bone or atypical femoral fractures.
It was concluded that PROLIA® constitutes an effective and safe adjuvant treatment for patients with postmenopausal hormone receptor-positive early breast cancer receiving Aromatase Inhibitor therapy, with a Disease Free Survival benefit similar to bisphophonates, but with a better toxicity profile and convenient subcutaneous injections twice yearly. Adjuvant denosumab in postmenopausal patients with hormone receptor-positive breast cancer (ABCSG-18): disease-free survival results from a randomised, double-blind, placebo-controlled, phase 3 trial. Gnant M, Pfeiler G, Steger GG, et al. Lancet Oncol 2019;20:339-351
Clinical and Genomic Risk to Guide the Use of Adjuvant Therapy for Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. About 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Approximately 50% of all breast cancers are Estrogen Receptor (ER) positive, HER2-negative, axillary node-negative tumors. Patients with early stage breast cancer often receive adjuvant chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for Hormone Receptor positive, HER negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
Oncotype Dx assay categorizes patients on the basis of Recurrence Scores into Low risk (less than 18), Intermediate risk (18-30), and High risk (31 or more). It has been unclear whether patients in the Intermediate risk group benefited from the addition of chemotherapy to endocrine therapy. TAILORx was specifically designed to address this question and provide a very definitive answer. In this study, the Intermediate risk Recurrent Score (18-30) was changed to 11-25, to account for exclusion of higher-risk patients with HER2-positive disease and to minimize the potential for under treatment.
TAILORx ((Trial Assigning Individualized Options for Treatment) is a phase III, randomized, prospective, non-inferiority trial, and is the largest breast cancer treatment trial ever conducted, and the first precision medicine trial ever done, according to the authors. In this study, 10,273 women, 18-75 years of age, with hormone receptor-positive, HER2-negative, axillary node-negative breast cancer were enrolled. Patients had tumors 1.1-5.0 cm in size (or 0.6-1.0 cm and intermediate/high grade). Patients were divided into three groups based on their Recurrence Score. Women with a Low Recurrence Score of 0-10 received endocrine therapy alone and those with a High Recurrence Score of 26-100 received endocrine therapy in combination with standard adjuvant chemotherapy. Patient with Intermediate Recurrence Score of 11-25 (N=6711) were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. Patients were followed up for 9 years. The Primary endpoint was invasive Disease Free Survival, defined as recurrence of cancer in the breast, regional lymph nodes, and/or distant organs, a second primary cancer in the opposite breast or another organ, or death from any cause. Results reported in June 2018 showed that while most women with an Intermediate Recurrence Score of 11-25 did not benefit from chemotherapy, women 50 years or younger with a Recurrence Score of 16-25 did indeed benefit from adjuvant chemotherapy.
The authors in this publication provided additional results from the same data set showing that adding “Clinical Risk” provides additional prognostic information. The investigators used a binary classification system from the MINDACT trial (Microarray in Node-Negative Disease May Avoid Chemotherapy), which used a 70-gene assay, and divided patients into high or low “Clinical Risk” based on tumor size and histologic grade. Clinical Risk was defined as low if the tumor was 3 cm or less in diameter and had a low histologic grade, 2 cm or less and had an intermediate histologic grade, or 1 cm or less in diameter and had a high grade. The Clinical Risk was defined as high if the low-risk criteria were not met. This additional reporting provided prognostic information about recurrent risk, but not benefits of chemotherapy particularly in the Intermediate Recurrence Score group.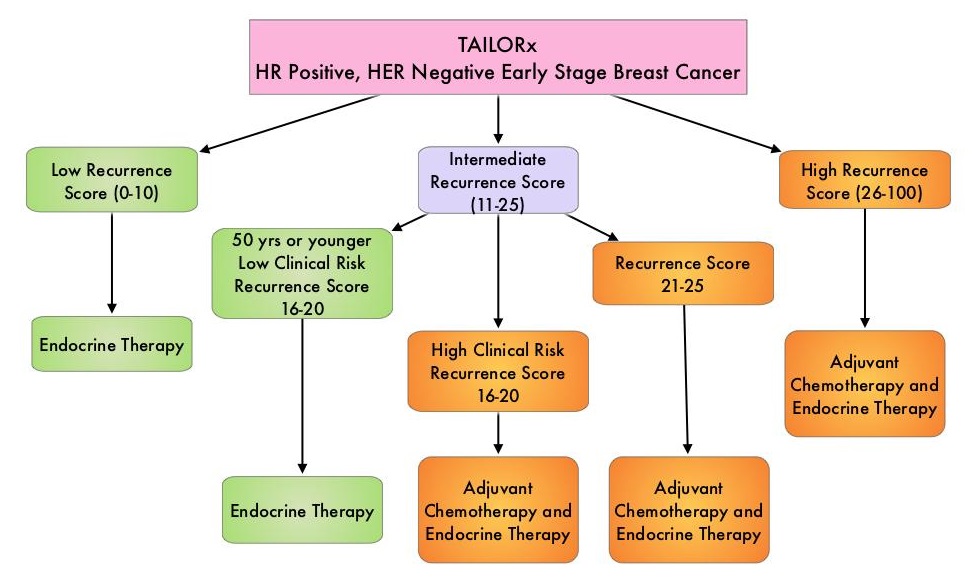
Among women who were 50 years of age or younger with a low Recurrence Score, the distant recurrence rate at 9 years was less than 5%, irrespective of Clinical Risk, and about 5% among those with an intermediate Recurrence Score with low Clinical Risk. However in sharp contrast, among women 50 years of age or younger, with high Clinical Risk and an intermediate Recurrence Score who had received endocrine therapy alone, the rate of distant recurrence at 9 years was 12.3%, compared with 6.1% among women who had received adjuvant chemotherapy. It is possible that younger women with a Recurrence Score of 11-25 and high Clinical Risk, receiving endocrine therapy alone, may have been undertreated with Tamoxifen, and the authors speculated that based on previously published studies, adding Ovarian Suppression and an Aromatase Inhibitor might result in risk reduction, equivalent to that observed using adjuvant chemotherapy.
The authors concluded that these new findings complement the original, definitive TAILORx conclusion and integration of genomic (Recurrence Score) and Clinical Risk may provide a more accurate estimate of prognosis for individual patients, than could be provided by either the genomic or clinical information alone. They added that this Clinical Risk stratification facilitates more refined estimates of absolute chemotherapy benefit for women 50 years of age or younger, with a Recurrence Score 16-25. Clinical and Genomic Risk to Guide the Use of Adjuvant Therapy for Breast Cancer. Sparano JA, Gray RJ, Ravdin PM, et al. N Engl J Med 2019;380:2395-2405
Prognostic Value of Progesterone Receptor in Estrogen Receptor‐Positive, HER‐2 Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. About 70% of breast tumors express Estrogen Receptors (ER) and/or Progesterone Receptors (PR) and these patients are often treated with anti-estrogen therapy.
It has been well established that the detection of Estrogen, Progesterone and HER-2 neu receptors on the tumor cell expressed either alone or together, is a significant prognostic factor, and is associated with the conditional gene expression in the tumor cell itself. The St. Gallen International Expert Consensus in 2011 classified breast cancer into five molecular subtypes with prognostic significance, based on these genetically determined expressions in the tumor cell. The five molecular subtypes of breast cancer include 1) Luminal A (ER+ and/or PR+, HER-2 negative, Ki-67 less than 14%) constituting about 26% of patients 2) Luminal B with HER-2 negative (ER+ and/or PR+, HER-2 negative, Ki-67 14% or more) constituting about 32% 3) Luminal B with HER-2 positive (ER+ and/or PR+, HER-2+, any Ki-67) constituting about 25% 4) HER-2 enriched (ER-, PR-, HER-2+) accounting for about 8% of patients 5) Basal-like (triple negative) (ER-, PR-, HER-2 negative, CK5/6+ and/or EGFR+) constituting about 9% of all breast cancer patients. According to the 2015 St. Gallen guidelines, adjuvant chemotherapy can avoided in Luminal A patients, whereas patients with ER+ HER‐2 negative tumors with low or no PR expression or with high Ki‐67 expression are considered to have a high risk of relapse and adjuvant chemotherapy is often recommended. It should be noted however that low PR expression in Luminal A patients can carry a poor prognosis as well.
The Progesterone Receptor (PR) is a member of the nuclear receptor family and is regulated by the Estrogen Receptor. It is expressed in over two-thirds of ER-positive breast cancers and is more highly expressed in the Luminal A breast cancer subtype. Several studies have demonstrated improved prognosis among PR-positive breast cancer patients. Further, in ER+, HER‐2 negative breast cancers, the Progesterone Receptor is an independent prognostic marker. However, the prognostic value of PR by tumor grade has been unclear.
The authors hypothesized that patient with high‐grade tumors might do well if PR is positive. The objective of this study therefore was to study the prognostic value of PR in tumors with high tumor proliferative index, using tumor grade as a surrogate for the proliferative activity of ER+ HER‐2 negative breast cancer. This retrospective study included women with primary operable, invasive, ER+ HER‐2 negative breast cancer, diagnosed between 2000 and 2012, treated at University Hospitals Leuven. The association of PR status and subtype (Grade 1-2 versus Grade 3) was assessed with Distant Recurrence‐Free Interval (DRFI) and Breast Cancer‐specific survival. The data set from BIG 1‐98 trial was used for validation. A total of 4,228 patients from Leuven and 5,419 from BIG 1‐98 were analyzed. The median patient age was 58 years, 73% had Luminal A‐like subtype, 27% had Luminal B‐like subtype, lymph node status was predominantly negative (62%) and PR was positive in 89% of patients. Median tumor size was 2.1 cm and most of the patients received adjuvant endocrine therapy (96%) and/or radiotherapy (87%). Adjuvant chemotherapy was administered to 29% of the patients. The median follow up period was 8.6 years.
It was noted in this study that PR positive tumors were more strongly associated with better prognosis in Luminal B‐like than in luminal A‐like breast cancer, suggesting that PR is more prognostic in Luminal B‐like versus Luminal A‐like tumors. Among the Luminal B‐like tumors, the 5‐year cumulative incidences of distant recurrence were 18.7% in PR negative and 9.2% in PR positive tumors.
It was concluded PR positivity may be more protective against metastatic relapse in Luminal B‐like versus Luminal A‐like breast cancer. An absent PR is a clinically more important negative prognostic factor in tumors with a high proliferative index than in low proliferative ER+ HER‐2 negative breast tumors. Prognostic Value of the Progesterone Receptor by Subtype in Patients with Estrogen Receptor‐Positive, HER‐2 Negative Breast Cancer. Van Astena K, Slembroucka L, Olbrecht S, et al. The Oncologist 2019;24:165-171
FDA Approves First PI3K Inhibitor, PIQRAY®, for Breast Cancer
SUMMARY: The FDA on May 24, 2019 approved PIQRAY® (Alpelisib) in combination with FASLODEX® (Fulvestrant), to treat postmenopausal women and men, with Hormone Receptor (HR) positive, Human Epidermal growth factor Receptor 2 (HER2) negative, PIK3CA-mutated, advanced or metastatic breast cancer (as detected by an FDA-approved test), following progression on or after an endocrine-based regimen. The FDA also approved the companion diagnostic test, therascreen PIK3CA RGQ PCR Kit, to detect the PIK3CA mutation in a tissue and/or a liquid biopsy. Patients who are negative by the therascreen test using the liquid biopsy should undergo tumor biopsy for PIK3CA mutation testing. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.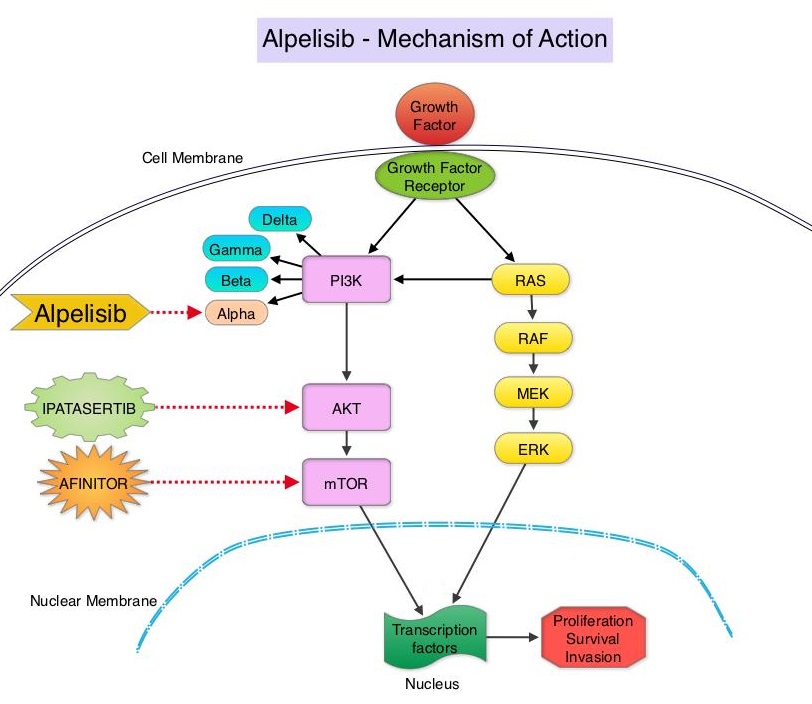
The PhosphoInositide 3-Kinase (PI3K) pathway is an intracellular signaling pathway important in the regulation of cancer cell proliferation and metastasis. PI3K is a lipid kinase and has four distinct isoforms - alpha, beta, gamma and delta, which play a unique role in the survival of different tumor types and establishment of supportive tumor microenvironments. The alpha and beta isoforms are expressed in a wide variety of tissues whereas the gamma and delta isoforms are primarily expressed in hematopoietic cells such as B and T cells. The PI3K alpha isoform is particularly important in breast cancer and plays an important role in tumorigenesis, supporting tumor angiogenesis and stromal interactions, making this a viable target. PIK3CA is an oncogene that codes for the alpha isoform of PI3K, (PI3Kα), more specifically for the alpha isoform of p110. The PI3k pathway is the most frequently altered pathway in human cancers including breast cancer, and has been implicated in disease progression in a significant number of patients with breast cancer. Activation of the PI3K pathway in breast cancer has been associated with resistance to endocrine therapy and disease progression. Approximately 40% of patients with Hormone Receptor positive (HR+), HER2 negative breast cancers, harbor activating mutations in the PIK3CA isoform of PI3K, which is the most common mutation in HR+ breast cancer. Patients with advanced breast cancer harboring PIK3CA mutations typically have a poor prognosis. This provides a strong rationale for targeting the PI3K pathway in breast cancer.
PIQRAY® is an oral, alpha-specific PI3K inhibitor that specifically inhibits PIK3 in the PI3K/AKT kinase signaling pathway. Further, it was shown in preclinical studies that cancer cells with PIK3CA mutations are more sensitive to PIQRAY® than those without the mutation, across a broad range of tumor types. SOLAR-1 clinical trial, which was conducted to test this hypothesis, became the basis for this FDA approval.
SOLAR-1 is a global, double-blind, placebo-controlled, randomized phase III trial, which studied the benefit of PIQRAY® in combination with FASLODEX® (Fulvestrant) among postmenopausal women and men with PIK3CA-mutated HR+/HER2 negative advanced or metastatic breast cancer, who had progressed on or following prior Aromatase Inhibitor (AI) treatment with or without a Cyclin-Dependent Kinase (CDK) 4/6 inhibitor. In this study, 572 patients were randomized in a 1:1 ratio to receive PIQRAY® 300 mg orally daily or placebo once daily, in combination with FASLODEX® 500mg IM on days 1 and 15 of the first cycle and day 1 of each subsequent 28-day cycle. Patients were stratified based on visceral metastases and prior CDK4/6 inhibitor treatment. A total of 341 patients had PIK3CA mutations upon testing of the tumor tissue with 169 patients receiving the PIQRAY® combination and 172 patients receiving FASLODEX® alone. Enrolled patients had received one or more prior lines of hormonal therapy, but no chemotherapy for advanced breast cancer. They had not previously received FASLODEX® or any PI3K, Akt or mTOR inhibitor, and were not on concurrent anticancer therapy. Approximately half of the patients in each treatment group had lung or liver metastases and 6% had received prior CDK4/6 inhibitor therapy. The Primary endpoint was Progression Free Survival (PFS) for patients with the PIK3CA mutation. Secondary endpoints included Overall Survival (OS), Overall Response Rate (ORR), Clinical Benefit Rate, Health-Related Quality of Life, Efficacy in PIK3CA non-mutant cohort, Safety and Tolerability.
The Primary endpoint was met and at a median follow up of 20 months, the PFS was nearly twice as long in patients with PIK3CA mutations randomized to PIQRAY® plus FASLODEX® compared to the placebo plus FASLODEX® group. The median PFS was 11.0 months in the PIQRAY® group compared to 5.7 months in the placebo group (HR=0.65; P=0.00065). In patients with measurable, PIK3CA-mutated advanced breast cancer (N=262), the Overall Response Rate was 36% for the PIQRAY® plus FASLODEX® group versus 16% for placebo plus FASLODEX® group (P=0.0002). There was no significant PFS benefit noted in the PIK3CA-nonmutant patient group receiving PIQRAY® plus FASLODEX® The most frequent toxicities with PIQRAY® were hyperglycemia which could be managed with Metformin, nausea, decreased appetite and skin rash.
It was concluded that PIQRAY® given along with FASLODEX® significantly improved Progression Free Survival compared to Placebo plus FASLODEX® with manageable toxicities. The authors commented that this is the first study to show statistically significant, clinically meaningful PFS improvement with an alpha-specific PI3K inhibitor in PIK3CA-mutated HR+, HER2 negative advanced breast cancer, highlighting the importance of clinical genomics in advanced breast cancer. PIQRAY® is also the first novel drug approved under the Real-Time Oncology Review pilot program. It however remains unclear whether PIQRAY® should be incorporated into the current treatment paradigm upfront, along with endocrine therapy and a CDK 4/6 inhibitor, or sequentially following disease progression on a combination of endocrine therapy and a CDK 4/6 inhibitor. Alpelisib (ALP) + fulvestrant (FUL) for advanced breast cancer (ABC): results of the phase 3 SOLAR-1 trial. André F, Ciruelos EM, Rubovszky G, et al. Presented during the Presidential Symposium 1 at: 2018 ESMO Congress; October 19-23; Munich, Germany. Abstract LBA3_PR.
FDA Approves KADCYLA® for Early Breast Cancer
SUMMARY: The FDA on May 3, 2019, approved KADCYLA® (Ado-Trastuzumab Emtansine) for the adjuvant treatment of patients with HER2-positive early breast cancer, who have residual invasive disease after neoadjuvant Taxane and HERCEPTIN® (Trastuzumab)-based treatment. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of invasive breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of HERCEPTIN®, several other HER2-targeted therapies have become available. The duration of adjuvant HERCEPTIN® therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials. 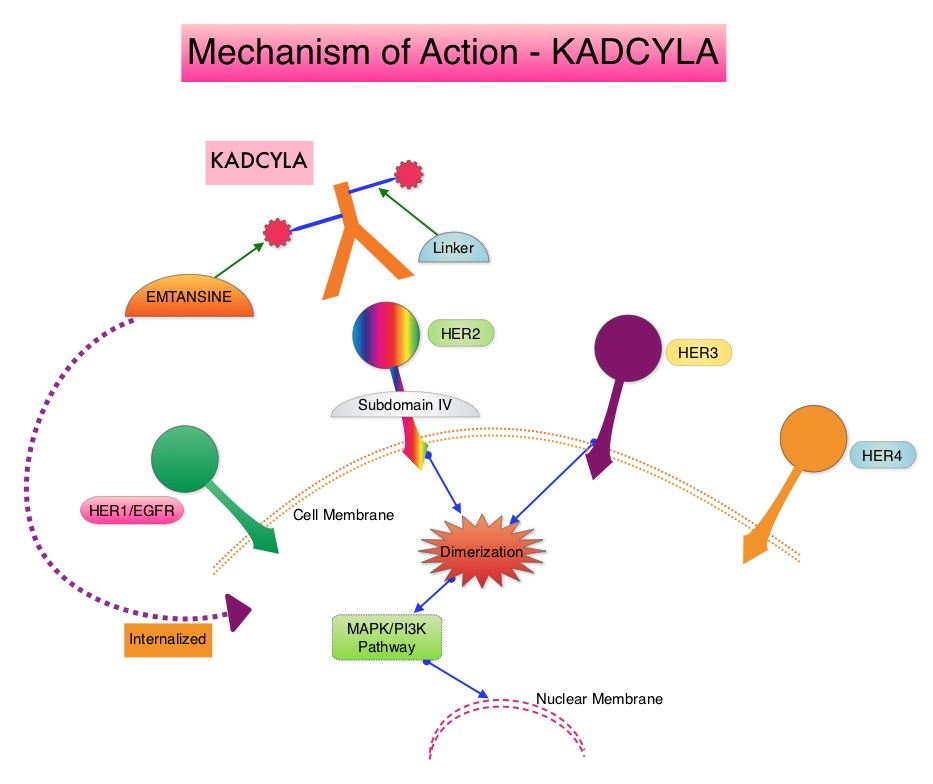
KADCYLA® is an Antibody-Drug Conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release. In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® (Lapatinib) plus XELODA® (Capecitabine), in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a Taxane.
It is well established that patients with HER2-positive early breast cancer following HERCEPTIN® based neoadjuvant therapies have a pathological Complete Response rate of 40-60%. Those without a pathological Complete Response tend to have significantly less favorable outcomes. These patients irrespective of pathological response status complete their standard adjuvant therapy which includes 12 months of HER2-targeted therapy. KATHERINE trial was conducted to evaluate the benefit of switching from standard HER2-directed therapy to single-agent KADCYLA®, after neoadjuvant chemotherapy along with either single or dual HER2 targeted therapy, in patients with residual invasive cancer at surgery. This study was conducted to address the unmet need of patients who have residual invasive breast cancer after receiving neoadjuvant chemotherapy plus HER2-targeted therapy.
The KATHERINE trial is an open-label, phase III global study, which compared KADCYLA® with HERCEPTIN®, as an adjuvant treatment for patients with HER2-positive early breast cancer, who had residual invasive disease following neoadjuvant chemotherapy and HERCEPTIN®. This study included 1,486 patients with HER2-positive early stage breast cancer, who were found to have residual invasive disease in the breast or axillary lymph nodes at surgery, following at least six cycles (16 weeks) of neoadjuvant chemotherapy with a Taxane (with or without Anthracycline) and HERCEPTIN®. Within 12 weeks of surgery, patients (N=1486) were randomly assigned in a 1:1 ratio to KADCYLA® 3.6 mg/kg IV every 3 weeks or HERCEPTIN® 6 mg/kg IV every 3 weeks, for 14 cycles (743 patients in each group). Both treatment groups were well balanced and hormone receptor positive disease was present in 72% of the patients. The majority of the patients (77%) had received an Anthracycline-containing neoadjuvant chemotherapy regimen, and in 19% of the patients, another HER2-targeted agent in addition to HERCEPTIN® (dual HER2 blockade) had been administered as a component of neoadjuvant therapy. The Primary end point was invasive Disease Free Survival (defined as freedom from ipsilateral invasive breast tumor recurrence, ipsilateral locoregional invasive breast cancer recurrence, contralateral invasive breast cancer, distant recurrence, or death from any cause). The median duration of follow up was 41.4 months in the KADCYLA® group and 40.9 months in the HERCEPTIN® group.
At the prespecified interim analysis, invasive disease occurred in 12.2% of patients who received KADCYLA® and 22.2% of patients who received HERCEPTIN®. The estimated percentage of patients who were free of invasive disease at 3 years was 88.3% in the KADCYLA® group and 77.0% in the HERCEPTIN® group. Invasive Disease Free Survival, which was the Primary end point of the study, was significantly higher in the KADCYLA® group than in the HERCEPTIN® group (HR=0.50; P<0.001).This suggested that KADCYLA® reduced the risk of developing an invasive breast cancer recurrence or death by 50%. Distant recurrence as the first invasive disease event occurred in 10.5% of patients in the KADCYLA® group and in 15.9% of the HERCEPTIN® group. A consistent benefit was seen across all prespecified subgroups. Adverse events were consistent with the known safety profile of KADCYLA®, with more toxicities associated with KADCYLA® than with HERCEPTIN®. Additional follow-up will be necessary to determine the Overall Survival benefit with adjuvant KADCYLA®.
It was concluded that among patients with HER2-positive early breast cancer who had residual invasive disease after completion of neoadjuvant therapy, substituting KADCYLA® for adjuvant HERCEPTIN® reduced the risk of recurrence of invasive breast cancer or death by 50%, with the benefit seen across all patient subgroups. The authors added that even though KATHERINE trial focused on higher-risk patients with residual invasive breast cancer after completion of neoadjuvant chemotherapy, CNS recurrence remains a persistent problem. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. von Minckwitz G, Huang C-S, Mano MS, et al. for the KATHERINE Investigators. N Engl J Med 2019;380:617-628
Six Prognostic Factors That Predict Invasive Breast Cancer Recurrence after DCIS
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Carcinoma in situ of the breast also known as Ductal Carcinoma In Situ (DCIS) is defined as a malignant proliferation of ductal epithelial cells that are confined to the milk ducts without invasion of the basement membrane, and is a precursor lesion to invasive carcinoma. DCIS accounts for approximately 25% of all newly diagnosed breast cancers. Patients with small, screening-detected lesions, are often treated with breast-conserving surgery (to prevent the development of invasive breast cancer), followed by adjuvant radiation and hormonal therapy, although neither of the latter two interventions have been shown to improve survival outcomes. As such, a significant number of patients are overtreated. There remains a large unmet need, to distinguish relatively benign DCIS from DCIS that will develop into invasive breast cancer.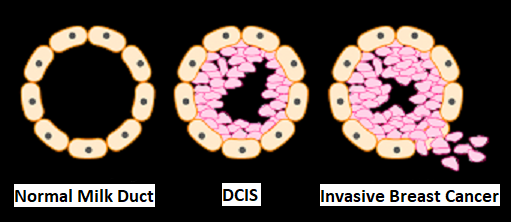
The authors in this study performed a systematic review from 1970 to 2018, with meta-analyses of 1,781 articles from the PubMed database, to summarize current knowledge on prognostic factors for invasive disease, after a diagnosis of DCIS. The number of patients in the included studies ranged from 52 to 37,692. Of all the articles reviewed, 40 articles met the inclusion criteria. Eligible studies assessed risk of invasive recurrence in women primarily diagnosed and treated for DCIS, and included at least 10 ipsilateral-invasive breast cancer events and 1 year of follow up. The mean follow up time ranged from 3.2 to 15.8 years. Quality in Prognosis Studies (QUIPS) tool was used for risk-of-bias assessment (A working group comprising epidemiologists, statisticians, and clinicians developed this tool based on previous research and this tool can inform judgements of risk-of-bias in prognostic research). Meta-analyses were performed to estimate the average effect size of the prognostic factors.
The researchers identified six prognostic factors in the meta-analyses that were statistically significant and were associated with a 36% to 84% increase in the relative risk of recurrence of invasive disease after a DCIS diagnosis. These six factors included-
1) African American race (43% higher risk)
2) Premenopausal status (59% higher risk)
3) Detection by palpation (84% higher risk)
4) Positive margins (63% higher risk)
5) High histologic grade (36% higher risk)
6) High p16 expression (51% higher risk).
Further, the authors identified frequently occurring biases in studies on invasive recurrence after DCIS and the highest risk of bias was attributable to insufficient handling of confounders and poorly described study groups. They added that avoiding these common methodological pitfalls can improve future study designs.
It was concluded that this study results may help clinicians distinguish indolent from potentially aggressive DCIS and prevent overtreatment.
Predictors of an Invasive Breast Cancer Recurrence after DCIS: A Systematic Review and Meta-analyses. Visser LL, Groen EJ, van Leeuwen FE, et al. Cancer Epidemiol Biomarkers Prev. 2019;28:835-845
High Body Fat Level Increases Breast Cancer Risk in Postmenopausal Women with Normal BMI
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Obesity is an important contributing factor to postmenopausal breast cancer incidence and mortality. Based on recently published meta-analysis, in women diagnosed with breast cancer, there is an approximately 30% increased risk of disease recurrence or death in those who are obese, compared to those with ideal body weight. Increasing physical activity may lower the risk of breast cancer recurrence. According to the consensus from the St Gallen Consensus Conference in 2015, obesity has been associated with poor breast cancer outcomes. Obesity is associated with alterations in Insulin/glucose homeostasis, adipokines, and sex hormones, which may all play a role in breast cancer outcomes. Weight loss can lead to reductions in C-reactive protein, Insulin, glucose, and Leptin. These mediators have all been implicated to have prognostic significance in breast cancer.
Body Mass Index (BMI) measures body size and is calculated based on height and weight. It is used as a measure of obesity. BMI however does not discriminate between adiposity and muscle and individuals deemed healthy based on a normal BMI may still be prone to cardiometabolic disorders due to high levels of visceral fat. Dual-energy X-ray absorptiometry (DXA or DEXA) is often utilized to measure bone mineral density and is the most accurate method presently available for the diagnosis of osteoporosis and to estimate fracture risk. DEXA scan can also be used to measure total body composition and fat content including the amount of visceral fat, with a high degree of accuracy by its ability to break down fat, bone and muscle tissue. It has been reported that approximately 18% of women with normal BMI had excess fat, detected on DEXA scan.
There are two types of adipose tissue in the human body, White Adipose Tissue (WAT) and Brown Adipose Tissue (BAT) which have antagonistic functions. White Adipose Tissue or white fat cells represent the body’s main type of fat tissue and each fat cell has a single lipid droplet. They are distributed in the subcutaneous tissue, around a person's waist and thighs and around internal organs (visceral fat). WAT stores excess energy as triglycerides and serves as an energy reservoir. Brown Adipose tissue (BAT) which is abundant in small mammals and in newborns generates heat by burning calories and helps them to survive cold temperatures. Brown adipocytes contain several small lipid droplets, and a high number of iron-containing mitochondria which gives brown fat its dark tan color. Most BAT is distributed in the lower neck and interscapular area of an adult, and above the collarbone. Higher quantities of BAT are associated with lower body weight and BAT decreases and body weight increases with increasing age.
Leptin is a hormone produced primarily by adipose tissue and circulating Leptin levels correlate with the body fat stores, with increased circulating Leptin levels noted in individuals with excess adiposity. Leptin can induce Aromatase which synthesizes estrogen, can directly stimulate cancer cell proliferation and survival, and activate Estrogen Receptor α via ligand-independent mechanism.
The increased risk of postmenopausal breast cancer in women with normal BMI is poorly understood. Recent studies have shown that in these women with normal BMI, excess body fat is associated with adipocyte hypertrophy which correlates with WAT inflammation, increased circulating Leptin levels, elevated levels of Aromatase and elevated Insulin levels. Dysregulation of Insulin signaling can activate the PI3K/Akt/mTOR and Ras/Raf/MAPK pathways which in turn can enhance cell proliferation and increase the risk of breast cancer. Further, Insulin also induces insulin like growth factor-1 (IGF-1), which can activate ERα. Insulin resistance leads to reduced levels of sex hormone–binding globulin, resulting in elevated levels of free estradiol. It has been suggested that all of these changes collectively may play a role in the pathogenesis of obesity-related breast cancer. The present study was conducted to investigate the association between body fat and breast cancer risk in women with normal BMI.
The authors in this long-term prospective study examined the association between body fat mass, measured by DEXA scan, and the risk of breast cancer, in a secondary analysis of 3460 postmenopausal women with normal BMI (18.5-24.9), enrolled in the Women’s Health Initiative (WHI) clinical trials or observational study. The goal of this study was to understand whether excess adipose tissue is associated with an increased breast cancer risk in women with normal BMI. Participants 50-79 years old with a mean age of 64 years underwent body fat measurement (the percentage of whole-body fat, trunk fat, and fat mass in both legs) with DEXA scan at 3 US designated centers at the time of study entry into the WHI clinical trials, and years 1, 3, 6, and 9. Levels of Insulin, glucose, C-reactive protein, interleukin-6, triglycerides, HDL cholesterol, estradiol, sex hormone-binding globulin, adiponectin, and Leptin were measured in 3-13% of participants using baseline fasting blood specimens.
At a median follow up of 16 years, 182 incident breast cancers were confirmed, and 146 (80%) were ER positive. It was noted that among postmenopausal women with normal BMI, relatively high body fat levels were associated with an elevated risk of invasive breast cancer. The authors specifically, found a 56% increase in the risk of developing ER-positive breast cancer per 5-kg increase in trunk fat, despite a normal BMI. Elevated trunk fat levels were also associated with metabolic dysregulation and inflammation characterized by increased circulating levels of Insulin, Leptin, C-reactive protein, Interleukin 6 and triglycerides, whereas levels of HDL cholesterol and sex hormone–binding globulin were lower.
It was concluded from this large prospective study that normal BMI may not be an adequate proxy for the risk of breast cancer in postmenopausal women. High body fat levels and altered levels of circulating metabolic and inflammatory factors may be associated with a higher risk of invasive breast cancer. Association of Body Fat and Risk of Breast Cancer in Postmenopausal Women with Normal Body Mass Index. A Secondary Analysis of a Randomized Clinical Trial and Observational Study. Iyengar NM, Arthur R, Manson JE, et al. JAMA Oncol. 2019;5:155-163
FDA Approves TECENTRIQ® and ABRAXANE® Combination for Advanced Triple Negative Breast Cancer
SUMMARY: The FDA on March 8, 2019 granted accelerated approval to TECENTRIQ® (Atezolizumab) in combination with ABRAXANE® (Paclitaxel protein-bound) for adult patients with unresectable locally advanced or metastatic Triple Negative Breast Cancer (TNBC) whose tumors express PD-L1 (PD-L1 stained tumor-infiltrating immune cells [IC] of any intensity covering 1% or more of the tumor area), as determined by an FDA-approved test. The FDA also approved the VENTANA PD-L1 (SP142) Assay as a companion diagnostic device for selecting TNBC patients for TECENTRIQ®. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease.
Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. Those who do not achieve a pathological Complete Response tend to have a poor prognosis. It therefore appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway. Previously published studies have shown that tumor-infiltrating lymphocytes were associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC.
TECENTRIQ® (Atezolizumab) is an anti PD-L1 monoclonal antibody, designed to directly bind to PD-L1 expressed on tumor cells and tumor-infiltrating immune cells, thereby blocking its interactions with PD-1 and B7.1 receptors, and thus enabling the activation of T cells. Single-agent TECENTRIQ® is presently approved for the treatment of metastatic Urothelial carcinoma and Non Small Cell Lung Cancer (NSCLC). TECENTRIQ® has also been shown to have clinical activity with acceptable safety profile in patients with other solid tumors including Triple Negative Breast Cancer. The premise for combining chemotherapy with immune checkpoint inhibitors is that chemotherapy may enhance release of tumor antigens and antitumor responses to immune checkpoint inhibition. Taxanes in particular may additionally activate toll-like receptor activity and promote dendritic-cell activity. ABRAXANE® (Nanoparticle Albumin-Bound - nab Paclitaxel) was selected as a chemotherapy partner in the present study because at the time that this trial was designed, the glucocorticoid premedication that is required with solvent-based Paclitaxel (TAXOL®), had been hypothesized to affect immunotherapy activity.
IMpassion130 is an international, randomized, double-blind, placebo-controlled phase III trial in which first-line treatment with TECENTRIQ® plus ABRAXANE®, was compared with placebo plus ABRAXANE®, in patients with locally advanced or metastatic Triple Negative Breast Cancer. Patients with untreated metastatic Triple Negative Breast Cancer (N=902) were randomly assigned in a 1:1 ratio and received TECENTRIQ® 840 mg IV or placebo on days 1 and 15 and ABRAXANE® 100 mg/m2 IV on days 1, 8, and 15 of every 28-day cycle. Treatment was continued until disease progression or unacceptable toxicity. Stratification factors included presence or absence of liver metastases, use or non-use of neoadjuvant or adjuvant taxane treatment, and PD-L1 expression on tumor-infiltrating immune cells as a percentage of tumor area (less than 1% was considered PD-L1 negative versus1% or more considered PD-L1 positive) according to ImmunoHistochemical testing (IHC). Both treatment groups were well balanced. Approximately 40% of the patients were PD-L1 positive. The two Primary end points were Progression Free Survival (PFS) and Overall Survival (OS).
At a median follow up of 12.9 months, the median PFS in the intent-to-treat population was 7.2 months with TECENTRIQ® plus ABRAXANE®, as compared with 5.5 months with placebo plus ABRAXANE® (HR=0.80; P=0.002). This suggested a 20% improvement in PFS with the TECENTRIQ® combination. At the time of the first interim analysis, the median overall survival was 21.3 months in the TECENTRIQ® plus ABRAXANE® group and 17.6 months in the placebo plus ABRAXANE® group (HR for death=0.84; P=0.08). The Objective Response Rate (ORR) in the intent-to-treat population was 56% in the TECENTRIQ® and ABRAXANE® group versus 45.9% in the placebo plus ABRAXANE® group.
Among patients with PD-L1–positive tumors, the benefit was even more significant. The addition of TECENTRIQ® improved PFS by 38% (HR=0.62; P<0.001) and similar benefit was noted in the OS, with a median OS of 25 months with the TECENTRIQ® combination and 15.5 months with placebo plus ABRAXANE® (HR=0.62). Grade 3 or 4 adverse events were 48.7% in the TECENTRIQ® and ABRAXANE® and 42.2% in the placebo plus ABRAXANE® group.
It was concluded that TECENTRIQ® plus ABRAXANE® significantly prolonged Progression Free Survival among patients with metastatic Triple Negative Breast Cancer in both the intent-to-treat population and the PD-L1 positive subgroup, and could potentially change how we manage patients with Triple Negative Breast Cancer. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. Schmid P, Adams S, Rugo HS, et al. N Engl J Med 2018; 379:2108-2121
Adjuvant KADCYLA® Superior to HERCEPTIN® in High Risk HER2-Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of HERCEPTIN®, several other HER2-targeted therapies have become available. The duration of adjuvant HERCEPTIN® therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials.
KADCYLA® (Ado-Trastuzumab Emtansine, T-DM1) is an Antibody-Drug Conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release. In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® (Lapatinib) plus XELODA® (Capecitabine), in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a Taxane.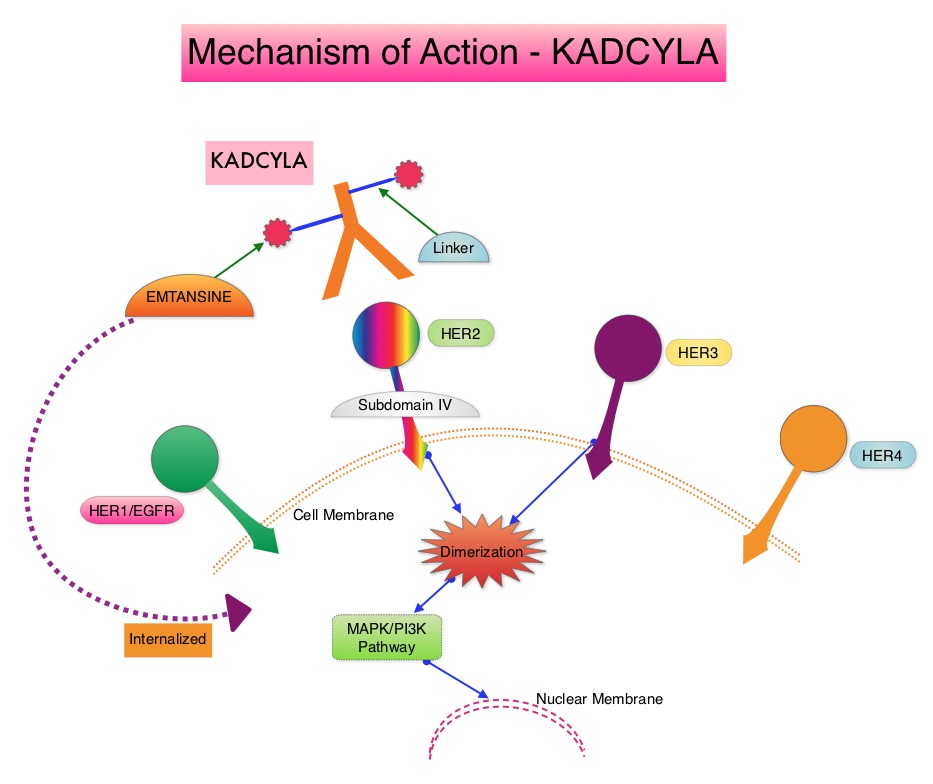
It is well established that patients with HER2-positive early breast cancer following HERCEPTIN® based neoadjuvant therapies have a pathological Complete Response rate of 40-60%. Those without a pathological Complete Response tend to have significantly less favorable outcomes. These patients irrespective of pathological response status complete their standard adjuvant therapy which includes 12 months of HER2-targeted therapy. KATHERINE trial was conducted to evaluate the benefit of switching from standard HER2-directed therapy to single-agent KADCYLA®, after neoadjuvant chemotherapy along with either single or dual HER2 targeted therapy, in patients with residual invasive cancer at surgery. This study was conducted to address the unmet need of patients who have residual invasive breast cancer after receiving neoadjuvant chemotherapy plus HER2-targeted therapy.
The KATHERINE trial is an open-label, phase III global study, which compared KADCYLA® with HERCEPTIN®, as an adjuvant treatment for patients with HER2-positive early breast cancer, who had residual invasive disease following neoadjuvant chemotherapy and HERCEPTIN®. This study included 1,486 patients with HER2-positive early stage breast cancer, who were found to have residual invasive disease in the breast or axillary lymph nodes at surgery, following at least six cycles (16 weeks) of neoadjuvant chemotherapy with a Taxane (with or without Anthracycline) and HERCEPTIN®. Within 12 weeks of surgery, patients (N=1486) were randomly assigned in a 1:1 ratio to KADCYLA® 3.6 mg/kg IV every 3 weeks or HERCEPTIN® 6 mg/kg IV every 3 weeks, for 14 cycles (743 patients in each group). Both treatment groups were well balanced and hormone receptor positive disease was present in 72% of the patients. The majority of the patients (77%) had received an Anthracycline-containing neoadjuvant chemotherapy regimen, and in 19% of the patients, another HER2-targeted agent in addition to HERCEPTIN® (dual HER2 blockade) had been administered as a component of neoadjuvant therapy. The Primary end point was invasive Disease Free Survival (defined as freedom from ipsilateral invasive breast tumor recurrence, ipsilateral locoregional invasive breast cancer recurrence, contralateral invasive breast cancer, distant recurrence, or death from any cause). The median duration of follow up was 41.4 months in the KADCYLA® group and 40.9 months in the HERCEPTIN® group.
At the prespecified interim analysis, invasive disease occurred in 12.2% of patients who received KADCYLA® and 22.2% of patients who received HERCEPTIN®. The estimated percentage of patients who were free of invasive disease at 3 years was 88.3% in the KADCYLA® group and 77.0% in the HERCEPTIN® group. Invasive Disease Free Survival, which was the Primary end point of the study, was significantly higher in the KADCYLA® group than in the HERCEPTIN® group (HR=0.50; P<0.001).This suggested that KADCYLA® reduced the risk of developing an invasive breast cancer recurrence or death by 50%. Distant recurrence as the first invasive disease event occurred in 10.5% of patients in the KADCYLA® group and in 15.9% of the HERCEPTIN® group. A consistent benefit was seen across all prespecified subgroups. Adverse events were consistent with the known safety profile of KADCYLA®, with more toxicities associated with KADCYLA® than with HERCEPTIN®. Additional follow-up will be necessary to determine the Overall Survival benefit with adjuvant KADCYLA®.
It was concluded that among patients with HER2-positive early breast cancer who had residual invasive disease after completion of neoadjuvant therapy, substituting KADCYLA® for adjuvant HERCEPTIN® reduced the risk of recurrence of invasive breast cancer or death by 50%, with the benefit seen across all patient subgroups. The authors added that even though KATHERINE trial focused on higher-risk patients with residual invasive breast cancer after completion of neoadjuvant chemotherapy, CNS recurrence remains a persistent problem. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. von Minckwitz G, Huang C-S, Mano MS, et al. for the KATHERINE Investigators. (published online December 5, 2018). N Engl J Med 2019;380:617-628
Increased Risk of Breast Cancer after Recent Childbirth
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. Breast cancer is the most common cancer type in reproductive-aged women. Women with biological children (parous women) are at a lower risk for developing breast cancer compared to nulliparous women. However, parity as a protective factor largely applies to breast cancer developing after age 60 years and may not apply for younger premenopausal women. Evidence from national registry linkage studies in Scandinavian countries suggested that recent childbirth confers a short-term increase in breast cancer risk which may last for 10 years or more, and this risk may be further increased in women who are older at first childbirth. Studies published thus far have not shown consistent findings and have had limited ability to account for factors influencing breast cancer risk such as breastfeeding and family history of breast cancer.
One biological explanation for an increase in breast cancer risk after childbirth may be due to proliferation of breast cells during pregnancy which could promote accelerated development of latent initiated tumor cells. This may also explain the higher breast cancer risk, conferred by older age at first childbirth, as a result of higher proportion of latent initiated tumor cells at older ages. Further, the postpartum breast microenvironment, characterized by lactational involution may also facilitate cancer cell migration and metastasis. Childbirth also brings about maternal changes beyond breast tissue including altered immune function and microbiota, increased stress, and accelerated aging processes.
The authors in this study used data from the International Premenopausal Breast Cancer Collaborative Group and conducted a pooled analysis of individual-level data from 15 prospective cohort studies. In this analysis, the researchers included women younger than 55 years and evaluated the risk of breast cancer in relation to recent childbirth, while taking into account other factors that relate to breast cancer risk such as breastfeeding, numbers of pregnancies and births and family history of breast cancer. It is felt that understanding these risk patterns may have implications for identifying risk-reducing strategies among vulnerable subgroups. A total of 889, 944 women, were available for analysis after excluding women who reported a first birth before age 13 years, women who were 50 years or older at study entry and at most recent birth, or reached parity greater than 10 births before enrollment. All these events were considered to have greater potential for data errors. The mean age at study entry was 42 years.
The researchers noted that compared with nulliparous women, parous women had an increased risk for breast cancer that peaked about 5 years after childbirth and then gradually decreased about 24 years after childbirth. These findings however were not noted among women who had only 1 child, or had their first child before age 25 years. The risk was highest in women who were older at the time of first childbirth, multiparous women and those who had a family history of breast cancer. Among those women with a family history of breast cancer, the risk was the greatest for Estrogen Receptor negative breast cancer. Breast feeding did not influence breast cancer risk patterns.
It was concluded that compared with nulliparous women, parous women have an increased risk for breast cancer after childbirth that is highest the first 5 years but decreases over the following 20 years. Health Care Professionals should take these factors into account when considering individual risk profiles for breast cancer in premenopausal women. Breast Cancer Risk After Recent Childbirth: A Pooled Analysis of 15 Prospective Studies. Nichols HB, Schoemaker MJ, Cai J, et al. Ann Intern Med. 2019;170:22-30
Late Breaking Abstract - ESMO 2018: Targeting PIK3CA Mutations Improves Outcomes in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 268,600 new cases of female breast cancer will be diagnosed in 2019 and about 41,760 women will die of the disease. About 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.
The PhosphoInositide 3-Kinase (PI3K) pathway is an intracellular signaling pathway important in the regulation of cancer cell proliferation and metastasis. PI3K is a lipid kinase and has four distinct isoforms - alpha, beta, gamma and delta, which play a unique role in the survival of different tumor types and establishment of supportive tumor microenvironments. The alpha and beta isoforms are expressed in a wide variety of tissues whereas the gamma and delta isoforms are primarily expressed in hematopoietic cells such as B and T cells. The PI3K alpha isoform is particularly important in breast cancer and plays an important role in tumorigenesis, supporting tumor angiogenesis and stromal interactions, making this a viable target. PIK3CA is an oncogene that codes for the alpha isoform of PI3K, (PI3Kα), more specifically for the alpha isoform of p110. The PI3k pathway is the most frequently altered pathway in human cancers including breast cancer, and has been implicated in disease progression in a significant number of patients with breast cancer. Activation of the PI3K pathway in breast cancer has been associated with resistance to endocrine therapy and disease progression. Approximately 40% of patients with Hormone Receptor positive (HR+), HER2 negative breast cancers, harbor activating mutations in the PIK3CA isoform of PI3K, which is the most common mutation in HR+ breast cancer. Patients with advanced breast cancer harboring PIK3CA mutations typically have a poor prognosis. This provides a strong rationale for targeting the PI3K pathway in breast cancer.
Alpelisib is an oral, alpha-specific PI3K inhibitor that specifically inhibits PIK3 in the PI3K/AKT kinase signaling pathway. Further, it was shown in preclinical studies that cancer cells with PIK3CA mutations are more sensitive to Alpelisib than those without the mutation, across a broad range of tumor types. SOLAR-1 clinical trial was conducted to test this hypothesis.
SOLAR-1 is a global, double-blind, placebo-controlled, randomized phase III trial, which studied the benefit of Alpelisib in combination with FASLODEX® (Fulvestrant) among postmenopausal women and men with PIK3CA-mutated HR+/HER2 negative advanced or metastatic breast cancer, who had progressed on or following prior Aromatase Inhibitor treatment with or without a Cyclin-Dependent Kinase (CDK) 4/6 inhibitor. In this study, 572 patients were randomized in a 1:1 ratio to receive Alpelisib 300 mg orally daily or placebo once daily, in combination with FASLODEX® 500mg IM on days 1 and 15 of the first cycle and day 1 of each subsequent 28-day cycle. Patients were stratified based on visceral metastases and prior CDK4/6 inhibitor treatment. A total of 341 patients had PIK3CA mutations upon testing of the tumor tissue with 169 patients receiving the Alpelisib combination and 172 patients receiving FASLODEX® alone. Enrolled patients had received one or more prior lines of hormonal therapy, but no chemotherapy for advanced breast cancer. They had not previously received FASLODEX® or any PI3K, Akt or mTOR inhibitor, and were not on concurrent anticancer therapy. Approximately half of the patients in each treatment group had lung or liver metastases and 6% had received prior CDK4/6 inhibitor therapy. The Primary endpoint was Progression Free Survival (PFS) for patients with the PIK3CA mutation. Secondary endpoints included Overall Survival (OS), Overall Response Rate (ORR), Clinical Benefit Rate, Health-Related Quality of Life, Efficacy in PIK3CA non-mutant cohort, Safety and Tolerability.
The Primary endpoint was met and at a median follow up of 20 months, the PFS was nearly twice as long in patients with PIK3CA mutations randomized to Alpelisib plus FASLODEX® compared to the placebo plus FASLODEX® group. The median PFS was 11.0 months in the Alpelisib group compared to 5.7 months in the placebo group (HR=0.65; P=0.00065). In patients with measurable, PIK3CA-mutated advanced breast cancer (N=262), the Overall Response Rate was 36% for the Alpelisib plus FASLODEX® group versus 16% for placebo plus FASLODEX® group (P=0.0002). There was no significant PFS benefit noted in the PIK3CA-nonmutant patient group receiving Alpelisib plus FASLODEX®. The most frequent toxicities with Alpelisib were hyperglycemia which could be managed with Metformin, nausea, decreased appetite and rash.
It was concluded that Alpelisib given along with FASLODEX® significantly improved Progression Free Survival compared to Placebo plus FASLODEX®, with manageable toxicities. The authors commented that this is the first study to show statistically significant, clinically meaningful PFS improvement with an alpha-specific PI3K inhibitor in PIK3CA-mutated HR+, HER2 negative advanced breast cancer, highlighting the importance of clinical genomics in advanced breast cancer. It however remains unclear whether Alpelisib should be incorporated into the current treatment paradigm upfront, along with endocrine therapy and a CDK 4/6 inhibitor, or sequentially following disease progression on a combination of endocrine therapy and a CDK 4/6 inhibitor. Alpelisib (ALP) + fulvestrant (FUL) for advanced breast cancer (ABC): results of the phase 3 SOLAR-1 trial. André F, Ciruelos EM, Rubovszky G, et al. Presented during the Presidential Symposium 1 at: 2018 ESMO Congress; October 19-23; Munich, Germany. Abstract LBA3_PR.
Axillary Radiotherapy is an Alternative to Complete Lymph Node Dissection in Early-Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. Axillary lymph node evaluation is an important part of breast cancer staging and the presence of axillary lymph metastases decreases the 5-year survival rate by 28-40%. Axillary lymph node status remains the most powerful predictor of breast cancer recurrence and survival. Axillary Lymph Node Dissection (ALND) was first advocated in the 18th century as part of the treatment of invasive breast cancer and has been standard practice until 2 decades back. ALND can be associated with significant morbidities such as upper limb lymphedema, pain, and sensitivity disorders and this can have a major psychological impact on breast cancer patients. Sentinel Lymph Node Biopsy (SLNB) which was introduced into clinical practice in the mid 1990’s, however has now become the standard for Stage I and II breast cancer. The sentinel node is the first lymph node(s) to which cancer cells are most likely to metastasize from a primary tumor. With the introduction of intraoperative lymphatic mapping in the 1990s, Sentinel Lymph Node Biopsy (SLNB) has now gained general acceptance and if the sentinel node is negative for metastatic disease or only has minimal disease, then no further axillary surgery is indicated. Unlike Axillary Lymph Node Dissection (ALND), SLNB is associated with a lower incidence of lymphedema/ seroma at the surgery site, paresthesias and restriction of joint movement. Nine randomized clinical trials have not shown any difference in mortality among patients who underwent ALND or SLNB for either lymph node metastases or negative sentinel lymph nodes, validating Sentinel Lymph Node Biopsy (SLNB). The American Society of Clinical Oncology (ASCO) first published guidelines on the use of SLNB for patients with early stage breast cancer in 2005, based on one randomized clinical trial. Since then, ASCO updated Clinical Practice Guideline based on additional information from 9 randomized clinical trials and13 cohort studies pertinent to SLNB and ALND.
Patients with T1-2 tumors with positive Sentinel Lymph Node Biopsy usually undergo complete ALND and there is increasing controversy about whether ALND is always necessary. AMAROS is a multicenter, randomized phase III trial, sponsored by the European Organisation for Research and Treatment of Cancer (EORTC), in which the effectiveness of complete Axillary Lymph Node Dissection (ALND) was compared with axillary Radiation Therapy (RT), in patients with invasive breast cancer. The rationale was that Radiation Therapy uses high-energy x-rays to damage tumor cells and may be a less invasive treatment and causes fewer side effects than complete ALND. This study was conducted to evaluate whether axillary RT could yield comparable outcomes to ALND with fewer adverse side effects, in this patient population. This trial enrolled 4806 patients with early-stage, clinically node-negative breast cancer of whom 1425 patients had a positive sentinel lymph node biopsy. Of these patients, 744 were randomly assigned to undergo complete ALND, whereas 681 patients received axillary RT. Both treatment groups were well balanced. The first 5-year follow up data published in 2013 showed that upper extremity lymphedema occurred significantly less often in those who received Radiotherapy compared with those who underwent complete ALND, and recent Quality of life and morbidity data supported these earlier findings.
The authors herein presented the 10 year follow up data of the AMAROS trial. It was noted that at 10 years, 1.82% of patients assigned to axillary RT had an axillary recurrence compared with 0.93% of those assigned to complete ALND, and this suggested that there was no significant difference (P=0.36). Further, there was no significant difference in the distant metastasis–free survival or Overall Survival between the two treatment groups. The distant metastasis–free survival was 78.2% among those assigned to axillary RT and 81.7% among those assigned to complete ALND and Overall Survival in the two treatment groups was 81.4% and 84.6%, respectively. It was noted that there was a higher 10-year cumulative incidence of second primaries among patients assigned to axillary RT compared with those assigned to complete Axillary Lymph Node Dissection (12% versus 8.3%). It remains unclear whether the addition of the radiation will increase the risk of second primary cancers.
It was concluded that axillary Radiotherapy is noninferior to complete Axillary Lymph Node Dissection in terms of locoregional control and this trial suggests that some patients with a positive sentinel lymph node biopsy may be appropriate candidates for axillary Radiotherapy. Rutgers, E. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer patients: 10-year results of the EORTC AMAROS trial. Presented at the 2018 San Antonio Breast Cancer Symposium; December 4-8; San Antonio, Texas.(Abstract GS4-01)
Adjuvant KADCYLA® Superior to HERCEPTIN® in High Risk HER2-Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of HERCEPTIN®, several other HER2-targeted therapies have become available. The duration of adjuvant HERCEPTIN® therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials.
KADCYLA® (Ado-Trastuzumab Emtansine, T-DM1) is an Antibody-Drug Conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release. In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® (Lapatinib) plus XELODA® (Capecitabine), in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a Taxane.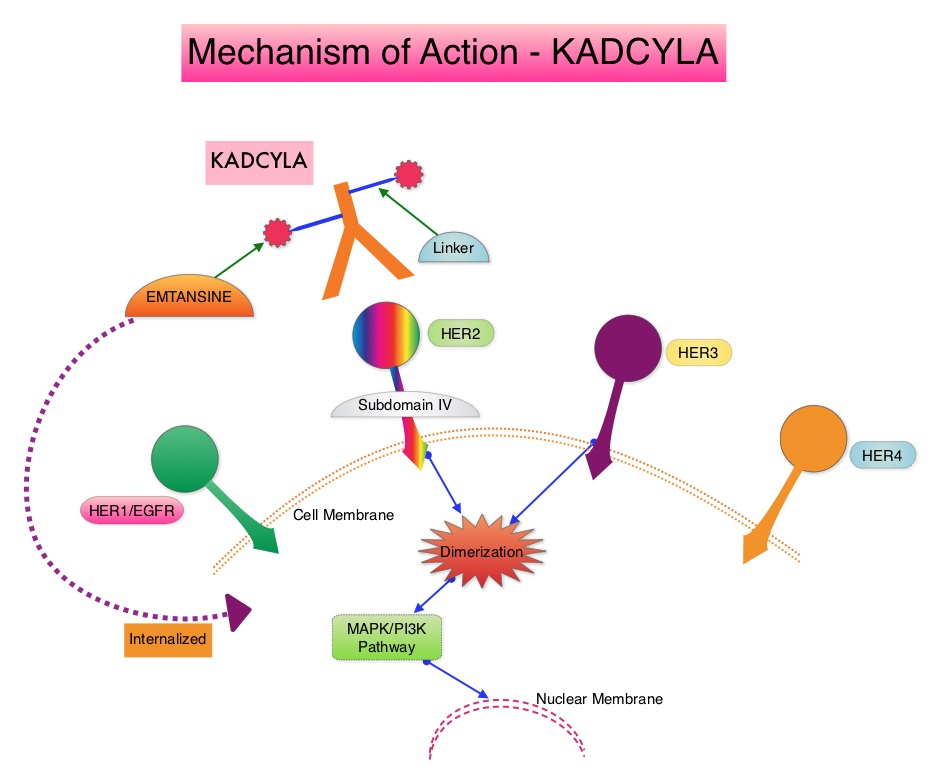
The present study was conducted to address the unmet need of patients who have residual invasive breast cancer after receiving neoadjuvant chemotherapy plus HER2-targeted therapy. These high risk patients have an unfavorable prognosis, compared to those who have no residual cancer following neoadjuvant therapy. The KATHERINE trial is an open-label, phase III global study, which compared KADCYLA® with HERCEPTIN®, as an adjuvant treatment for patients with HER2-positive early breast cancer, who had residual invasive disease following neoadjuvant chemotherapy and HERCEPTIN®.
This study included 1,486 patients with HER2-positive early stage breast cancer, who were found to have residual invasive disease in the breast or axillary lymph nodes at surgery, following at least six cycles (16 weeks) of neoadjuvant chemotherapy with a Taxane (with or without Anthracycline) and HERCEPTIN®. Within 12 weeks of surgery, patients (N=1486) were randomly assigned in a 1:1 ratio to KADCYLA® 3.6 mg/kg IV every 3 weeks or HERCEPTIN® 6 mg/kg IV every 3 weeks, for 14 cycles (743 patients in each group). Both treatment groups were well balanced and hormone receptor positive disease was present in 72% of the patients. The majority of the patients (77%) had received an Anthracycline-containing neoadjuvant chemotherapy regimen, and in 19% of the patients, another HER2-targeted agent in addition to HERCEPTIN® had been administered as a component of neoadjuvant therapy. The Primary end point was invasive Disease Free Survival (defined as freedom from ipsilateral invasive breast tumor recurrence, ipsilateral locoregional invasive breast cancer recurrence, contralateral invasive breast cancer, distant recurrence, or death from any cause). The median duration of follow up was 41.4 months in the KADCYLA® group and 40.9 months in the HERCEPTIN® group.
At the prespecified interim analysis, invasive disease occurred in 12.2% of patients who received KADCYLA® and 22.2% of patients who received HERCEPTIN®. The estimated percentage of patients who were free of invasive disease at 3 years was 88.3% in the KADCYLA® group and 77.0% in the HERCEPTIN® group. Invasive Disease Free Survival which was the Primary end point of the study was significantly higher in the KADCYLA® group than in the HERCEPTIN® group (HR=0.50; P<0.001). This suggested that KADCYLA® reduced the risk of developing an invasive breast cancer recurrence or death by 50%. Distant recurrence as the first invasive disease event occurred in 10.5% of patients in the KADCYLA® group and in 15.9% of the HERCEPTIN® group. A consistent benefit was seen across all prespecified subgroups. Adverse events were consistent with the known safety profile of KADCYLA®, with more toxicities associated with KADCYLA® than with HERCEPTIN®. Additional follow-up will be necessary to determine the Overall Survival benefit with adjuvant KADCYLA®.
It was concluded that among patients with HER2-positive early breast cancer who had residual invasive disease after completion of neoadjuvant therapy, substituting KADCYLA® for adjuvant HERCEPTIN® reduced the risk of recurrence of invasive breast cancer or death was 50%, with the benefit seen across all patient subgroups. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. von Minckwitz G, Huang C-S, Mano MS, et al. for the KATHERINE Investigators. (published online December 5, 2018). New Engl Jour Med. doi: 10.1056/NEJMoa1814017
Testing for BRCA1 and BRCA2 Mutations May Not Be Adequate in Breast Cancer Patients
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and 40,920 women are expected to die from the disease. DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. Mutations in BRCA1 and BRCA2 account for about 20 to 25 percent of hereditary breast cancers and about 5 to 10 percent of all breast cancers.These mutations can be inherited from either of the parents and a child has a 50 percent chance of inheriting this mutation and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal.
Breast cancer patients have a 5-12% lifetime risk of a second primary cancer. It remains unclear however whether patients with breast cancer and another primary cancer have mutations in genes other than BRCA1 and BRCA2, compared to those with a single breast cancer. There are well established data on the cancer risks, associated with different gene mutations. The authors hypothesized that among these patients, a number of factors including environmental exposures and genetic predisposition, may play a role in the development of more than one primary cancer in their lifetime. Recently published study suggested that there was a 85% cumulative breast cancer risk by age 60 years, among those with mutations in the TP53 gene (Cancer2016;122(23):3673-3681). Further with the increasing recognition that germline mutations in genes may have clinical and treatment implications, majority of patients are feeling comfortable opting for upfront multiple genetic mutation testing.
The researchers in this study looked at a panel of 15 actionable mutations beyond BRCA and the gene panel included TP53, PALB2, CDH1, PTEN, STK11, CHEK2, ATM, NBN, MSH6, PMS2, MSH2, MLH1, CDKN2A, MUTYH monoallelic, and CHEK2 Low Risk. Two cohorts of BRCA1 and BRCA2 negative patients were studied. The first cohort included high-risk breast cancer patients with either a single breast cancer (N=464) or breast cancer and an additional primary cancer (N=551). The second cohort comprised of patients with familial breast cancer (inherited risk) with either a single breast cancer (N=1464) or breast cancer and another primary cancer (N=340).
In a total of 891 patients in both cohorts who had breast cancer and an additional primary cancer, there was twice the risk of inheriting mutations in genes other than BRCA1 and BRCA2. In cohort 1, the mutation rate among patients who had breast cancer and an additional primary cancer was 8.7% compared to 4.1% among those with single breast cancer (P=0.003) and in cohort 2, the mutation rate was 8.2% versus 4.2%, respectively (P=0.003).
There was however a differences in individual gene mutation rates between the two cohorts. Among patients with breast cancer and an additional primary cancer in cohort 1, mutations in TP53 and MSH6 were significantly higher, whereas among the patients in cohort 2 with familial breast cancer, mutations in ATM, CHEK2 and PALB2 were significantly higher both in those with breast cancer and another primary cancer and those with a single breast cancer.
The authors concluded that patients with multiple primary cancers should be offered multiplex panel testing to identify patients at risk. Identifying mutations, especially mutations in the TP53 gene may have a bearing on appropriate recommendations such as risk-reducing bilateral mastectomy or mastectomy instead of a lumpectomy in this patient group. Thus, risk assessment using multiple genetic testing panels can be beneficial for clinical care and surveillance. Inherited mutations in breast cancer patients with and without multiple primary cancers. Maxwell KN, Vijai V, Lilyquist J, et al. DOI: 10.1200/JCO.2018.36.15_suppl.1503 Journal of Clinical Oncology 36, no. 15_suppl (May 2018) 1503-1503.
ASCO Endorses Complementary Therapy Guidelines for Breast Cancer Patients
SUMMARY: The American Society for Clinical Oncology (ASCO) after careful review, endorsed the Society for Integrative Oncology (SIO) guideline on the use of integrative therapies during and after breast cancer treatment. Integrative medicine is defined as a patient-centered, evidence-informed field of care that uses mind and body practices, natural products, and/or lifestyle modifications to improve health, quality of life, and clinical outcomes. The ASCO Expert Panel determined that the recommendations in the SIO guideline published in 2017 are clear, thorough, and based on the most relevant scientific evidence. The panel emphasized that these therapies are complementary and should be used along with conventional anticancer treatment, thereby taking a more comprehensive treatment approach across the cancer care continuum, to manage symptoms and adverse effects of breast cancer treatment. This ASCO guideline excluded lifestyle changes such as diet and exercise, mainstream interventions such as support groups, and practices like attention-restoration therapy, that are still being evaluated. Practices such as prayer and spirituality were not considered specifically integrative oncology therapy.
Guideline Question
What are evidence-based approaches to the use of integrative therapies in the management of symptoms and adverse effects during and after breast cancer treatment?
Target Population
Patients undergoing treatment of breast cancer and survivors of breast cancer
Target Audience
Oncologists, integrative medicine providers, supportive care specialists, nurses, pharmacists, primary care providers, and patients with breast cancer

Key Recommendations
Acute Radiation Skin Reaction
Aloe vera and hyaluronic acid cream should not be recommended for improving acute radiation skin reaction. (Grade D)
Anxiety and Stress Reduction
Meditation is recommended for reducing anxiety. (Grade A)
Music therapy is recommended for reducing anxiety. (Grade B)
Stress management is recommended for reducing anxiety during treatment, but longer group programs are likely better than self-administered home programs or shorter programs. (Grade B)
Yoga is recommended for reducing anxiety. (Grade B)
Acupuncture, massage, and relaxation can be considered for reducing anxiety. (Grade C)
Chemotherapy-Induced Nausea and Vomiting
Acupressure can be considered as an addition to antiemetic drugs to control nausea and vomiting during chemotherapy. (Grade B)
Electroacupuncture can be considered as an addition to antiemetic drugs to control vomiting during chemotherapy. (Grade B)
Ginger and relaxation can be considered as additions to antiemetic drugs to control nausea and vomiting during chemotherapy. (Grade C)
Glutamine should not be recommended for improving nausea and vomiting during chemotherapy. (Grade D)
Depression and Mood Disturbance
Meditation, particularly mindfulness-based stress reduction, is recommended for treating mood disturbance and depressive symptoms. (Grade A)
Relaxation is recommended for improving mood disturbance and depressive symptoms. (Grade A)
Yoga is recommended for improving mood disturbance and depressive symptoms. (Grade B)
Massage is recommended for improving mood disturbance. (Grade B)
Music therapy is recommended for improving mood disturbance. (Grade B)
Acupuncture, healing touch, and stress management can be considered for improving mood disturbance and depressive symptoms. (Grade C)
Fatigue
Hypnosis and ginseng can be considered for improving fatigue during treatment. (Grade C)
Acupuncture and yoga can be considered for improving post-treatment fatigue. (Grade C)
Acetyl-l-carnitine and guarana should not be recommended for improving fatigue during treatment. (Grade D)
Lymphedema
Low-level laser therapy, manual lymphatic drainage, and compression bandaging can be considered for improving lymphedema. (Grade C)
Neuropathy
Acetyl-l-carnitine is not recommended for the prevention of chemotherapy-induced peripheral neuropathy in patients with breast cancer due to potential harm. (Grade H)
Pain
Acupuncture, healing touch, hypnosis, and music therapy can be considered for the management of pain. (Grade C)
Quality of Life
Meditation is recommended for improving quality of life. (Grade A)
Yoga is recommended for improving quality of life. (Grade B)
Acupuncture, mistletoe, qigong, reflexology, and stress management can be considered for improving quality of life. (Grade C)
Sleep Disturbance
Gentle yoga can be considered for improving sleep. (Grade C)
Vasomotor/Hot Flashes
Acupuncture can be considered for improving hot flashes. (Grade C)
Soy is not recommended for hot flashes in patients with breast cancer due to lack of effect. (Grade D)
Integrative Therapies During and After Breast Cancer Treatment: ASCO Endorsement of the SIO Clinical Practice Guideline. Lyman GH, Greenlee H, Bohlke K, et al. J Clin Oncol. 2018;36:2647-2655
TECENTRIQ® and ABRAXANE® Significantly Prolonged Survival in Advanced Triple Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and 40,920 women are expected to die from the disease. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor-2) negative. TNBC accounts for 15-20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. Those who do not achieve a pathological Complete Response tend to have a poor prognosis. It therefore appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway.
Previously published studies have shown that tumor-infiltrating lymphocytes were associated with clinical benefit, when treated with chemotherapy and immunotherapy, in patients with TNBC, and improved clinical benefit was observed in patients with immune-enriched molecular subtypes of metastatic TNBC. TECENTRIQ® (Atezolizumab) is an anti PD-L1 monoclonal antibody, designed to directly bind to PD-L1 expressed on tumor cells and tumor-infiltrating immune cells, thereby blocking its interactions with PD-1 and B7.1 receptors, and thus enabling the activation of T cells. Single-agent TECENTRIQ® is presently approved for the treatment of metastatic Urothelial carcinoma and Non Small Cell Lung Cancer (NSCLC). TECENTRIQ® has also been shown to have clinical activity with acceptable safety profile in patients with other solid tumors including Triple Negative Breast Cancer. The premise for combining chemotherapy with immune checkpoint inhibitors is that chemotherapy may enhance release of tumor antigens and antitumor responses to immune checkpoint inhibition. Taxanes in particular may additionally activate toll-like receptor activity and promote dendritic-cell activity. ABRAXANE® (Nanoparticle Albumin-Bound - nab Paclitaxel) was selected as a chemotherapy partner in the present study because at the time that this trial was designed, the glucocorticoid premedication that is required with solvent-based Paclitaxel (TAXOL®), had been hypothesized to affect immunotherapy activity.
IMpassion130 is an international, randomized, double-blind, placebo-controlled phase III trial in which first-line treatment with TECENTRIQ® plus ABRAXANE®, was compared with placebo ABRAXANE®, in patients with locally advanced or metastatic Triple Negative Breast Cancer. Patients with untreated metastatic Triple Negative Breast Cancer (N=902) were randomly assigned in a 1:1 ratio and received TECENTRIQ® 840 mg IV or placebo on days 1 and 15 and ABRAXANE® 100 mg/m2 IV on days 1, 8, and 15 of every 28-day cycle. Treatment was continued until disease progression or unacceptable toxicity. Stratification factors included presence or absence of liver metastases, use or non-use of neoadjuvant or adjuvant taxane treatment, and PD-L1 expression on tumor-infiltrating immune cells as a percentage of tumor area (less than 1% was considered PD-L1 negative versus1% or more considered PD-L1 positive) according to ImmunoHistochemical testing (IHC). Both treatment groups were well balanced. Approximately 40% of the patients were PD-L1 positive. The two Primary end points were Progression Free Survival (PFS) and Overall Survival (OS).
At a median follow up of 12.9 months, the median PFS in the intent-to-treat population was 7.2 months with TECENTRIQ® plus ABRAXANE®, as compared with 5.5 months with placebo plus ABRAXANE® (HR=0.80; P=0.002). This suggested a 20% improvement in PFS with the TECENTRIQ® combination. Among patients with PD-L1–positive tumors, the addition of TECENTRIQ® improved PFS by 38% (HR=0.62; P<0.001) and similar benefit was noted in the OS, with a median OS of 25 months with the TECENTRIQ® combination and 15.5 months with placebo plus ABRAXANE® (HR=0.62). Grade 3 or 4 adverse events were 48.7% in the TECENTRIQ® and ABRAXANE® and 42.2% in the placebo plus ABRAXANE® group.
It was concluded that TECENTRIQ® plus ABRAXANE® significantly prolonged Progression Free Survival among patients with metastatic Triple Negative Breast Cancer in both the intent-to-treat population and the PD-L1 positive subgroup, and could potentially change how we manage patients with Triple Negative Breast Cancer. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. Schmid P, Adams S, Rugo HS, et al. October 20, 2018. DOI: 10.1056/NEJMoa1809615
Selection of Optimal Adjuvant Chemotherapy and Targeted Therapy for Early Breast Cancer: ASCO Clinical Practice Guideline Focused Update
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer.
ASCO published an adaptation of the Cancer Care Ontario guideline, on optimal adjuvant chemotherapy regimens for early breast cancer and adjuvant targeted therapy for HER2 (Human Epidermal Growth Factor receptor 2) positive breast cancers, in 2016. The recent publication of phase III studies relevant to the clinical care of breast cancer patients prompted the ASCO Update Steering Group of the original Expert Panel, to provide a focused update of the 2016 guideline. With the exception of this focused update, the remaining recommendations from the 2016 ASCO guideline are unchanged.
Selection of Optimal Adjuvant Chemotherapy and Targeted Therapy for Early Breast Cancer: ASCO Clinical Practice Guideline Focused Update
Questions Addressed in Focused Update
1) Should adjuvant Capecitabine be given following completion of standard preoperative Anthracycline- and Taxane-based combination chemotherapy in patients with early-stage HER2-negative breast cancer with residual invasive disease at surgery?
2) Should 1 year of adjuvant Pertuzumab be added to Trastuzumab-based combination chemotherapy in patients with early stage HER2-positive breast cancer?
3) Should Neratinib be offered as extended adjuvant therapy for patients after combination chemotherapy and Trastuzumab-based adjuvant therapy with early-stage, HER2-positive breast cancer?
Target Population
Patients who are being considered for, or who are receiving, systemic therapy following definitive surgery for early-stage invasive breast cancer, defined largely as invasive cancer anatomic Stages I to IIIC
Target Audience
Medical oncologists, Pathologists, Surgeons, Oncology nurses, Patients, and Caregivers.
Focused Update Recommendations
Patients with early-stage HER2-negative breast cancer with pathologic invasive residual disease at surgery following standard Anthracycline and Taxane-based preoperative therapy may be offered up to six to eight cycles of adjuvant Capecitabine (XELODA®).
Qualifying statements: If clinicians decide to use Capecitabine, then the Expert Panel preferentially supports the use of adjuvant Capecitabine in the subgroup of patients with Hormone Receptor negative, HER2-negative disease. The Capecitabine dosage used in the CREATE-X study (1,250 mg/m2 PO twice daily) is associated with higher toxicity in patients 65 years or older.
Clinicians may add 1 year of adjuvant Pertuzumab (PERJETA® ) to Trastuzumab (HERCEPTIN®)-based combination chemotherapy in patients with high-risk, early-stage, HER2-positive breast cancer.
Qualifying statements: The Expert Panel preferentially supports Pertuzumab in patients with node-positive, HER2-positive breast cancer in view of the clinically insignificant absolute benefit observed among node-negative patients. After a median follow up of 3.8 years, Pertuzumab offered a modest Disease Free Survival (DFS) benefit. The first planned interim analysis did not show an Overall Survival (OS) benefit in the trial population. There are no data to guide the duration of Pertuzumab in patients who received neoadjuvant Pertuzumab and achieved a pathologic Complete Response.
Clinicians may use extended adjuvant therapy with Neratinib (NERLYNX®) to follow Trastuzumab in patients with early stage, HER2-positive breast cancer. Neratinib causes substantial diarrhea and diarrhea prophylaxis must be used.
Qualifying statements: The Expert Panel preferentially favors use of Neratinib in patients with HER2-positive, Hormone Receptor positive, and node-positive disease. At a median follow-up of 5.2 years, no OS benefit has been observed. Patients who began Neratinib within 1 year of Trastuzumab completion appeared to derive the greatest benefit. There are no data on the added benefit of Neratinib in patients who also received Pertuzumab in the neoadjuvant or adjuvant setting.
Selection of Optimal Adjuvant Chemotherapy and Targeted Therapy for Early Breast Cancer: ASCO Clinical Practice Guideline Focused Update. Denduluri N, Chavez-MacGregor M, Telli ML, et al. J Clin Oncol. 2018;36:2433-2443.
HER2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant and neoadjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage as well as advanced metastatic breast cancer. Since the approval of HERCEPTIN®, several other HER2-targeted therapies have become available. Accurate determination of HER2 status of the tumor is therefore essential for patients with invasive breast cancer, to ensure that those most likely to benefit are offered a HER2-targeted therapy and those who are unlikely to benefit can avoid toxicities as well as financial burden associated with those drugs.
Laboratory testing for HER2 status in patients with breast cancer in the US is performed according to guidelines developed by an Expert panel of members of the American Society of Clinical Oncology (ASCO) and the College of American Pathologists (CAP). The ASCO/CAP guidelines were first published in 2007 and were updated in 2013. The Expert panel in 2018 developed and issued a focused update of the clinical practice guideline on HER2 testing in breast cancer. This new information made available since the previous update in 2013 addresses uncommon clinical scenarios and improves clarity, particularly for infrequent HER2 test results that are of uncertain biologic or clinical significance. There are currently two approved methods for determining HER2 status in breast cancer: ImmunoHistoChemistry (IHC) and In Situ Hybridization (ISH). This new guideline enables the Health Care Provider, how to best evaluate some of the less common patterns in HER2 results emerging from ISH.
Guideline Questions
1) What is the most appropriate definition for ImmunoHistoChemistry (IHC) 2+ (IHC equivocal)?
2) Must Human Epidermal growth factor Receptor 2 (HER2) testing be repeated on a surgical specimen if the initially tested core biopsy is negative?
3) What is the optimal algorithm for less common patterns observed when performing dual-probe In Situ Hybridization (ISH) HER2 testing in breast cancer?
Updated Recommendations
1) Immunohistochemistry (IHC) 2+ is defined as invasive breast cancer with weak to moderate complete membrane staining observed in more than 10% of tumor cells.
2) If the initial HER2 test result in a core needle biopsy specimen of a primary breast cancer is negative, a new HER2 test may (not “must”) be ordered on the excision specimen based on some criteria (such as tumor grade 3).
3)The HER2 testing algorithm now includes more rigorous interpretation criteria of the less common patterns that can be seen in about 5% of all cases when HER2 status in breast cancer is evaluated using a dual-probe ISH assay. These scenarios are described as ISH group 2 (HER2/Chromosome Enumeration Probe 17 [CEP17] ratio of 2.0 or more; average HER2 copy number less than 4.0 signals per cell), ISH group 3 (HER2/CEP17 ratio less than 2.0; average HER2 copy number 6.0 or more signals per cell), and ISH group 4 (HER2/CEP17 ratio less than 2.0; average HER2 copy number 4.0 or more and less than 6.0 signals per cell). These cases, described as ISH groups 2-4, should now be assessed using a diagnostic approach that includes a concomitant review of the IHC (ImmunoHistoChemistry) test, which will help the pathologist make a final determination of the tumor specimen as HER2 positive or negative.
4)The Expert Panel also preferentially recommends the use of dual-probe instead of single-probe ISH assays, but it recognizes that several single-probe ISH assays have regulatory approval in many parts of the world.
Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. Wolff AC, Hammond EH, Allison KH, et al. J Clin Oncol 2018; 36:2105-2122.
KISQALI® plus FASLODEX® Improve Progression Free Survival in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.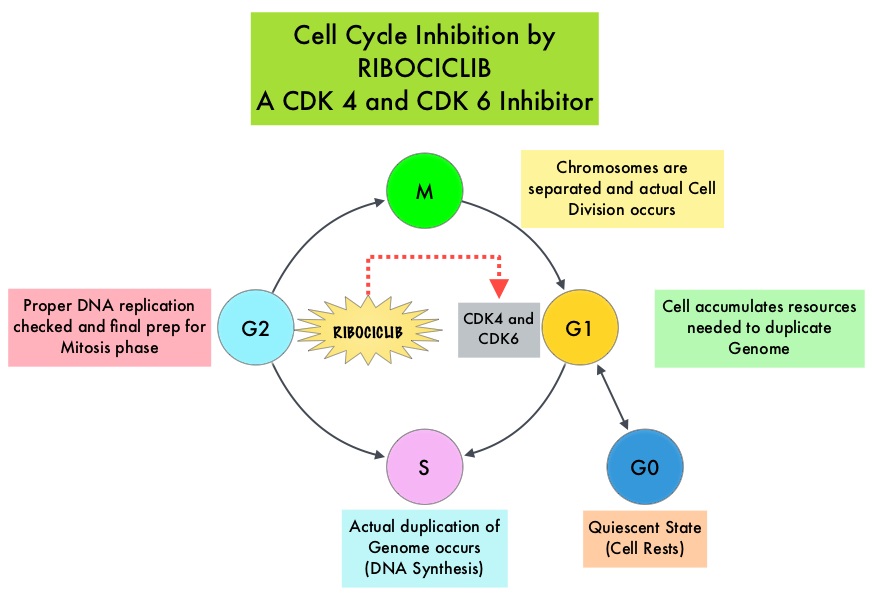
KISQALI® (Ribociclib) is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6 that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest. KISQALI® in combination with an Aromatase Inhibitor has been approved by the FDA for pre and perimenopausal women with HR (Hormone Receptor)-positive, HER2-negative advanced or metastatic breast cancer, as initial endocrine-based therapy. The efficacy of KISQALI® was evaluated in two prior randomized phase III studies. In the MONALEESA-2 trial which evaluated KISQALI® in combination with FEMARA® (Letrozole) compared to FEMARA® alone, in postmenopausal women with HR-positive, HER2-negative advanced breast cancer who received no prior therapy for their advanced breast cancer, the addition of KISQALI® to FEMARA® significantly prolonged Progression Free Survival (PFS) compared to FEMARA® alone. In the MONALEESA-7 study, KISQALI® in combination with Tamoxifen or a Non-Steroidal Aromatase Inhibitor plus ZOLADEX® (Goserelin) was compared with Tamoxifen or an Aromatase Inhibitor plus ZOLADEX®, in premenopausal or perimenopausal women with HR-positive, HER2- negative advanced breast cancer, who had not previously received endocrine therapy for advanced disease. In this study of premenopausal women, KISQALI® plus endocrine therapy significantly improved PFS compared with placebo plus endocrine therapy
MONALEESA-3 is a randomized, double-blind, placebo-controlled Phase III study which compared the efficacy of KISQALI® in combination with FASLODEX® with FASLODEX® alone, among postmenopausal women with HR-positive, HER2-negative advanced breast cancer who received no prior or only one line of prior endocrine therapy for advanced disease. In this trial, 726 women were randomized, of whom 367 were treatment-naïve and 345 patients had received up to one line of prior endocrine therapy for advanced disease. . Patients were randomized 2:1 to receive KISQALI® plus FASLODEX® (N=484) or placebo plus FASLODEX® (N=242). Treatment consisted of KISQALI® 600 mg orally daily 3 weeks on and 1 week off and FASLODEX® 500 mg IM on day 1 of each 28-day cycle, with an additional dose given on day 15 of cycle 1. Patients were stratified by the presence or absence of lung or liver metastases and prior endocrine therapy (first-line versus second-line). The median age in both groups was 63 years. The Primary endpoint was Progression Free Survival. Secondary end points included Overall Survival, Overall Response Rate, and Safety. The median time from randomization to data cutoff was 20.4 months.
Among all randomized patients, the median PFS in the KISQALI® plus FASLODEX® group was 20.5 months compared to 12.8 months in the FASLODEX® plus placebo group (HR= 0.59; P<0.001). This represented a 41% reduction in the risk of disease progression. The PFS benefit was consistent among the 367 patients who were treatment-naïve (HR=0.57) and 345 patients had received up to one line of prior endocrine therapy for advanced disease (HR=0.56). In the subgroup of patients taking KISQALI® plus FASLODEX® as first-line treatment, the median PFS was not reached and 70% were estimated to remain Progression Free at median follow up of 16.5 months. Among those patients with measurable disease at baseline, the Overall Response Rate was 40.9% for the KISQALI® plus FASLODEX® arm versus 28.7% for FASLODEX® plus placebo group (P=0.003). At first interim analysis, the Overall Survival data were immature. The most common grade 3/4 Adverse Events in patients receiving KISQALI® plus FASLODEX® compared to FASLODEX® alone were neutropenia (53.4% versus 0%) and leukopenia (14.1% versus 0%).
It was concluded that KISQALI® plus FASLODEX® might represent a new, first or second-line treatment option for patients with Hormone Receptor-positive, Human Epidermal growth factor Receptor 2-negative advanced breast cancer. MONALEESA-3 is the only randomized Phase III trial to study a CDK4/6 inhibitor plus FASLODEX® in the first-line setting, with demonstrable efficacy in patients with de novo advanced breast cancer and those who had not received adjuvant therapy in more than a year. Ribociclib (RIB) + fulvestrant (FUL) in postmenopausal women with hormone receptor-positive (HR+), HER2-negative (HER2 –) advanced breast cancer (ABC): Results from MONALEESA-3. Slamon DJ, Neven P, Chia SKL, et al. J Clin Oncol. 2018: 36, (suppl; abstr 1000).
Six Months Adjuvant HERCEPTIN® Safer and Efficacious in Early Stage HER2+ Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2, and adjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early stage breast cancer. The duration of adjuvant HERCEPTIN® therapy has been 12 months and this length of treatment was empirically adopted from the pivotal registration trials.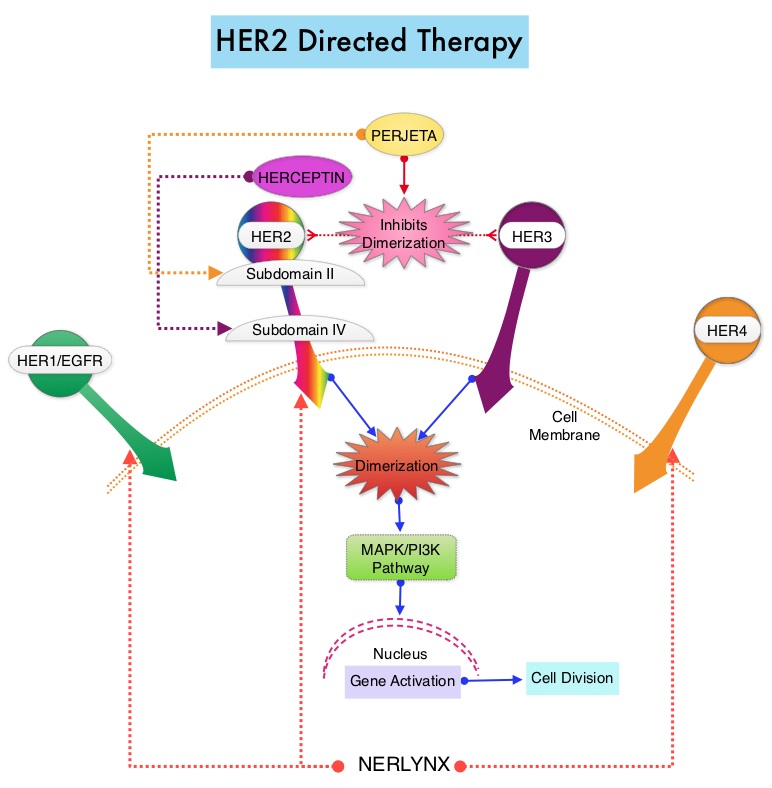
PERSEPHONE is a randomized, phase III, noninferiority trial in which a 6-month course of adjuvant HERCEPTIN® was compared with the standard 12-month course, among patients with HER2-positive early breast cancer. This study was conducted based on the hypothesis that shorter course of treatment with HERCEPTIN® could reduce cardiotoxicities as well as cost without compromising efficacy. This trial randomized 4089 patients across 152 sites in a 1:1 ratio to receive HERCEPTIN® for 6 months (N=2044) or 12 months (N=2045). In this trial, 69% of patients had ER-positive tumors, 41% received Anthracycline-based chemotherapy, 49% received Anthracycline and Taxane-based chemotherapy, 10% received Taxane-based chemotherapy, 85% received adjuvant chemotherapy, and sequential HERCEPTIN® was administered in 54% of patients. This study also included assessment of Left Ventricular Ejection Fraction (LVEF) every 3 months until month 12, as well as continued Quality of Life and health economic assessments at months 18 and 24. The Primary endpoint was Disease Free Survival (DFS) from the time of diagnosis.
At a median follow-up period of 5 years, the 4-year DFS rate was identical in both treatment groups. DFS was 89.8% with 12 months of HERCEPTIN® compared with 89.4% with the 6-month course, which met the criteria for noninferiority (P=0.01). Further, only 4% of the patients enrolled in the 6-month HERCEPTIN® group discontinued HERCEPTIN® treatment due to cardiotoxicities compared with 8% in the 12-month group (P<0.0001), suggesting that the number of patients stopping treatment due to cardiac toxicities was cut in half with the shorter duration of treatment with HERCEPTIN®. Patients receiving shorter course of HERCEPTIN® also had a more rapid recovery of their cardiac LVEF following treatment, compared with the standard of care group (P=0.02).
It was concluded from this largest, reduced duration, noninferiority trial that a shorter 6-month course of adjuvant HERCEPTIN® was noninferior for Disease Free Survival, compared with the standard 12-month schedule, among patients with HER2-positive early breast cancer, with an additional benefit of reduction in cardiac toxicities, as well as cost both to the patients and healthcare systems. Overall Survival data was not available at the time of this analysis. PERSEPHONE: 6 versus 12 months (m) of adjuvant trastuzumab in patients (pts) with HER2 positive (+) early breast cancer (EBC): Randomised phase 3 non-inferiority trial with definitive 4-year (yr) disease-free survival (DFS) results. Earl HM, Hiller L, Vallier A-L, et al. J Clin Oncol 36, 2018 (suppl; abstr 506)
Late Breaking Abstract - ASCO 2018: Endocrine Therapy Alone is Adequate for Early Stage Breast Cancer Patients with Intermediate Risk Recurrence Score
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. Approximately 50% of all breast cancers are Estrogen Receptor (ER) positive, HER2-negative, axillary node-negative tumors. Patients with early stage breast cancer often receive adjuvant chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for early stage, hormone receptor positive, HER negative, early stage breast cancer patients, are often made based on tumor size, grade, ImmunoHistoChemical (IHC) markers such as Ki-67, nodal status and Oncotype DX Recurrence Score (RS) assay.
Oncotype Dx assay categorizes patients on the basis of Recurrence Scores into Low risk (less than 18), Intermediate risk (18-30), and High risk (31 or more). It has been unclear whether patients in the Intermediate risk group benefited from the addition of chemotherapy to endocrine therapy. TAILORx was specifically designed to address this question and provide a very definitive answer. In this study, the Intermediate risk Recurrent Score (18-30) was changed to 11-25, to account for exclusion of higher-risk patients with HER2-positive disease and to minimize the potential for under treatment.
TAILORx ((Trial Assigning Individualized Options for Treatment) is a phase III, randomized, prospective, non-inferiority trial, and is the largest breast cancer treatment trial ever conducted, and the first precision medicine trial ever done, according to the authors. In this study, 10,273 women, 18-75 years of age, with hormone receptor-positive, HER2-negative, axillary node-negative breast cancer were enrolled. Patients had tumors 1.1-5.0 cm in size (or 0.6-1.0 cm and intermediate/high grade). Patients were divided into three groups based on their Recurrence Score. Women with a Low Recurrence Score of 0-10 received endocrine therapy alone and those with a High Recurrence Score of 26-100 received endocrine therapy in combination with standard adjuvant chemotherapy. Patient with Intermediate Recurrence Score of 11-25 (N=6711) were randomly assigned to receive endocrine therapy alone or endocrine therapy and adjuvant chemotherapy. The Primary endpoint was invasive Disease Free Survival, defined as recurrence of cancer in the breast, regional lymph nodes, and/or distant organs, a second primary cancer in the opposite breast or another organ, or death from any cause.
At a median follow-up of 7.5 years, the study met its Primary endpoint, and it was noted that that endocrine therapy alone was non-inferior to chemotherapy plus endocrine therapy, in patients with Intermediate Recurrence Score of 11-25. At 9 years, patients with Intermediate Recurrence Scores receiving endocrine therapy or chemotherapy in combination with endocrine therapy showed similar invasive Disease Free Survival rates (83.3% vs 84.3%), distant Recurrence Free Interval (94.5% vs 95.0%), Recurrence Free Interval (92.2% vs 92.9%) and Overall Survival (93.9% vs 93.8%) respectively. These findings suggested that there was no benefit from adding chemotherapy to endocrine therapy, for this patient group.
The authors also conducted an exploratory analysis of patients in the Intermediate Risk group to determine which patients would benefit from added chemotherapy. They noted that there was no significant interaction between menopause, tumor size or grade, with Recurrence Score. There was however an interaction between age and Recurrence Score. In women 50 years or younger with a Recurrence Score of 16-20, there were 2% fewer distant recurrences, and in those with a recurrence score of 21-25, there were 7% fewer distant recurrences with the addition of chemotherapy, suggesting that younger women with a Recurrence Score of 16-25 had some benefit with the addition of chemotherapy to endocrine therapy.
It was concluded that women older than 50 years with hormone receptor-positive, HER2-negative, node-negative breast cancer and a Recurrence Score of 0-25, as well as women 50 years or younger with hormone receptor-positive, HER2-negative, node-negative breast cancer and a Recurrence Score of 0-15, could be spared from chemotherapy, based on this study. This study showed that chemotherapy could be avoided in about 70% of these patients, by allowing this test to tailor treatment. Further, this prospective study reflects outcomes with current modern chemotherapy and endocrine therapy regimens. The authors recommended that any patient 75 years or younger with early-stage breast cancer should therefore be offered Oncotype DX assay test, for guidance regarding chemotherapy recommendations after surgery. TAILORx: phase III trial of chemoendocrine therapy versus endocrine therapy alone in hormone receptor-positive, HER2-negative, node-negative breast cancer and an intermediate prognosis 21-gene recurrence score. Sparano JA, Gray RJ, Wood WC, et al. J Clin Oncol. 2018;36(suppl; abstr LBA1).
Increased Risk of Anaplastic Large-Cell Lymphoma with Breast Implants
SUMMARY: Breast implants are among the most commonly used medical devices and the number of women with breast implants diagnosed with Anaplastic Large Cell Lymphoma in the breast (breast-ALCL) have increased over the past 10 years. The FDA in 2011, identified a possible association between breast implants and the development of Anaplastic Large Cell Lymphoma (ALCL), and there has been growing body of medical literature describing the natural history and long term outcomes of the disease. In 2016, the World Health Organization designated Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) as a T-Cell Lymphoma that can develop following breast implants.
The incidence of ALCL is higher with macrotextured implants than smooth implants. It has been postulated that textured implants have an increased surface area and a local inflammatory response, elicited by silicone-derived products or specific bacterial species adherent to the prosthesis surface (Biofilm) may play a role, potentiating capsular contracture and increasing T-cell response and possible conversion to BIA-ALCL. It remains unclear however, whether certain women including those with proven BRCA mutations have a genetically determined increased risk to develop lymphoma when exposed to breast implants via a genetically determined altered or exaggerated local immunological response. There are presently no evidence-based guidelines on how this condition should be detected, treated or followed up. It is however, suggested that any seroma more than 6-12 months after breast implantation, in the absence of infection, should be evaluated with ultrasonography and mammography, to identify a mass or lymph nodes that are suspicious for lymphoma. The seroma should be aspirated and the fluid analyzed for cytology, cultures, flow cytometry and cell block. If cytology is negative, patients should be closely monitored for recurrence of seroma. Biopsy should be performed if a mass is present, and if ALCL is confirmed, patients will respond to capsulectomy and removal of the implant, as ALCL confined within the capsule often have an indolent course and good prognosis. In contrast, patients who present with a distinct mass may have a more aggressive disease course and poor prognosis, and require chemotherapy and/or radiation therapy. Postoperatively evaluation if BIA-ALCL is confirmed, should include a PET-CT scan and bone marrow biopsy to rule out systemic disease. It is recommended that patients receive clinical follow up at least every 6 months for 5 years along with breast ultrasonography for 2 years, and those who undergo reinsertion of the implant should be followed up beyond 5 years.
In this publication, a case-control study was conducted, comparing the prevalence of breast implants between women with primary breast-ALCL and women with primary breast lymphomas other than ALCL, using a comprehensive Dutch pathology database. The purpose of this study was to determine the relative and absolute risks of breast ALCL in women with breast implants. The authors identified all patients diagnosed with primary Non Hodgkin Lymphoma in the breast between 1990 and 2016 and retrieved clinical data, including breast implant status, from the treating physicians, through the population-based nationwide Dutch pathology registry. They then calculated the risk of breast-ALCL in women both with and without breast implants.
The estimated prevalence of breast implants in women aged 20-70 years was 3.3%. It was noted that among 43 patients with breast-ALCL (median age 59 years), 32 had ipsilateral breast implants, compared with 1 among 146 women with other primary breast lymphomas. The median time to development of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) was 13 years after an implant was placed. Most cases of BIA-ALCL (75%) in this Dutch study were associated with macrotextured implants and the Complete Remission rate was 90% after treatment and only 6% of the 32 affected women died of disseminated disease. In this study, the number of women with implants needed to cause 1 breast-ALCL case before age 75 years was 6920. Affected patients had implants for cosmetic reasons alone, for reconstruction in transgender surgery, after breast cancer surgery, and after prophylactic mastectomy for high breast cancer risk.
It was concluded that breast implants are associated with increased risk of breast-ALCL, but the absolute risk remains small. With an increasing number of breast implant insertions, the need for increased awareness among the public, medical professionals, and regulatory bodies is imperative and it is essential to promote alternative cosmetic procedures, and recognize this rare clinical entity. Breast Implants and the Risk of Anaplastic Large-Cell Lymphoma in the Breast. de Boer M, van Leeuwen FE, Hauptmann M, et al. JAMA Oncol. 2018;4:335-341.
Dual HER2 Blockade Along with an Aromatase Inhibitor is an Effective Chemotherapy-Sparing Alternative Treatment in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1 (EGFR), HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. Approximately 50% of HER2-positive breast cancers are Hormone Receptor positive.
Patients with HER2-positive metastatic breast cancer are often treated with anti-HER2 targeted therapy along with chemotherapy, irrespective of hormone receptor status, and this has resulted in significantly improved treatment outcomes. HER2-targeted therapies include HERCEPTIN® (Trastuzumab), TYKERB® (Lapatinib), PERJETA® (Pertuzumab) and KADCYLA® (ado-Trastuzumab emtansine).
Dual HER2 blockade with HERCEPTIN® and PERJETA®, given along with chemotherapy (with or without endocrine therapy), as first line treatment, in HER2 positive metastatic breast cancer patients, was shown to significantly improve Progression Free Survival (PFS) as well as Overall Survival. The superior benefit with dual HER2 blockade has been attributed to differing mechanisms of action and synergistic interaction between HER2 targeted therapies. Not all HER2-positive, Hormone Receptor positive metastatic breast cancer patients, are candidates for chemotherapy. These patients however may benefit from anti-HER2 targeted therapy given along with endocrine therapy.
Preclinical evidence suggested that endocrine resistance may be related to cross talk between HER2- and Hormone Receptor-signaling pathways. HER2 inhibition may in turn enable the Estrogen Receptor (ER) to become the primary driver of cell proliferation, resulting in relative resistance to anti-HER2 therapy. Therefore, targeting both HER2 and ER simultaneously may be essential to derive optimal benefit among patients with HER2-positive Hormone Receptor positive metastatic breast cancer. Previously published studies demonstrated improved median PFS when single HER2 blockade combined with endocrine therapy was compared with endocrine therapy alone, among treatment naïve patients with HER2-positive and Hormone Receptor positive metastatic breast cancer.
Based on the improved outcomes with dual anti-HER2 blockade compared with single HER2 blockade in both neoadjuvant as well as metastatic settings, this present study was designed to evaluate the superiority of dual HER2 blockade with TYKERB® and HERCEPTIN® given along with an Aromatase Inhibitor (AI), over single HER2 blockade with HERCEPTIN® given along with an AI, in patients with HER2-positive, Hormone Receptor positive metastatic breast cancer, who experienced disease progression after prior neo(adjuvant)/first-line HERCEPTIN® based chemotherapy. This study also included a third treatment arm of TYKERB® plus an AI, which was compared with the other two treatment groups.
The ALTERNATIVE study is an open-label, phase III trial, in which 355 patients were randomly assigned in a 1:1:1 ratio to receive either TYKERB® along with HERCEPTIN® plus an AI (N=120), HERCEPTIN® plus an AI (N=117) or TYKERB® plus an AI. TYKERB® was administered at 1000 mg orally daily in the dual HER2 blockade group and at 1500 mg orally daily in the TYKERB® plus AI group. Patients receiving TYKERB® were urged to initiate treatment with Loperamide at the onset of diarrhea. HERCEPTIN® was administered IV at a loading dose of 8 mg/kg, followed by the maintenance dose of 6 mg/kg IV every 3 weeks. Physician’s choice of AIs included either Letrozole 2.5 mg, Anastrozole 1 mg or Exemestane 25 mg, orally daily. Enrolled patients were postmenopausal women, with histologically or cytologically confirmed ER-positive and/or Progesterone Receptor-positive, HER2-positive metastatic breast Cancer, as determined in a local laboratory. Prior treatment with endocrine therapy and disease progression during or after a prior HERCEPTIN® based chemotherapy regimen in the neo(adjuvant) setting and/or in the first-line metastatic setting, was a requirement for enrollment in this study. Only one prior regimen in the metastatic setting was allowed. Patients for whom chemotherapy was felt appropriate per treating physician’s judgement, were excluded from the study. Two thirds of the patients had received HERCEPTIN® based regimens in adjuvant setting and approximately one third in metastatic setting. The Primary end point was Progression Free Survival (PFS) with dual HER2 blockade plus AI compared with HERCEPTIN® plus AI. Secondary end points included PFS comparison of other treatment groups, Overall Survival, Overall Response Rate (ORR), Clinical Benefit Rate and Safety.
The study met its Primary end point and the median PFS utilizing dual HER2 blockade with a combination of TYKERB® along with HERCEPTIN® plus an AI was 11 months, compared with 5.7 months for HERCEPTIN® plus an AI (HR=0.62, P=0.0064). This represented a 38% reduction in the risk of disease progression. The PFS benefit was consistently observed in various predefined subgroups of patients. Further, the ORR and Clinical Benefit Rate were superior in the TYKERB® along with HERCEPTIN® plus AI group compared to HERCEPTIN® plus AI group ((31.7% versus 13.7% and 41% versus 31%, respectively). Although survival data were immature, there was also a trend favoring treatment with dual HER2 (median 46 months versus 40 months). When other treatment groups were compared, the median PFS with TYKERB® plus an AI was 8.3 months compared to 5.7 months with HERCEPTIN® plus an AI (HR=0.71, P=0.036), suggesting that among HER2-positive, Hormone Receptor positive metastatic breast cancer patients, who had progressed after prior treatment with HERCEPTIN®, anti-HER2 treatment with TYKERB® along with an AI may be a reasonable alternative, although this hypothesis will need to be confirmed. Common adverse events included higher incidence of diarrhea and rash in the groups treated with TYKERB® and serious adverse events were similar across the three treatment groups. Treatment discontinuation due to adverse events was lower in the dual HER2 blockade group.
The authors concluded that dual HER2 blockade with a combination of TYKERB®, HERCEPTIN® and an Aromatase Inhibitor (AI) resulted in improved PFS compared with HERCEPTIN® plus an AI, among HER2-positive and Hormone Receptor positive metastatic breast cancer patients, who had prior HERCEPTIN® based chemotherapy and endocrine therapy in the neo(adjuvant) and/or first line metastatic setting. This dual HER2 blockade combination offers an effective and safe chemotherapy-sparing alternative treatment regimen for this patient population. Phase III, Randomized Study of Dual Human Epidermal Growth Factor Receptor 2 (HER2) Blockade With Lapatinib Plus Trastuzumab in Combination With an Aromatase Inhibitor in Postmenopausal Women With HER2-Positive, Hormone Receptor–Positive Metastatic Breast Cancer: ALTERNATIVE. Johnston SR, Hegg R, Im S, et al. J Clin Oncol 2017;36:741-748
ASTRO Issues New Updated Clinical Guideline for Whole Breast Radiation Therapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. When it comes to adjuvant radiation therapy in breast cancer, the standard of care has been Whole Breast Irradiation (WBI), administered in Conventional Fractions (CF-WBI) of 180 to 200 cGy daily, to approximately 4500 to 5000 cGy, with or without a tumor bed boost. More recently, with clinical trials supporting the safety and effectiveness of HypoFractionated Whole Breast Irradiation (HF-WBI), the American Society for Radiation Oncology (ASTRO) task force, has issued a new clinical guideline for the use of whole-breast radiation therapy for breast cancer, which replaces the existing ASTRO Whole Breast Irradiation guideline published in 2011. With HypoFractionated Whole Breast Irradiation, patients receive larger doses of radiation over a shorter period of time, typically completing treatment in 3-4 weeks, compared with 5-7 weeks for CF-WBI.
The guideline recommendations were based on a systematic literature review, between January 2009 and May 2016, and created using ASTRO-approved tools, for grading evidence quality and recommendation strength. The ASTRO convened a task force to address 5 key questions focused on dose-fractionation for WBI, indications and dose fractionation for tumor bed boost, and treatment planning techniques for WBI and tumor bed boost. The recommendations are summarized below.
Whole-Breast Irradiation (without irradiation of regional lymph nodes) - Delivery and Dosing
1) Treatment decisions, including radiation techniques (Hypofractionated versus Conventional Fractions) should be a shared decision between patient and physician and should be individualized to each patient.
2) For women with invasive breast cancer receiving WBI with or without inclusion of the low axilla, the preferred dose-fractionation scheme is HF-WBI to a dose of 4000 cGy in 15 fractions or 4250 cGy in 16 fractions.
3) The decision to offer HF-WBI should be independent of age, tumor grade, hormone receptor status, HER2 receptor status, surgical margin status, breast size, breast cancer laterality, chemotherapy received prior to radiation and trastuzumab or endocrine therapy received prior to or during radiation.
4) HF-WBI may be used as an alternative to CF-WBI in patients with DCIS.
5) CF-WBI may be preferred over HFWBI when treating primary breast cancers with rare histologies that are most commonly treated with CF when arising in other parts of the body.
6) In patients with breast augmentation, either HF-WBI or CF-WBI may be used.
Tumor Bed Boost
1) A tumor bed boost is recommended for patients with invasive breast cancer 50 years or younger with any grade tumor, age 51-70 years with high grade tumor, or a positive margin. Omitting a tumor bed boost is suggested in patients with invasive breast cancer who are older than 70 years with hormone receptor-positive tumors of low or intermediate grade, resected with widely negative (2 or more mm) margins.
2) A tumor bed boost may be used for patients with DCIS, 50 years and younger, high grade tumors, or close (less than 2 mm) or positive margins following resection. A tumor bed boost may be omitted for patients with DCIS who are older than 50 years, screen detected tumor, total size 2.5 cm or less, low to intermediate nuclear grade, and have widely negative surgical margins (3 mm or more).
3) The decision to use a tumor bed boost should be based on the clinical indications for a boost and should be independent of whether the patient received Conventional or Hypofractionated WBI.
Preferred Techniques for WBI Treatment Planning
1) Three-dimensional conformal radiotherapy planning with a field-in-field technique is recommended to achieve radiation dose homogeneity and full coverage of the tumor bed.
2) Techniques that incorporate deep inspiration breath hold, prone positioning, and/or heart blocks are recommended to minimize heart dose. Treatment techniques should also minimize dose to the contralateral breast, lung, and other normal tissues.
3) Treatment plans should be individualized after consideration of many factors, including tumor characteristics, patient anatomy and comorbidities.
Radiation therapy for the whole breast: Executive summary of an American Society for Radiation Oncology (ASTRO) evidence-based guideline. Smith BD, Bellon JR, Blitzblau R, et al. DOI: https://doi.org/10.1016/j.prro.2018.01.012
Circulating Tumor Cells Predict Late Recurrence in Early Stage HR-Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. Circulating Tumor Cells (CTCs) are epithelial cells that are shed into the circulation from a primary or metastatic tumor. After being shed, CTCs can remain in the circulation or undergo apoptosis. Evaluation of CTCs during the course of disease has prognostic value. Because of the very low concentrations of CTCs (1 CTC in the background of millions of normal hematopoietic cells) in the peripheral blood, different technologies have been developed that will allow enrichment and detection of these CTCs. One such technology is the CELLSEARCH® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of patients with breast cancer. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK positive, CD45 negative and DAPI positive. In essence, CTC assessment is a real time, peripheral blood evaluation (“Liquid Biopsy”) in breast cancer patients.
In women with Hormone Receptor (HR)-positive breast cancer, late recurrences occurring 5 or more years after diagnosis, account for approximately 50% of recurrences. In a meta-analysis from the EBCTCG (Early Breast Cancer Trialists’ Collaborative Group) group, the 10-year risk of recurrence after 5 years of endocrine therapy was found to be 5% in patients with node negative breast cancer, 10% in patients with 1-3 positive lymph nodes, and 22% in patients with 4-9 positive lymph nodes. Late recurrence in patients with HR-positive breast cancer has therefore become a major concern, as this risk for recurrence and death from breast cancer can be ongoing for at least 20 years after the original diagnosis..
The blood and tumor samples of patients are valuable for research and allow the exploration of possible tumor and host-related factors contributing to recurrence. The Coalition of Cancer Cooperative Groups, a non-profit member organization of the National Cancer Institute-sponsored Cooperative Groups, in 2013, created the North American Breast Cancer Groups (NABCG) Biospecimen Bank for Determinants of Late Relapse in Operable Breast Cancer. The purpose for the collection and analysis of samples is, to improve our understanding of breast cancer, and identify which patients are most likely to benefit from specific therapies. The NABCG Biospecimen Bank remains a unique resource of archived primary and metastatic tumor tissue, blood, and DNA, collected from more than 15,000 women, who participated in two large cooperative group cancer treatment trials, TAILORx and E5103.
The authors in this analysis included 546 patients with HER-2-negative, stage II to stage III breast cancer from the E5103 clinical trial, with no clinical evidence of recurrence between 4.5 to 7.5 years following their initial diagnosis. ECOG-ACRIN clinical trial E5103 is a randomized phase III trial, which evaluated the addition of Bevacizumab (AVASTIN®), a Vascular Endothelial Growth Factor (VEGF) inhibitor, to adjuvant chemotherapy following surgery, in patients with lymph node-positive or high-risk, lymph node-negative breast cancer. All patients had undergone surgery and adjuvant chemotherapy as well as endocrine therapy for at least 5 years, if hormone receptor positive. The CTCs from the blood samples of these patients were measured using the CELLSEARCH® CTC assay. None of the patients had clinical evidence of recurrence at the time of this analysis. More than half of the patients (56%) were 50 years or older, 59% of the patients had a tumor 2 cm or larger, 73% had positive lymph nodes and 65% had Hormone Receptor (HR) positive disease. This Primary endpoint of this study was time to recurrence and the primary focus in this analysis was on the group with Hormone Receptor positive disease.
After a median follow up of 1.6 years, 4.8% of patients had a positive CTC assay result overall (1 or more cells/7.5 mL in the blood). Among those with Hormone Receptor positive breast cancer, 5.1% had a positive CTC assay result whereas positive CTC assay result was noted in 4.1% of patients with hormone receptor negative disease. Of the 353 patients with HR-positive disease in this analysis, 4% had a recurrence whereas only 0.5% with HR-negative disease had a local recurrence. The Hazard Ratio for patients with CTC negative results compared with CTC positive results was 21.7 (P<0.001). A positive CTC assay result was associated with a nearly 21.7 fold increased risk of breast cancer recurrence in patients with HR-positive disease. A positive CTC assay was not associated with recurrence in the HR-negative group. The Positive Predictive Value of a positive CTC assay for recurrence at 2 years, in patients with HR-positive disease was 35%, and the Negative Predictive Value for patients in this cohort was 98%.
It was concluded that detection of positive Circulating Tumor Cells in the blood, 5 years after breast cancer diagnosis, was associated with an increased risk for late recurrence, in women with HR-positive, HER2-negative breast cancer. Sparano JA, O’Neill A, Alpaugh K, et al. Circulating tumor cells and late recurrence of breast cancer. Presented at: 2017 San Antonio Breast Cancer Symposium; December 5-9, 2017; San Antonio, TX. Abstract GS6-03.
Long Term Disease Free Survival Benefit with Adjuvant NERLYNX® in HER2-positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 266,120 new cases of invasive breast cancer will be diagnosed in 2018 and about 40,920 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2 and adjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early breast cancer. Nonetheless, approximately 25% of patients will develop recurrent disease within 10 years following this adjuvant intervention. Extending the duration of adjuvant HERCEPTIN® therapy or adding TYKERB® (Lapatinib), a Tyrosine Kinase Inhibitor that targets HER1 and HER2, has not improved outcomes.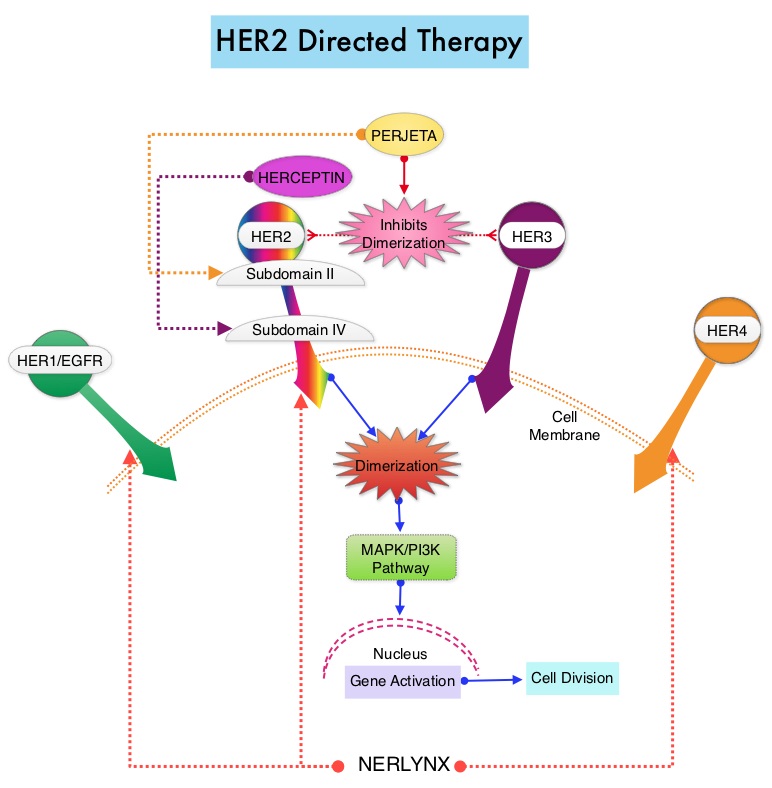
NERLYNX® is a potent, irreversible, oral Tyrosine Kinase Inhibitor, of HER1, HER2 and HER4 (pan-HER inhibitor).NERLYNX® interacts with the catalytic domain of HER1, HER2, and HER4 and blocks their downstream signaling pathways, resulting in decreased cell proliferation and increased cell death. Clinical data has suggested that NERLYNX® has significant activity in suppressing HER-mediated tumor growth and is able to overcome tumor escape mechanisms experienced with current HER2-targeted and chemotherapeutic agents. It has been well known that hormone receptor positive breast cancer patients, who are also HER2-positive, have relative resistance to hormone therapy. Preclinical models had suggested that the addition of NERLYNX® could improve responses in ER positive, HER2-positive breast cancer patients. Further, NERLYNX® has clinical activity in patients with HER2-positive metastatic breast cancer.
ExteNET trial is a multicentre, randomized, double-blind, placebo-controlled, phase III study, in which the efficacy and safety of 12 months of NERLYNX® after HERCEPTIN®-based adjuvant therapy was evaluated, in patients with early stage HER2-positive breast cancer. Patients with early stage HER2-positive breast cancer (N=2,840), and within two years of completing adjuvant HERCEPTIN®, were randomized in a 1:1 ratio to receive either oral NERLYNX® 240 mg per day (N=1420) or placebo (N=1420), for one year. Patients were stratified by hormone receptor status, nodal status (0, 1-3, or 4 or more), and HERCEPTIN® adjuvant regimen (sequentially versus concurrently with chemotherapy). The authors had previously reported that NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly improved 2-year invasive Disease Free Survival.
They now reported updated efficacy outcomes, from a protocol-defined 5-year follow-up sensitivity analysis, and long-term toxicity findings. The predefined endpoint of the 5-year analysis was invasive Disease Free Survival (iDFS), defined as the time between the randomization date to the first occurrence of invasive recurrence (local/regional, ipsilateral or contralateral breast cancer), distant recurrence, or death from any cause. The median follow up was 5.2 years.
It was noted that patients in the NERLYNX® group had significantly fewer invasive Disease Free Survival events, than those in the placebo group (HR=0.73; P=0.0083). The 5-year invasive Disease Free Survival was 90.2% in the NERLYNX® group and 87.7% in the placebo group. The most common grade 3-4 adverse events associated with NERLYNX® were diarrhea, vomiting and nausea. Patients can experience diarrhea early, in the first 2 or 3 days and this can be alleviated using antidiarrheal prophylaxis with Loperamide, initiated with the first dose of NERLYNX® and continued for the first 2 months of treatment and as needed thereafter. There was no evidence of long-term toxicities associated with NERLYNX®, when compared to placebo.
It was concluded that at a median follow up of 5 years, NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly reduced the proportion of clinically relevant breast cancer relapses that might lead to death, such as distant and locoregional recurrences outside the preserved breast. This benefit was seen without increase in the risk of long term toxicity. Overall Survival data will be available at a later date. NERLYNX® is the first TKI approved by the FDA, shown to reduce the risk for disease recurrence, in patients with early stage HER2-positive breast cancer. Neratinib after trastuzumab-based adjuvant therapy in HER2-positive breast cancer (ExteNET): 5-year analysis of a randomised, double-blind, placebo-controlled, phase 3 trial. Martin M, Holmes FA, Ejlertsen B, et al. for the ExteNET Study Group. Lancet Oncol 2017;18:1688-1700
FDA Approves LYNPARZA® for Germline BRCA-Mutated Metastatic Breast Cancer
SUMMARY: The FDA on January 12, 2018, granted regular approval to LYNPARZA® (Olaparib), a Poly ADP-Ribose Polymerase (PARP) inhibitor, for the treatment of patients with deleterious or suspected deleterious germline BRCA-mutated (gBRCAm), HER2-negative metastatic breast cancer, who have been treated with chemotherapy either in the neoadjuvant, adjuvant, or metastatic setting. This is the first FDA-approved treatment for patients with gBRCAm HER2-negative metastatic breast cancer. Patients with Hormone Receptor (HR) positive breast cancer should have been treated with a prior endocrine therapy or be considered inappropriate for endocrine treatment. Patients must be selected for therapy based on an FDA-approved companion diagnostic for LYNPARZA®.
Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 266,120 new cases of invasive breast cancer are expected to be diagnosed in 2018 and about 40,920 women are expected to die of the disease. DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. Mutations in BRCA1 and BRCA2 account for about 20 to 25 percent of hereditary breast cancers and about 5 to 10 percent of all breast cancers. These mutations can be inherited from either of the parents and a child has a 50 percent chance of inheriting this mutation and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal.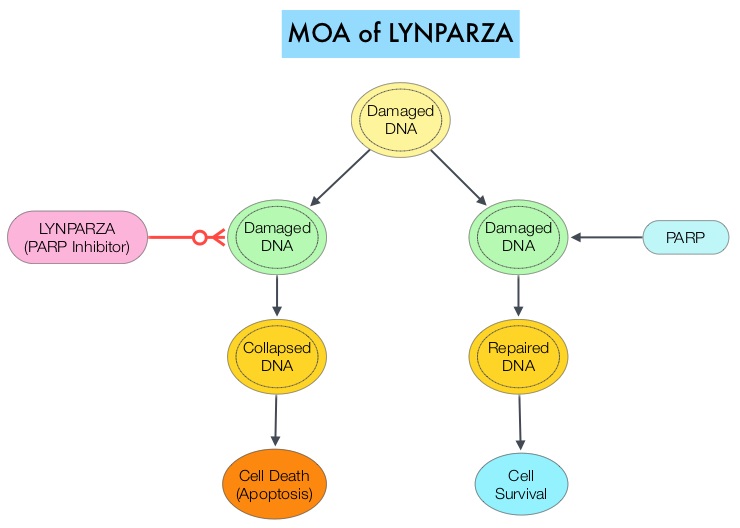
The PARP (Poly ADP Ribose Polymerase) family of enzymes, which include PARP1 and PARP2 repair damaged DNA. LYNPARZA® is a PARP enzyme inhibitor that causes cell death in tumors that already have a DNA repair defect, such as those with BRCA1 and BRCA2 mutations. The FDA approved LYNPARZA® in 2014 as monotherapy for the treatment of patients with deleterious or suspected deleterious germline BRCA mutated advanced ovarian cancer.
The present approval was based on data from OlympiAD, a randomized, open-label, phase III study that evaluated the efficacy and safety of LYNPARZA® compared with physician’s choice of standard single agent chemotherapy, in patients with HER2-negative metastatic breast cancer, with inherited, germline BRCA mutations. In this study, 302 patients were randomized in a 2:1 ratio to receive LYNPARZA® tablets 300 mg PO BID (N=205) or physician’s choice of standard chemotherapy (N=97). The later included 21-day cycles of either XELODA® (Capecitabine) 2500 mg/m2 orally on days 1-14, NAVELBINE® (Vinorelbine) 30 mg/m2 IV days 1 and 8 or HALAVEN® (Eribulin)1.4 mg/m2 IV days 1 and 8. Treatment was continued until disease progression or unacceptable toxicity. The median age was 44 years, and all patients had received prior chemotherapy in the neoadjuvant, adjuvant, or metastatic setting. Patients in this study were stratified based on prior use of chemotherapy for metastatic disease, Hormone Receptor status (HR positive versus triple negative), and previous use of platinum-based chemotherapy. The Primary endpoint was Progression Free Survival (PFS). Secondary endpoints included Overall Survival (OS), time to second progression or death, Objective Response Rate (ORR) and effect on health-related Quality of Life.
At a median follow up of about 14 months, the median PFS was 7 months in the LYNPARZA® group versus 4.2 months with standard chemotherapy (HR=0.58; P=0.0009), suggesting a 42% reduced risk of cancer progression in the LYNPARZA® group compared to those who received chemotherapy. Following disease progression, the time to second progression (which meant duration of time before the cancer worsened again), was also longer in the LYNPARZA® group (HR 0.57), suggesting that recurrent disease was not more aggressive following progression on LYNPARZA®. The ORR was 60% and 29% in LYNPARZA® and chemotherapy groups respectively. Severe side effects were more common in chemotherapy treated patients (50%) compared with LYNPARZA® group (37%). The most common side effects in the LYNPARZA® group included nausea, fatigue and cytopenias, whereas rash on hands and feet were most common in the chemotherapy group.
The authors concluded that LYNPARZA® monotherapy significantly improved Progression Free Survival in HER2-negative metastatic breast cancer patients, with inherited germline BRCA mutations, compared to standard chemotherapy. This “proof of the principle” study demonstrated that breast cancers with defects in a specific DNA damage repair pathway are sensitive to targeted therapy and this is the first of several phase III studies with PARP inhibitors that are underway. OlympiAD: Phase III trial of olaparib monotherapy versus chemotherapy for patients (pts) with HER2-negative metastatic breast cancer (mBC) and a germline BRCA mutation (gBRCAm). Robson ME, Im S-A, Senkus E, et al. J Clin Oncol 35, 2017 (suppl; abstr LBA4).
Hormonal Contraception Increases Breast Cancer Risk
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease. It is estimated that approximately 140 million women worldwide use hormonal contraception and this number accounts for approximately 13% of women between the ages of 15 and 49 years. The use of hormonal contraception has been on the rise.
Estrogen promotes the development of breast cancer. Previous published studies had shown positive associations between the use of oral contraceptives and breast cancer risk, but the results have been inconsistent from no increase in risk to a 20-30% increase in risk. Further, unlike contemporary hormonal contraception, oral contraceptives in the past contained a higher estrogen dose in the combined estrogen/progestin hormonal contraceptives, and the higher dose of estrogen has been implicated in the development of breast cancer. Contemporary products with new progestins and new routes of delivery (intrauterine system, contraceptive patches, vaginal rings, progestin-only implants, and injections) has raised new concerns, with some studies suggesting that the addition of progestin appears to increase the risk of breast cancer among postmenopausal women who receive hormone therapy. There is evidence to suggest that use of hormonal contraception at a young age may confer a higher risk of breast cancer than initiation of use later in life.
This Danish study, using their national registry, reported the risk of invasive breast cancer risk among women of reproductive age, who were using currently available hormonal contraception. This prospective cohort study conducted in Denmark included 1.8 million women between 15-49 years of age, who did not have a diagnosis of cancer or venous thromboembolism, and who had not received treatment for infertility. Individually updated information about the use of hormonal contraception, breast cancer diagnoses, and potential contributing factors, was obtained from nationwide registries.
It was noted that when compared with women who had never used hormonal contraception, the relative risk of breast cancer among all current and recent users of hormonal contraception was 1.20 (20% higher than average). This risk increased from 1.09 (9% higher than average) with less than 1 year of use to 1.38 (38% higher than average) with more than 10 years of hormonal contraception use (P=0.002). After discontinuation of hormonal contraception, the risk of breast cancer continued to be higher among the women who had used hormonal contraceptives for 5 years or more than among women who had not used hormonal contraceptives. Women who currently or recently used the progestin-only intrauterine system also had a higher risk of breast cancer than women who had never used hormonal contraceptives, with a relative risk of 1.21 (21% higher than average). Outcomes in this study however, could not be adjusted for age at menarche, breast feeding, alcohol consumption, physical activity or Body Mass Index.
It was concluded from this large study population that, the risk of breast cancer was higher among women who currently or recently used contemporary hormonal contraceptives than among women who had never used hormonal contraceptives, and this risk increased with longer durations of use. Additionally, these results unequivocally suggest that no hormonal contraceptives are free of risk. Contemporary Hormonal Contraception and the Risk of Breast Cancer. Mørch LS, Skovlund CW, Hannaford PC, et al. N Engl J Med 2017; 377:2228-2239
Long Term Risk of Breast Cancer Recurrence after Stopping Endocrine Therapy at 5 Years
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease.
It has been well established that treatment with 5 years of endocrine therapy in early stage, Estrogen Receptor (ER) positive breast cancer, significantly reduces the risks of locoregional and distant recurrence, contralateral breast cancer, death from breast cancer, and therefore death from any cause. Extended adjuvant endocrine therapy with either Tamoxifen or an Aromatase Inhibitor (AI) beyond 5 years can further reduce breast cancer recurrence. This however can result in treatment related side effects. Therefore, when considering extended adjuvant endocrine therapy beyond 5 years, the potential benefits should be weighed against the associated risk with such therapy. The absolute benefit of continuing endocrine therapy after 5 years depends on the absolute risk of later recurrence, if patient’s receives no further therapy.
The authors in this publication reported the influence of original tumor characteristics, on the incidence of breast cancer outcomes, over a 20 year period. This meta-analysis included data from 88 trials in the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) database of randomized trials, involving 62,923 women with ER-positive breast cancer, who were disease-free after 5 years of adjuvant endocrine therapy. Data was analyzed from women who had T1 disease (Tumor diameter 2 cm or less) or T2 disease (Tumor diameter more than 2 cm up to 5 cm), no positive nodes (N0), 1-3 positive nodes (N1-3), 4-9 positive nodes (N4-9), and no distant metastases. The authors then assessed the associations of Tumor diameter and Nodal status (TN), tumor grade, and other factors with patients’ outcomes, during the period from 5 to 20 years.
It was noted that distant breast cancer recurrences occurred at a steady rate for at least another 15 years after completing 5 years of adjuvant hormonal treatment. The risk of distant recurrence strongly correlated with the original Tumor diameter and Nodal status (TN status).
Among women with T1 disease, the risk of distant recurrence was 13% in those with T1N0 disease, 20% in those with T1N1-3 disease and 34% in those with T1N4-9 disease. Even in those women with low grade T1N0 breast cancer, the absolute risk of distant recurrence from 5-20 years was 10%.
Among those with T2 disease, the risk of distant recurrence was 19% with T2N0 disease, 26% with T2N1-3 disease, and 41% with T2N4-9 disease. TN status was also a strong determinant of locoregional recurrence, but not predictive for contralateral breast cancer.
There was a strong association of tumor grade and Ki-67 status with the risk of distant recurrence during the first 5 years but had limited additional prognostic relevance during years 5-20. Similarly, patients with negative Progesterone Receptor (PR) had a worse prognosis during the first 5 years but not thereafter.
The authors concluded that even after 5 years of adjuvant endocrine therapy, women with ER-positive, early stage breast cancer, continue to be at risk for recurrence and death from breast cancer, for at least 20 years after the original diagnosis. This risk varies from 10-41% and is strongly dependent on TN status at the time of initial diagnosis. Even those with low-grade T1N0 disease were at an appreciable risk of distant recurrence. Extended adjuvant endocrine therapy beyond 5 years may reduce this risk, and this study highlights the need for new approaches to reduce late recurrence. 20-Year Risks of Breast-Cancer Recurrence after Stopping Endocrine Therapy at 5 Years. Pan H, Gray R, Braybrooke J, et al. for the EBCTCG. N Engl J Med 2017; 377:1836-1846
Circulating Tumor Cells in the Peripheral Blood May Predict Outcomes in Breast Cancer
SUMMARY: Circulating Tumor Cells (CTCs) are epithelial cells that are shed into the circulation from a primary or metastatic tumor. After being shed, CTCs can remain in the circulation or undergo apoptosis. Evaluation of CTCs during the course of disease has prognostic value. Because of the very low concentrations of CTCs (1 CTC in the background of millions of normal hematopoietic cells) in the peripheral blood, different technologies have been developed that will allow enrichment and detection of these CTCs. One such technology is the CellSearch® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of patients with breast cancer. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK positive, CD45 negative and DAPI positive. In essence, CTC assessment is a real time, peripheral blood evaluation (“Liquid Biopsy”) in breast cancer patients.
Despite advances in treatment, approximately 30% of patients with node-negative breast cancer and 50% of patients with node-positive breast cancer may relapse within 5 years. This is due to cancer cells shed from primary tumors that migrate to distal sites as Circulating Tumor Cells (CTCs) via the circulatory system. CTCs are therefore precursors of metastatic disease and may not only predict risk of metastatic disease but may also be useful in monitoring treatment efficacy. There have been conflicting reports about the effectiveness of different treatments to reduce CTCs in breast cancer patients, including the different molecular subtypes.
To further address these controversies, the authors conducted a meta-analysis of the published studies which included measurement of CTCs before and after treatment in breast cancer patients, and estimated the benefit of reducing CTC on patient outcomes. Data base searches included 1004 publications and 50 studies conducted between 2009 and 2016 in the US, Europe and Asia. A total of 6712 patients from these studies were eligible for meta-analysis. Enrolled patients had pathologically diagnosed breast cancer, CTCs were detected by any method, including cell capture and quantitative PCR and the patient’s CTC status both pre- and post-therapy was available. The CTC-positive rate was reported using different cut-off values of CTC count and different expression thresholds of epithelial genes (EpCAM, CK18, CK19) using RT-PCR in the various studies.
An overall analysis of the 6712 patients with CTC-positive rate by the random-effects model suggested that treatment intervention significantly decreased CTC-positive rate compared to the baseline (Relative Risk (RR)=0.68, P<0.00001, which meant a 32% reduction in the Relative Risk, compared to baseline values.
Subgroup analyses revealed that when compared to pre-treatment, CTCs were decreased after neoadjuvant treatment (RR=0.65, P=0.006), adjuvant treatment (RR=0.89, P=0.10), treatment in metastatic setting (RR=0.59, P<0.00001) and the combination therapy (RR=0.78, P=0.03). Reduction in CTCs was not seen after surgery (RR=1.27, P=0.42), suggesting that local intervention with surgery does not eliminate CTCs and patients with positive CTCs should receive other therapies after surgery, to decrease the risk of recurrence.
When compared to pre-treatment levels, treatment resulted in significant reduction in CTCs in HER2-positive patients (RR=0.68, P<0.0001) and HER2-negative patients (RR=0.52, P=0.01). This reduction in CTCs was however not noted in patients with triple-negative breast cancer patients (RR=0.38, P=0.29), indicating that current therapies for this group is inadequate and should be further optimized with newer therapies.
More importantly, reduction in CTCs was associated with lower probability of disease progression (P=0.01), longer Progression Free Survival (P<0.0001) and longer Overall Survival (P<0.00001).
It was concluded that based on this large meta-analysis, CTCs can help monitor the effectiveness of treatment and guide subsequent therapies in breast cancer patients. Circulating tumor cell status monitors the treatment responses in breast cancer patients: a meta-analysis. Yan W-T, Cui X, Chen Q, et al. Sci. Rep. 7, 43464; doi: 10.1038/srep43464 (2017).
Targeting AKT Improves Outcomes in a Subset of Triple Negative Breast Cancer Patients
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease. Triple Negative Breast Cancer (TNBC) is a heterogeneous, molecularly diverse group of breast cancers and are ER (Estrogen Receptor), PR (Progesterone Receptor) and HER2 (Human Epidermal Growth Factor Receptor 2) negative. TNBC accounts for 15% to 20% of invasive breast cancers, with a higher incidence noted in young patients. It is usually aggressive, and tumors tend to be high grade and patients with TNBC are at a higher risk of both local and distant recurrence. Those with metastatic disease have one of the worst prognoses of all cancers with a median Overall Survival of 13 months. The majority of patients with TNBC who develop metastatic disease do so within the first 3 years after diagnosis, whereas those without recurrence during this period of time have survival rates similar to those with ER-positive breast cancers. The lack of known recurrent oncogenic drivers in patients with metastatic TNBC, presents a major therapeutic challenge. Nonetheless, patients with TNBC often receive chemotherapy in the neoadjuvant, adjuvant or metastatic settings and approximately 30-40% of patients achieve a pathological Complete Response (pCR) in the neoadjuvant setting. Those who do not achieve a pathological Complete Response tend to have a poor prognosis. It therefore appears that there are subsets of patients with TNBC who may be inherently insensitive to cytotoxic chemotherapy. Three treatment approaches appear to be promising and they include immune therapies, PARP inhibition and inhibition of PI3K pathway.
Using gene expression profiling, TNBC can be classified into 4 distinct molecular subtypes- two Basal-Like (BL1, BL2), Mesenchymal type (M) and Luminal Androgen Receptor type (LAR). BL1 molecular subtype of TNBCs are characterized by high levels of expression of genes involved in the cell cycle and DNA-damage repair pathways and accounts for up to 18% of TNBCs. These tumors are more sensitive to therapies targeting the DNA-repair pathways such as platinum based chemotherapy and Poly-ADP Ribose Polymerase (PARP) inhibition. BL2 molecular subtype of TNBCs represent 13% of TNBCs and in contrast are characterized by upregulation of growth factor signaling pathways, including the Epidermal Growth Factor (EGF), MET pathways, as well as genes involved in glycolysis and gluconeogenesis. These tumors may better respond to small molecule inhibitors of growth factor pathways. An alternate classification of Basal-Like subtype includes Basal-Like Immune Suppressed (BLIS) which is associated with downregulation of B cell, T cell, and Natural Killer cell immune-regulating pathways, and has the worse prognosis and Basal-Like Immune Activated (BLIA) subtype, which has the best prognosis due to upregulated immune-associated pathways. Mesenchymal TNBCs constitute approximately 10-30% of TNBC tumors and are associated with aberrations in the PI3K/AKT/ mTOR pathway as well as increased angiogenesis and may benefit from agents targeting these pathways. Metaplastic breast cancer belongs to this TNBC group. The Luminal Androgen Receptor (LAR) subtype accounts for approximately 11% of TNBCs. These tumors have a high expression of Androgen Receptor by IHC (Immuno HistoChemistry) and benefit from Androgen Receptor blockade and do not respond to cytotoxic chemotherapy.
Ipatasertib is a highly selective oral ATP-competitive, small-molecule, AKT inhibitor and sensitivity to Ipatasertib has been associated with high levels of phosphorylated AKT, PTEN protein loss or genetic mutations in PTEN, and PIK3CA mutations. KRAS and BRAF mutations are typically associated with resistance to Ipatasertib. It is estimated that approximately 50% of TNBCs have deficient expression of the tumor suppressor PTEN, which is associated with a higher degree of AKT pathway activation Preclinical studies showed synergy between Ipatasertib and Taxanes. Because of the high prevalence of PI3K/AKT pathway activation in TNBCs, the authors in this study evaluated the benefit of a combination of Ipatasertib and Paclitaxel as first line therapy, for TNBC.
The LOTUS trial is a randomized, placebo controlled, double blind, phase II study in which 124 treatment naïve patients with inoperable, locally advanced or metastatic Triple Negative Breast Cancer were randomly assigned (1:1) to receive Paclitaxel 80 mg/m2 IV Days 1, 8, 15 of a 28 day cycle in combination with either Ipatasertib 400 mg PO daily (N=62) or placebo (N=62), administered on days 1-21 of each 28 day cycle. Treatment was continued until disease progression or unacceptable toxicity. Patients in this study were stratified based on expression of the PTEN tumor suppressor gene and alteration of PIK3CA/AKT1/PTEN in their tumors. The co-primary endpoints were Progression Free Survival (PFS) in the intent-to-treat population and Progression Free Survival in the PTEN-low population. Secondary endpoints included Objective Response Rate and Duration of Response. The median follow up was 10.3 months.
It was noted that the median PFS in the intent-to-treat population was 6.2 months with Ipatasertib versus 4.9 months with placebo (HR=0.60; P=0.037). In the 48 patients with low PTEN expression tumors, the median PFS however was 6.2 months with Ipatasertib and 3.7 months with placebo (HR=0.59; P=0.18), and this was not statistically significant. The PFS benefit was more pronounced in the patient group with PIK3CA/AKT1/PTEN-altered tumors, with a median PFS of 9.0 months in the Ipatasertib group versus 4.9 months in the placebo group (HR=0.44; P=0.041). The most common grade 3/4 adverse events in the Ipatasertib group were diarrhea and neutropenia.
The authors concluded that Ipatasertib prolonged Progression Free Survival compared to placebo and is the first study supporting AKT-targeted therapy for Triple Negative Breast Cancer, supporting the use of gene expression profiling in this heterogeneous malignancy. Ipatasertib plus paclitaxel versus placebo plus paclitaxel as first-line therapy for metastatic triple-negative breast cancer (LOTUS): a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Kim S, Dent R, Im S, et al on behalf of the LOTUS investigators. The Lancet Oncology 2017;18:1360-1372
African American Women with Benign Breast Disease and Triple Negative Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease. Benign Breast Disease (BBD) is an important risk factor for the later development of breast cancer in either breast. With the routine use of mammography, the identification of BBD has become more common, and this has in turn enabled the calculation of breast cancer risk. The Breast Cancer Risk Assessment Tool which is an interactive tool designed by researchers at the National Cancer Institute (NCI) and the National Surgical Adjuvant Breast and Bowel Project (NSABP) to estimate a woman's risk of developing invasive breast cancer, utilizes the number of breast biopsies and atypical hyperplasia, in addition to other factors, to estimate women’s risk of developing breast cancer. Benign Breast Disease (BBD) encompasses a variety of histologic entities. FibroCystic Changes (FCCs) constitute the most frequent benign disorder of the breast. FCCs may be multifocal and bilateral and women usually present with symptoms of breast pain and tender nodularities in breasts. Hormonal imbalance, with estrogen predominance over progesterone, has been implicated as an important contributing factor in its development. Fibrocystic changes are usually subdivided into nonproliferative lesions, proliferative lesions without atypia, and proliferative lesions with atypia (atypical hyperplasia). Proliferative or atypical lesions are associated with an increased risk of breast cancer. However a significant majority of breast biopsies (about 70%) show nonproliferative lesions.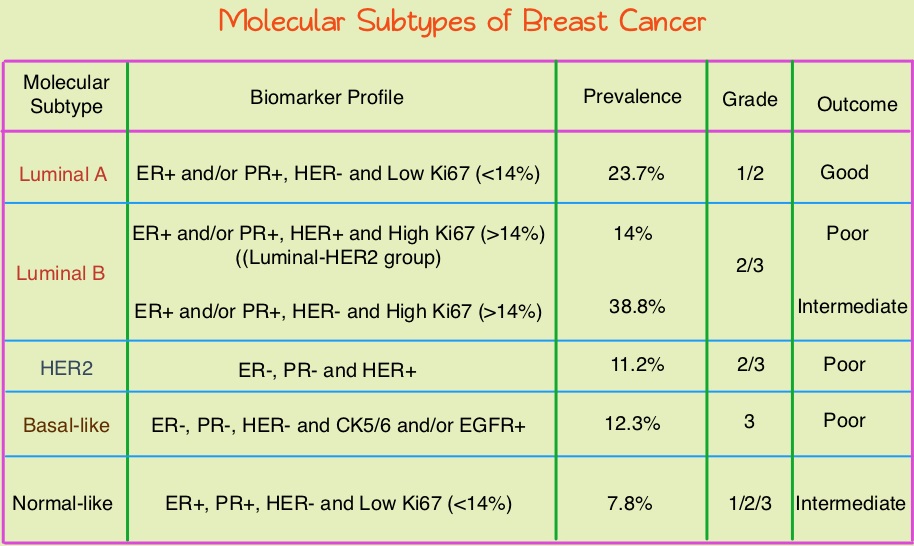
Breast cancer is heterogeneous malignancy and using global gene expression analyses, 6 breast cancer intrinsic subtypes have been established. They include Luminal A, Luminal B, HER2-enriched, Claudin-low, Basal-like, and a Normal breast-like group. Triple Negative Breast Cancer (TNBC) accounts for 12–24% of all breast carcinomas and is negative for Estrogen Receptor (ER), Progesterone Receptor (PR), and HER2. There is a 2 fold increase in the incidence of TNBC among African American (AA) women compared with White American (WA) women and is a surrogate for the inherently aggressive Basal-like breast cancer subtype. This group has the worse prognosis compared to other breast cancer subtypes. Basal-like breast cancer sub-type is also a marker of hereditary breast cancer susceptibility. Multiparity may increase the risk of TNBC and reduce the likelihood of developing ER-positive breast cancer. Triple Negative Breast Cancer (TNBC) compared with non-TNBC, likely arises from different pathogenetic pathways. NF-κB pathway, which controls immune response, angiogenesis, the cell cycle, extracellular matrix degradation, and apoptosis, may represent a key regulator of TNBC. Activation of PI3K/AKT pathway in turn, has been implicated in oncogenic transformation.
Benign breast disease (BBD) has been associated with increased risk for ER positive/non-TNBC. The purpose of this study was to determine whether African American (AA) identity is associated with TNBC among a cohort of both AA and White American (WA) women, who were initially diagnosed with BBD. The authors in this study conducted a retrospective analysis of a cohort comprising 2588 African American women and 3566 White American women aged between 40 and 70 years, with a biopsy-proven Benign Breast Disease. This data was obtained from the Pathology Information System of Henry Ford Health System (HFHS), an integrated multihospital and multispecialty health care system in Detroit, Michigan. These individuals had breast biopsies performed between January 1, 1994, and December 31, 2005 and data analysis was performed from November 1, 2015, to June 15, 2016. Patients with prior breast cancer and those with breast cancer diagnosed within 6 months of BBD biopsy were excluded. Benign Breast Disease was classified as fibrocystic/proliferative/hyperplasia without atypia, with atypia, or with lobular carcinoma in situ. The mean follow up was 10.2 years for both AA patients and WA patients.
It was noted that more than 75% of the subsequent breast cancers in each subset were Ductal Carcinoma in Situ (DCIS) or Stage I. Among the African American (AA) patients who developed subsequent invasive breast cancer, 24.2% developed TNBC compared with 7.4% of the White American (WA) patients who developed subsequent invasive breast cancers (P=0.01). The 10 year probability estimate for developing TNBC was 0.56% for AA patients and 0.25% for WA patients.
The authors concluded that in this largest analysis to date of TNBC and its relationship to racial/ethnic identity and Benign Breast Disease as risk factors, African American identity persisted as a significant risk factor for Triple Negative Breast Cancer, suggesting that African American identity is associated with inherent susceptibility for TNBC pathogenetic pathways. This important finding should be taken into consideration, when discussing chemoprevention strategy among African American women with Benign Breast Diseases. Association Between Benign Breast Disease in African American and White American Women and Subsequent Triple-Negative Breast Cancer. Newman LA, Stark A, Chitale D, et al. JAMA Oncol. 2017;3:1102-1106.
FDA Approves VERZENIO® for Hormone Receptor Positive, HER2-Negative Breast Cancer
SUMMARY: The FDA on September 28, 2017, approved VERZENIO® (Abemaciclib) in combination with FASLODEX® (Fulvestrant) for women with Hormone Receptor positive (HR-positive), HER2-negative, advanced or metastatic breast cancer, with disease progression following endocrine therapy. In addition, VERZENIO® was approved as monotherapy for women and men with HR-positive, HER2-negative advanced or metastatic breast cancer with disease progression following endocrine therapy and prior chemotherapy in the metastatic setting. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease.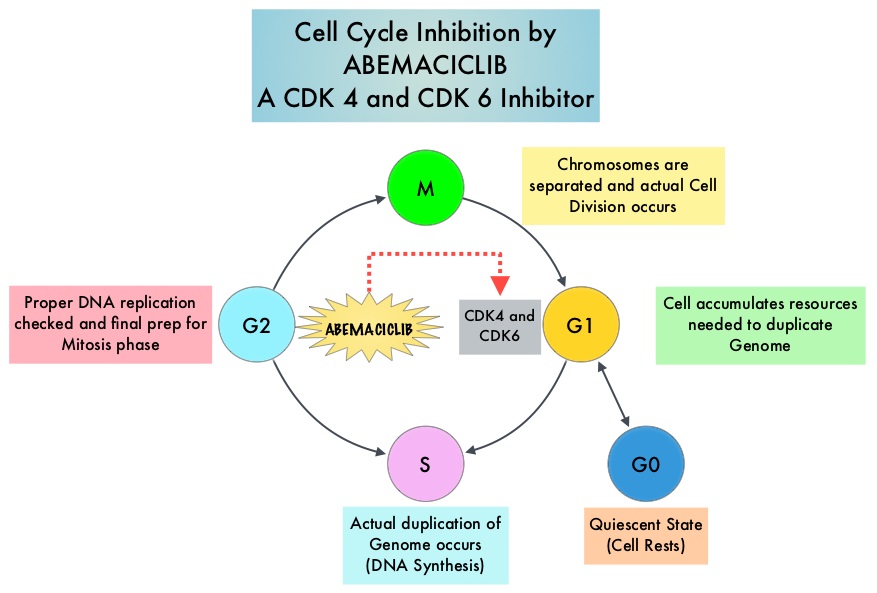
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
VERZENIO® is an oral, selective inhibitor of CDK4 and CDK6 kinase activity, and prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. VERZENIO® is structurally distinct from other CDK 4 and 6 inhibitors (such as Ribociclib and Palbociclib) and is 14 times more potent against cyclin D1/CDK 4 and cyclin D3/CDK 6, in enzymatic assays. VERZENIO® in the phase I trials was noted to be active in HR-positive, metastatic breast cancer, as monotherapy as well as in combination with FASLODEX®. Based on this preliminary data, two clinical trials, MONARCH 1 and MONARCH 2, were conducted.
The approval of VERZENIO® as monotherapy in HR-positive, metastatic breast cancer was based on MONARCH 1, which is a a single-arm, open-label, phase II, multicenter study, which enrolled women with measurable HR-positive, HER2-negative metastatic breast cancer, whose disease progressed during or after endocrine therapy, had received a taxane in any setting, and who received one or two prior chemotherapy regimens in the metastatic setting. This trial included 132 patients who received VERZENIO® 200 mg orally twice daily on a continuous schedule, until disease progression. The Objective Response Rate was 19.7%, with a median response duration of 8.6 months.
The approval of VERZENIO® in combination with FASLODEX® was based on MONARCH 2, which is an international, double-blind, phase III study in which 669 patients were randomized in a 2:1 ratio to receive either VERZENIO® plus FASLODEX® (N=446) or placebo plus FASLODEX® (N=223). Enrolled patients had HR-positive, HER2-negative metastatic breast cancer, with disease progression while receiving neoadjuvant or adjuvant endocrine therapy, within 12 months of adjuvant endocrine therapy, or while receiving endocrine therapy for metastatic breast cancer. Patients must not have received more than one endocrine therapy or any prior chemotherapy for metastatic breast cancer. Randomized patients received either VERZENIO® 150 mg or placebo orally twice daily plus FASLODEX® 500 mg IM on Day 1 and Day 15 of cycle 1 and then on Day 1 of cycle 2 and beyond (28-day cycles). Treatment was continued until disease progression or unmanageable toxicities. The mean patient age was 60 years, 82% of patients were postmenopausal, 72% had measurable disease, 56% had visceral disease, and 25% had primary endocrine therapy resistance. About 60% of patients had received chemotherapy in the adjuvant or neoadjuvant setting and 69% of the patients had prior therapy with Aromatase Inhibitors (AI). The Primary end point was Progression Free Survival (PFS), and Secondary end points included Overall Survival (OS), Objective Response Rate (ORR), Duration of Response, Clinical Benefit Rate, Quality of Life, and safety.
The median PFS for the group receiving VERZENIO® plus FASLODEX® was 16.4 months compared with 9.3 months for those taking placebo with FASLODEX® (HR= 0.55; P<0.0001). In patients with measurable disease, the Objective Response Rate for the group receiving VERZENIO® plus FASLODEX® was 48.1% compared to 21.3% in the placebo with FASLODEX® treated patients. The most common adverse events in the VERZENIO® versus placebo groups were diarrhea neutropenia, nausea and fatigue.
It was concluded that a combination of VERZENIO® plus FASLODEX® significantly improved Progression Free Survival and Objective Response Rates, with a tolerable safety profile, in patients with Hormone Receptor-positive and HER 2-negative metastatic breast cancer who progressed while receiving endocrine therapy. MONARCH 2: Abemaciclib in Combination With Fulvestrant in Women With HR+/HER2− Advanced Breast Cancer Who Had Progressed While Receiving Endocrine Therapy. Sledge GW, Toi M, Neven P, et al. DOI: 10.1200/JCO.2017.73.7585 Journal of Clinical Oncology 35, no. 25 (September 2017) 2875-2884.
MONARCH 1: Results from a phase II study of abemaciclib, a CDK4 and CDK6 inhibitor, as monotherapy, in patients with HR+/HER2- breast cancer, after chemotherapy for advanced disease. Dickler MN, Tolaney SM, Rugo HS, et al. J Clin Oncol 34, 2016 (suppl; abstr 510).
Screening Mammography Starting at Age 40 years May Reduce Breast Cancer Deaths by 40 percent
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. It is estimated that 252,710 new cases of invasive breast cancer and 63,410 new cases of non-invasive breast cancer will be diagnosed in women in 2017 and 40,610 women are expected to die from the disease. In the US, about 33 million screening mammograms are performed each year.
Currently, the major national health care organizations in the US have different recommendations for screening mammography which has led to some confusion and emotional counterarguments. These several different recommendations include 1) Annual screening at ages 40 to 84 years 2) Annual screening at ages 45 to 54 years and then biennially at ages 55 to 79 years 3) Biennial screening at ages 50 to 74 years.
To address this varied recommendations and help women make informed decisions regarding mammography screening, the authors used computer modeling (CISNET models) to assess the three major screening mammography recommendations, and estimate the number of breast cancer deaths that might be prevented with the different screening mammography schedules. Cancer Intervention and Surveillance Modeling Network (CISNET) is a consortium of NCI-sponsored investigators who use statistical modeling to improve understanding of cancer control interventions in prevention, screening and treatment, and their effects on population trends in incidence and mortality. CISNET has been cited by the International Society Pharmacoeconomics and Outcomes Research (ISPOR) Task Force on Good Modeling Practices for its role in establishing a forum that enables researchers to compare results and articulate reasons for discrepancies.
It was noted in this study that the mean mortality reduction in breast cancer-specific deaths was greatest with the recommendation of annual screening at ages 40 to 84 years (39.6%), which meant that 29,369 lives were saved from breast cancer, compared with the recommendation of screening annually at ages 45 to 54 years, then biennially at ages 55 to 79 years (30.8%), which meant that 22,829 were lives saved from breast cancer, and the recommendation of biennial screening at ages 50 to 74 years (23.2%) which meant that 17,153 lives were saved from breast cancer.
The study also took into consideration risks associated with screening, including callbacks for additional imaging following indeterminate or suspicious mammographic finding and in some cases, a breast biopsy, only to find out that the findings were benign. The authors commented that the average woman in her 40s getting annual screening can expect additional and unnecessary screening about once every 12 years and unnecessary breast biopsy recommendations once every 150 years. Other rare risks with screening mammography include breast cancer that could be missed and breast cancer caused by mammogram radiation.
It was concluded that based on the CISNET models, the greatest breast cancer-specific mortality reduction is achieved with annual screening of women starting at age 40 years. They added that this is the first study to compare the three most widely discussed recommendations for screening mammography, head to head. These findings will guide women and their Health Care Providers in deciding when to begin screening mammography and how often to get screened. Comparison of recommendations for screening mammography using CISNET models. Arleo EK, Hendrick E, Helvie MA, et al. CANCER; Published Online: August 21, 2017. http://doi.wiley.com/10.1002/cncr.30842
FDA Approves NERLYNX® for Adjuvant Treatment of HER2 Positive Breast Cancer
SUMMARY: The FDA on July 17, 2017 approved NERLYNX® (Neratinib) for the extended adjuvant treatment of adult patients with early stage HER2-overexpressed/amplified breast cancer, to follow adjuvant Trastuzumab (HERCEPTIN®)-based therapy. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2 and adjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early breast cancer. Nonetheless, approximately 25% of patients will develop recurrent disease within 10 years following this adjuvant intervention. Extending the duration of adjuvant HERCEPTIN® therapy or adding TYKERB® (Lapatinib), a Tyrosine Kinase Inhibitor that targets HER1 and HER2, has not improved outcomes.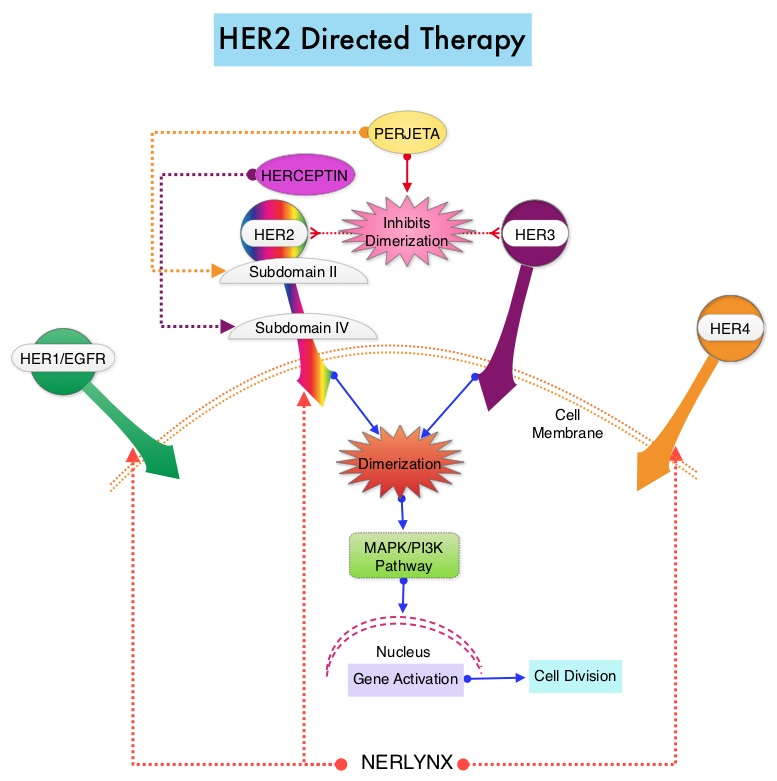
NERLYNX® is a potent, irreversible, oral Tyrosine Kinase Inhibitor, of HER1, HER2 and HER4 (pan-HER inhibitor). NERLYNX® interacts with the catalytic domain of HER1, HER2, and HER4 and blocks their downstream signaling pathways, resulting in decreased cell proliferation and increased cell death. Clinical data has suggested that NERLYNX® has significant activity in suppressing HER-mediated tumor growth and is able to overcome tumor escape mechanisms experienced with current HER2-targeted and chemotherapeutic agents. It has been well known that hormone receptor positive breast cancer patients, who are also HER2-positive, have relative resistance to hormone therapy. Preclinical models had suggested that the addition of NERLYNX® could improve responses in ER positive, HER2-positive breast cancer patients. Further, NERLYNX® has clinical activity in patients with HER2-positive metastatic breast cancer.
The approval of NERLYNX® was based on ExteNET trial, which is a multicentre, randomized, double-blind, placebo-controlled, phase III study, in which the efficacy and safety of 12 months of NERLYNX® after HERCEPTIN®-based adjuvant therapy was evaluated, in patients with early stage HER2-positive breast cancer. Patients with early stage HER2-positive breast cancer (N=2,840), and within two years of completing adjuvant HERCEPTIN®, were randomized in a 1:1 ratio to receive either oral NERLYNX® 240 mg per day (N=1420) or placebo (N=1420), for one year. Patients were stratified by hormone receptor status, nodal status (0, 1-3, or 4 or more), and HERCEPTIN® adjuvant regimen (sequentially versus concurrently with chemotherapy). The Primary endpoint was invasive Disease Free Survival (iDFS), defined as the time between the randomization date to the first occurrence of invasive recurrence (local/regional, ipsilateral or contralateral breast cancer), distant recurrence, or death from any cause, within two years of follow up. The median follow up was 2 years.
In the updated analysis, the two year iDFS was 94.2% in patients treated with NERLYNX® compared with 91.9% in those receiving placebo (HR 0.66; P=0.008). Patients with ER positive breast cancer were noted to have greater benefit. The most common grade 3-4 adverse events associated with NERLYNX® were diarrhea, vomiting and nausea. Patients can experience diarrhea early, in the first 2 or 3 days and this can be alleviated using antidiarrheal prophylaxis with Loperamide, initiated with the first dose of NERLYNX® and continued for the first 2 months of treatment and as needed thereafter.
It was concluded that NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly improved 2-year invasive Disease Free Survival. Longer follow up will hopefully address if there is an Overall Survival benefit with this treatment intervention. NERLYNX® is the first TKI approved by the FDA, shown to reduce the risk for disease recurrence, in patients with early stage HER2-positive breast cancer. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial Chan A, Delaloge S, Holmes FA, et al. The Lancet Oncology 2016; 17:367- 377
Weight Gain Increases the Risk for Postmenopausal Breast cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. Obesity is an important contributing factor to postmenopausal breast cancer incidence and mortality. Based on recently published meta-analysis, in women diagnosed with breast cancer, there is an approximately 30% increased risk of disease recurrence or death in those who are obese compared to those with ideal body weight. Increasing physical activity may lower the risk of breast cancer recurrence. According to the consensus from the St Gallen Consensus Conference in 2015, obesity has been associated with poor breast cancer outcomes. Obesity is associated with alterations in insulin/glucose homeostasis, adipokines, and sex hormones, which may play a role in breast cancer outcomes. Weight loss can lead to reductions in C-reactive protein, insulin, glucose, and leptin. These mediators have all been implicated to have prognostic significance in breast cancer. 
The Nurses’ Health Studies (NHS) are the largest and longest running investigations focused on women’s health. This was established in 1976 and the information provided by its 238,000 dedicated nurse-participants has allowed NHS to produce key advances impacting women’s health. These studies are conducted by researchers at Harvard School of Public Health and Brigham and Women's Hospital in Boston, Massachusetts. The authors conducted a clinical trial in this NHS cohort and studied the effects of weight and weight changes in early adulthood and risk of breast cancer later in life. A prospective observational study was conducted among 74,177 women from the Nurses' Health Study from 1980-2012. These women provided information on breast cancer risk factors such as reproductive factors, hormone therapy, anthropometric variable, benign breast disease, and family history of breast cancer. This information was updated every 2 years up to the time of data analysis. Each individual’s weight at age 18 was collected in 1980.
During the observation period, 4965 cases of invasive breast cancer were reported for the 74,177 women followed from 1980 to 2012. Weight gain over a long period of time from age 18, both during premenopause and postmenopause, were positively associated with postmenopausal breast cancer risk. However, premenopausal weight gain was not related to premenopausal breast cancer risk. Further, weight gain from age 18 yrs onwards was positively associated with ER+/PR+ postmenopausal breast cancer and there was a 50% increased risk for breast cancer with a weight gain of 30 kg. This direct association was not seen for ER+/PR- or ER-/PR- breast cancer. The authors noted that overall, 17% of ER+/PR+ postmenopausal breast cancer and 14% of total postmenopausal breast cancer are attributable to weight gain of more than 5 kg after age 18.
It was concluded that 14% of postmenopausal breast cancer could be prevented if women avoided excessive weight gain of more than 5 kg after age 18. This study adds new insights on weight gain during premenopausal years and risk for postmenopausal breast cancer. Weight and weight changes in early adulthood and later breast cancer risk. Rosner B, Eliassen AH, Toriola AT, et al. Int J Cancer. 2017 Jan 30. doi: 10.1002/ijc.30627 [Epub ahead of print]
Late Breaking Abstract – ASCO 2017: Adjuvant Dual HER2 Regimen of PERJETA® and HERCEPTIN® Improves Outcomes in Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. Trastuzumab binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). Adjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive early breast cancer.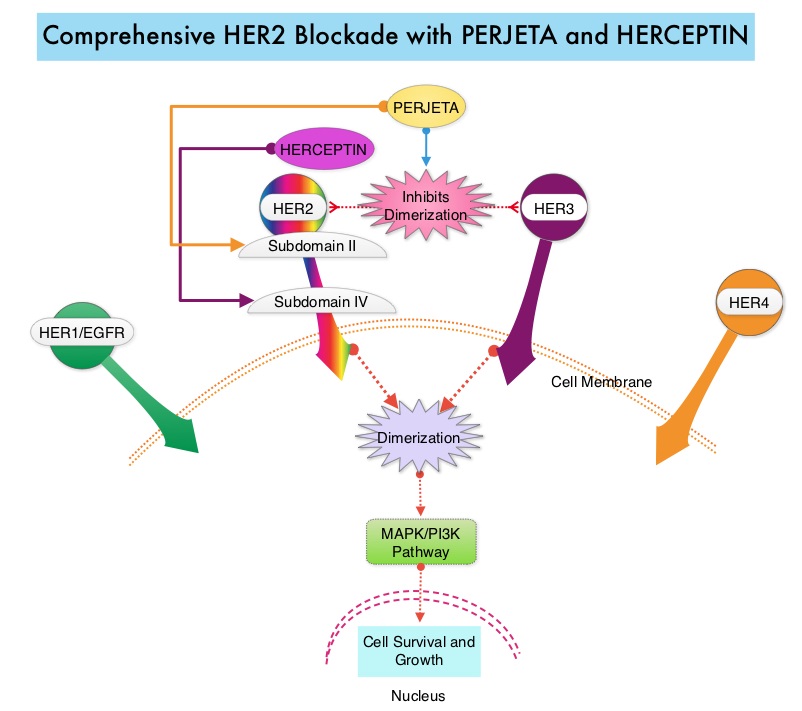
PERJETA® (Pertuzumab) is a recombinant humanized monoclonal antibody that binds to the HER2 at a different epitope of the HER2 extracellular domain (subdomain II) compared to HERCEPTIN® and prevents the dimerization of HER2 with HER3 receptor. PERJETA® induces ADCC similar to HERCEPTIN®. By combining HERCEPTIN® and PERJETA®, a more comprehensive blockade of HER2 signaling can be accomplished, as these two agents bind to different HER2 epitopes and may complement each other and improve efficacy. In the CLEOPATRA study, the addition of PERJETA® to HERCEPTIN® and Docetaxel resulted in significant improvement in Progression Free Survival (PFS) and Overall Survival (OS), in patients with HER-positive metastatic breast cancer. This triple drug combination also resulted in a significantly increased pathological Complete Response rate, when given in a neoadjuvant setting (NeoSphere trial).
Based on these previously published efficacy data, the authors in this study investigated whether the addition of PERJETA® to adjuvant HERCEPTIN® and chemotherapy, improves outcomes, among patients with HER2-positive early breast cancer. APHINITY is a prospective, randomized, multicenter, multinational, double-blind, placebo-controlled phase III trial in which a total of 4805 patients were randomly assigned in a 1:1 ratio, to receive standard adjuvant anthracycline or non-anthracycline chemotherapy regimen along with HERCEPTIN® plus either PERJETA® (2400 patients) or placebo (2405 patients). Anti-HER2 therapy was administered for a total of 1 year. Patients could receive radiotherapy and/or endocrine therapy following completion of adjuvant chemotherapy. Eligible patients had node-positive or high-risk node-negative (tumor diameter greater than 1.0 cm), HER2-positive, non-metastatic, adequately excised breast cancer. Both treatment groups were well balanced and about 37% of the patients had 1-3 positive lymph nodes and 25% of the patients had 4 or more positive lymph nodes. Two thirds of the patients were hormone receptor positive and about 78% of the patients received an anthracycline containing adjuvant chemotherapy regimen. The median follow up was 45.4 months and one year of treatment was completed by approximately 85% of the patients in both treatment groups. The primary end point was Disease Free Survival (DFS) from invasive breast cancer and secondary end points included Overall Survival (OS) and DFS from non-invasive breast cancers.
The addition of PERJETA® to chemotherapy and HERCEPTIN® resulted in a higher rate of DFS for invasive breast cancer with a 3-year invasive DFS of 94.1% in the PERJETA® group and 93.2% in the placebo group (HR=0.81; P=0.045), in favor of PERJETA®. Patients in the high risk subgroups benefited the most. The 3-year invasive DFS for patients with node-positive disease was 92.0% in the PERJETA® group, compared with 90.2% in the placebo group (HR=0.77; P=0.02). In the cohort of patients with hormone receptor negative tumors, the 3-year invasive DFS was 92.8% in the PERJETA® group and 91.2% in the placebo group (HR=0.76; P=0.08). The site of first distant recurrence was visceral or in the CNS rather than the bone. Cardiac toxicities were uncommon in both treatment groups and patients in the PERJETA® group had higher incidence of diarrhea while on concurrent chemotherapy.
It was concluded that for patients with HER2-positive early breast cancer, the addition of PERJETA® to standard postoperative HERCEPTIN® based adjuvant chemotherapy, significantly improved Disease Free Survival for invasive breast cancer. This benefit was more so for those patients with high risk disease. APHINITY trial (BIG 4-11): A randomized comparison of chemotherapy (C) plus trastuzumab (T) plus placebo (Pla) versus chemotherapy plus trastuzumab (T) plus pertuzumab (P) as adjuvant therapy in patients (pts) with HER2-positive early breast cancer (EBC). von Minckwitz G, Procter MJ, De Azambuja E, et al. J Clin Oncol. 2017;35(suppl; abstr LBA500).
Late Breaking Abstract - ASCO 2017: LYNPARZA® Improves Progression Free Survival in BRCA Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. DNA can be damaged due to errors during its replication or as a result of environmental exposure to ultraviolet radiation from the sun or other toxins. The tumor suppressor genes such as BRCA1 (Breast Cancer 1) and BRCA2 help repair damaged DNA and thus play an important role in maintaining cellular genetic integrity, failing which these genetic aberrations can result in malignancies. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. Mutations in BRCA1 and BRCA2 account for about 20 to 25 percent of hereditary breast cancers and about 5 to 10 percent of all breast cancers. These mutations can be inherited from either of the parents and a child has a 50 percent chance of inheriting this mutation and the deleterious effects of the mutations are seen even when an individual’s second copy of the gene is normal. 
The PARP (Poly ADP Ribose Polymerase) family of enzymes which include PARP1 and PARP2, repair damaged DNA. LYNPARZA® (Olaparib) is a PARP enzyme inhibitor that causes cell death in tumors that already have a DNA repair defect, such as those with BRCA1 and BRCA2 mutations. The FDA approved LYNPARZA® (Olaparib) in 2014 as monotherapy for the treatment of patients with deleterious or suspected deleterious germline BRCA mutated advanced ovarian cancer.
OlympiAD is a randomized, open-label, phase III study that evaluated the efficacy and safety of LYNPARZA® (Olaparib) compared with physician’s choice of standard single agent chemotherapy (TPC), in patients with HER2-negative metastatic breast cancer, with inherited, germline BRCA mutations. In this study, 302 patients were randomized in a 2:1 ratio to receive LYNPARZA® tablets 300 mg PO BID (N=205) or physician’s choice of standard chemotherapy (N=97). The later included 21-day cycles of either XELODA® (Capecitabine) 2500 mg/m2 orally on days 1-14, NAVELBINE® (Vinorelbine) 30 mg/m2 IV days 1 and 8 or HALAVEN® (Eribulin)1.4 mg/m2 IV days 1 and 8. Treatment was continued until disease progression or unacceptable toxicity. The median age was 44 years, 50% of the patients had triple negative disease, 71% of the patients had prior chemotherapy for metastatic breast cancer, 28% had prior platinum based chemotherapy regimen and those with hormone receptor positive breast cancer had received hormonal therapy. The primary endpoint was Progression Free Survival (PFS). Secondary endpoints included Overall Survival, time to second progression or death, Objective Response Rate and effect on health-related Quality of Life.
At a median follow up of about 14 months, the median PFS was 7 months in the LYNPARZA® group versus 4.2 months with standard chemotherapy (HR=0.58; P=0.0009), suggesting a 42% reduced risk of cancer progression in the LYNPARZA® group compared to those who received chemotherapy. Following disease progression, the time to second progression (which meant duration of time before the cancer worsened again), was also longer in the LYNPARZA® group (HR 0.57), suggesting that recurrent disease was not more aggressive following progression on LYNPARZA®. The Objective Response Rate was 60% and 29% in LYNPARZA® and chemotherapy group respectively. Severe side effects were more common in chemotherapy treated patients (50%) compared with LYNPARZA® group (37%). The most common side effects in the LYNPARZA® group included nausea, fatigue and cytopenias, where as rash on hands and feet were most common in the chemotherapy group.
The authors concluded that LYNPARZA® monotherapy significantly improved Progression Free Survival in HER2-negative metastatic breast cancer patients, with inherited germline BRCA mutations, compared to standard chemotherapy. This “proof of the principle” study demonstrated that breast cancers with defects in a specific DNA damage repair pathway are sensitive to targeted therapy and this is the first of several phase III studies with PARP inhibitors that are underway. OlympiAD: Phase III trial of olaparib monotherapy versus chemotherapy for patients (pts) with HER2-negative metastatic breast cancer (mBC) and a germline BRCA mutation (gBRCAm). Robson ME, Im S-A, Senkus E, et al. J Clin Oncol 35, 2017 (suppl; abstr LBA4).
Final Overall Survival Results with KADCYLA® in Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. It binds to the extracellular domain of the receptor and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Despite this benefit, majority of these patients develop progressive disease within 18 months. The tumors in these patients continue to express HER2 although the lower sensitivity to HER2 targeted agents has been attributed to HER2 independent escape mechanisms. Treatment strategies for this patient population have included switching chemotherapy in subsequent lines of treatment and continuing HERCEPTIN®, combining another HER2 targeted agent, Lapatinib (TYKERB®) with Capecitabine (XELODA®) and dual HER2 inhibition with a combination of HERCEPTIN® and TYKERB®.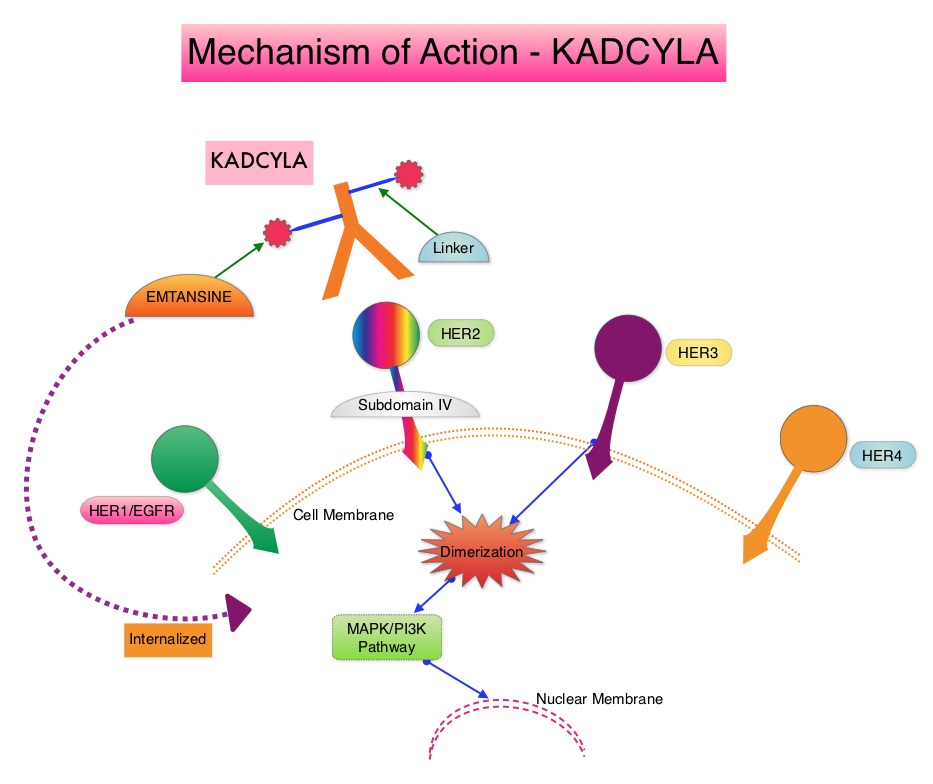
KADCYLA® (Ado-Trastuzumab Emtansine, T-DM1) is an Antibody-Drug Conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release. In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® plus XELODA®, in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a taxane.
TH3RESA is an open label randomized phase III trial in which KADCYLA® was compared with treatment of physician’s choice, in patients with unresectable locally advanced, recurrent or metastatic breast cancer. Eligible patients had a left ventricular ejection fraction of 50% or more and had HER2-positive advanced breast cancer and had received two or more HER2-directed regimens in the advanced setting, and had progressed on both HERCEPTIN® and TYKERB® containing regimens in metastatic setting, and also had disease progression on a taxane, in any setting. Patients were randomized in a 2:1 ratio to receive either KADCYLA® 3.6 mg/kg IV every 21 days (N=404) or treatment of physician’s choice (N=198), which included HER2 directed therapy for the majority of patients. Treatment was continued until disease progression or unmanageable toxicity. The Co-primary endpoints were Progression Free Survival (PFS) and Overall Survival (OS). Secondary endpoints included Response Rates, Duration of Response, Safety and Quality of Life. The authors had previously reported a significant improvement in PFS with KADCYLA® compared with physician's treatment choice (6.2 months vs 3.3 months, HR= 0.528, P<0.0001) and an OS trend favoring KADCYLA®.
The authors now reported the results from the final Overall Survival analysis of the TH3RESA trial. At data cutoff, 47% of the patients in the physician's choice group had crossed over to KADCYLA®. The Overall Survival was significantly longer with KADCYLA® compared with treatment of physician's choice (median 22.7 months versus 15.8 months, HR=0.68, P=0.0007). This benefit was seen in all pre-specified subgroups. Patients in the KADCYLA® group had a lower incidence of grade 3 toxicities compared to the patients in the physician’s treatment choice group (40% vs 47%). Grade 3 thrombocytopenia however was more common in the KADCYLA® group compared to the physician’s choice group (6% vs 3%) and this has been attributed to the inhibition of megakaryocyte differentiation by KADCYLA®.
The authors concluded that for patients who had progressed on two or more HER2-directed regimens, treatment with KADCYLA® significantly improved Overall Survival, compared with treatment of physician's choice, thereby validating HER2 as a therapeutic target, even after multiple lines of previous therapy. Trastuzumab emtansine versus treatment of physician's choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): final overall survival results from a randomised open-label phase 3 trial. Krop IE, Kim SB, González-Martín A, et al. Lancet Oncol. DOI: http://dx.doi.org/10.1016/S1470-2045(17)30313-3
Late Breaking Abstract - ECCO 2017: Breast Conserving Therapy Better Than Mastectomy in Some Patients with Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. The National Surgical Adjuvant Breast and Bowel Project (NASBP) protocols B-04 and B-06 have clearly established after more than a 2 decades of evaluation and follow up that, in Stage I and II breast cancer, there is no significant difference in either distant Disease Free or Overall Survival between the Breast Conserving Therapy (BCT) and Breast Removal Surgery (Mastectomy). This data established Breast Conserving Therapy (BCT) as the preferred local-regional procedure. These trials however often excluded elderly patients or patients with co-morbidities. Radiotherapy after breast- conserving surgery significantly decreases the risk of local recurrence and improves Breast Cancer Specific Survival (BCSS) in certain subgroups of patients. According to the American Cancer Society, 42% of all invasive breast cancers in the US occur in women 65 years of age or older. These patients may have associated co-morbidities and may therefore be appropriate candidates for breast- conserving surgery rather than mastectomy. These patients also have better outcomes, as post-op recovery time is shorter. There is however limited data to confirm these findings, as most studies evaluated limited numbers of patients, lacked long term follow up and the cause of death in these patients could not be clearly determined.
To further address this question, the authors in this study compared breast-conserving surgery plus radiation therapy (BCT) with Mastectomy, for Breast Cancer Specific (BCSS) and Overall Survival (OS), in a population-based study of 129,692 breast cancer patients without metastatic disease, in the Netherlands. Patients were selected from the Netherlands Cancer Registry, who had T1-2, N0-2, M0 breast cancer, diagnosed between1999 and 2012. Patients were divided into two time cohorts: those diagnosed between 1999 and 2005 (long term follow up), and those diagnosed between 2006 and 2012, (contemporary adjuvant systemic therapy). The influence of prognostic factors such as age, stage, adjuvant systemic therapy, hormonal and HER2 receptor status and co-morbidities was studied in these two groups of patients, in order to identify possible prognostic factors, that might predict patient groups, who could benefit the most from Breast Conserving Therapy (BCT). Information on the cause of death was obtained from Statistics Netherlands, also known as the Dutch Central Bureau of Statistics.
It was noted that for patients in the long-term follow up cohort, Breast Conservation Therapy was associated with a statistically significant improvement in Breast Cancer Specific Survival and Overall Survival compared to Mastectomy in all T1-2, N0-2 stages. For patients diagnosed between 2006 to 2012 (contemporary adjuvant systemic therapy), Breast Conserving Therapy was again associated with a statistically significant improvement in Breast Cancer Specific Survival and Overall Survival for patients in the T1-2, N0-1 stage but not those with T1-2, N2 disease, and in this later group, Breast Cancer Specific Survival (BCSS) with conservation therapy was equal to that with mastectomy. Subgroup analyses in the T1-2, N0-1 subset showed superior BCSS with breast conservation in patients older than 50 years, those who did not receive chemotherapy and those who had co-morbid conditions, irrespective of hormone receptor or HER2 status. The Overall Survival (OS) results were similar. Among patients younger than 50 years of age without co-morbidities, and those who received chemotherapy, BCSS with breast conservation was equal to that with mastectomy, but OS was better with Breast Conservation Therapy than with Mastectomy.
It was concluded that in this large population of “real world” patients as seen in daily clinical practice, Breast Conserving Therapy is associated with superior Breast Cancer-Specific and Overall Survival when compared to Mastectomy in patients over 50 years of age, T1-2, N0-1 M0 stage, patients who had not received chemotherapy and patients with co-morbidities. This benefit was confirmed for patients in both time cohorts. This study information allows the Health Care Provider to decide which type of surgical treatment is best suited for some subtypes of Breast cancer. Breast conserving therapy and mastectomy revisited: Breast cancer-specific survival and the influence of prognostic factors in 129,692 patients. Lagendijk M, van Maaren MC, Saadatmand S, et al. ECCO2017 European Cancer Congress. Abstract number: 4LBA
Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: A Cancer Care Ontario and American Society of Clinical Oncology Clinical Practice Guideline
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop breast cancer during their life time. Approximately, 255,180 new cases of breast cancer will be diagnosed in 2017 and 41,070 women will die of the disease. Cancer Care Ontario and ASCO convened a Working Group and Expert Panel and following a systematic review of the literature, developed evidence-based recommendations regarding the use of bisphosphonates and other bone-modifying agents, as adjuvant therapy, for patients with breast cancer. These guidelines are based on several important findings noted in previously published studies and in the more recently published Oxford Overview (Early Breast Cancer Trialists’ Collaborative Group) analysis of individual patient data.
1) Adjuvant bisphosphonates were found to reduce bone recurrence and improve survival in postmenopausal patients with non-metastatic breast cancer (including those with natural menopause or menopause induced by ovarian suppression or ablation)
2) The absolute benefit with adjuvant bisphosphonates was greater in patients who were at a higher risk of recurrence, and almost all clinical trials were conducted in patients who also received systemic therapy.
3) Most studies evaluated ZOMETA® (Zoledronic acid) or BONEFOS® (Clodronate), and there was extremely limited data for other bisphosphonates. Although XGEVA® (Denosumab) was found to reduce fractures, long-term survival data is awaited.
The following is a summary of the panel’s recommendations:
Recommendation 1
a) Administration of bisphosphonates as adjuvant therapy should be considered for postmenopausal patients with breast cancer (including patients who are premenopausal before treatment and have menopause induced by ovarian suppression as detailed in Recommendation 5) deemed candidates for adjuvant systemic therapy.
b) The final decision of whether or not to administer bisphosphonates should be made during consultation between the patient and oncologist, taking into account patient and disease characteristics, including the risk of recurrence, and weighing the potential benefits and risks.
Recommendation 2
a) Zoledronic acid and Clodronate are the recommended bisphosphonates for adjuvant therapy in breast cancer.
b) There is a need for more information comparing different agents and schedules, and it is recommended that such trials be conducted to establish the utility and optimal administration of other bisphosphonates for adjuvant therapy.
Recommendation 3
a) There is insufficient evidence at this time to make any recommendation regarding the use of Denosumab in the adjuvant setting.
b) It is recommended that studies directly comparing Denosumab with bisphosphonates and evaluating administration schedules, be conducted.
Recommendation 4
a) For patients who will receive adjuvant bisphosphonates (Recommendation 1), Zoledronic acid 4 mg IV over 15 minutes (or longer) every 6 months for 3 to 5 years or Clodronate PO 1,600 mg daily for 2 to 3 years is recommended. Different durations may be considered.
b) More research is recommended comparing different bone-modifying agents, doses, dosing intervals, and durations.
Recommendation 5
For purposes of adjuvant bisphosphonate use, the definition of menopause should include natural menopause (at least 12 months of amenorrhea prior to initiation of chemotherapy or endocrine therapy) and menopause induced by ovarian ablation or suppression (but not the cessation of menses due to chemotherapy alone). In women aged 60 years or less with a previous hysterectomy and ovaries left in place, Luteinizing Hormone, Follicle Stimulating Hormone and Serum Estradiol should be in the postmenopausal range and measured prior to initiation of any systemic therapy, to receive adjuvant bisphosphonates.
Recommendation 6
a) A dental assessment is recommended prior to commencement of bisphosphonates, where feasible, and any pending dental or oral health problems should be dealt with prior to starting treatment, if possible. Patients should be informed of the risk of developing osteonecrosis of the jaw, especially with tooth extractions and other invasive dental procedures. Patients should inform their dental practitioner of their treatment. Patients with suspected osteonecrosis of the jaw should be referred to a dental practitioner with expertise in treating this condition. Recent guidelines or position papers by groups such as the International Task Force on Osteonecrosis of the Jaw, the American Association of Oral and Maxillofacial Surgeons, and the American Dental Association should be consulted.
b) Patients should have Serum Calcium measured prior to starting treatment. Patients receiving intravenous bisphosphonates (Zoledronic acid) should be monitored for renal function, prior to starting this treatment and for Serum Calcium and increase in Serum Creatinine, throughout the treatment period.
c) Calcium and vitamin D supplementation is recommended unless otherwise contraindicated. Oral bisphosphonates and Calcium should not be taken concurrently. Several monographs suggest an interval of at least 2 hours to allow for maximum absorption.
d) Symptoms such as ocular pain or loss of vision may be due to serious inflammatory conditions such as uveitis or scleritis and should be promptly evaluated by an ophthalmologist.
Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: A Cancer Care Ontario and American Society of Clinical Oncology Clinical Practice Guideline. Dhesy-Thind S, Fletcher GG, Blanchette PS, et al. DOI: 10.1200/JCO.2016.70.7257 Journal of Clinical Oncology - published online before print March 6, 2017
FDA Approves KISQALI® for First-Line Treatment of Hormone Receptor Positive Advanced Breast cancer
SUMMARY: The FDA on March 13, 2017 approved KISQALI® (Ribociclib), a cyclin-dependent kinase 4/6 inhibitor, in combination with an Aromatase Inhibitor, as initial endocrine-based therapy for the treatment of postmenopausal women with Hormone Receptor (HR)-positive, Human Epidermal growth factor Receptor 2 (HER2)-negative advanced or metastatic breast cancer. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop breast cancer during their life time. Approximately, 255,180 new cases of breast cancer will be diagnosed in 2017 and 41,070 women will die of the disease. Approximately 70% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein nullifies its beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors.
KISQALI® is an orally bioavailable, selective, small-molecule inhibitor of CDK4/6 that blocks the phosphorylation of RetinoBlastoma protein, thereby preventing cell-cycle progression and inducing G1 phase arrest. In a phase 1b study involving postmenopausal women with ER positive, HER2-negative advanced breast cancer, KISQALI® in combination with FEMARA® (Letrozole) demonstrated an Overall Response Rate (ORR) of 46% and a Clinical Benefit Rate of 79%, in treatment-naïve patients with advanced breast cancer.
MONALEESA-2 trial is a randomized, double-blind, placebo-controlled, phase III study in which 668 patients were randomly assigned in a 1:1 ratio to receive either KISQALI® plus FEMARA® (Letrozole) or placebo plus FEMARA®. Eligible patients included post-menopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer who had received no prior therapy for advanced disease. Treatment consisted of oral KISQALI® 600 mg daily on a 3-weeks on and 1-week off schedule, in 28-day treatment cycles plus FEMARA® 2.5 mg orally daily on a continuous schedule or placebo plus FEMARA®. Patients were stratified according to the presence or absence of liver or lung metastases and treatment was continued until disease progression or unacceptable toxicity. No treatment crossover was allowed. The median age was 62 years and close to 60% of the patients had visceral metastases. The primary end point was Progression Free Survival (PFS) and secondary end points included Overall Survival (OS), Overall Response Rate (ORR), Clinical Benefit Rate (Overall Response plus stable disease lasting 24 weeks or more), safety, and Quality of Life assessments.
A pre-planned interim efficacy analysis demonstrated a significant improvement in the PFS amongst the KISQALI® group compared to the placebo group (HR=0.56; P<0.0001). The median duration of follow-up was 15.3 months. The estimated median PFS had not been reached in the KISQALI® group and was 14.7 months in the placebo containing arm. The Overall Response Rate (ORR) in patients with measurable disease was 52.7% in the KISQALI® group and 37.1% in the placebo plus FEMARA® group (P<0.001). Overall Survival data was mature at the time of this analysis. The rates of discontinuation because of adverse events were 7.5% in the KISQALI® group and 2.1% in the placebo group. The most common adverse reactions observed in patients taking KISQALI® were myelosuppression, nausea, vomiting, diarrhea and fatigue, as well as abnormal liver function tests. KISQALI® has been shown to prolong the QT interval in a dose-dependent manner and prolongation of the QT interval occurred in 3.3% of patients treated at the 600 mg dose, with changes mostly occurring within the first 4 weeks of treatment.
The authors concluded that among patients receiving initial systemic treatment for HR-positive, HER2-negative advanced breast cancer, the addition of KISQALI® to FEMARA® significantly prolonged PFS compared to FEMARA® alone, with a higher rate of myelosuppression noted in the KISQALI® group. Ribociclib as First-Line Therapy for HR-positive, Advanced Breast Cancer. Hortobagyi GN, Stemmer SM, Burris HA, et al. N Engl J Med 375:1738-1748, 2016.
IBM Artificial Intelligence Platform Highly Concordant with Physician Recommendations
SUMMARY: Watson for Oncology, is an Artificial Intelligence (AI) computer developed by IBM in collaboration with Memorial Sloan Kettering Cancer Center. This revolutionary tool has the advanced ability to analyze the meaning and context of structured and unstructured data in the patients chart and is able to assimilate key patient information and then deliver evidence based treatment recommendations, through analytical approaches. The authors conducted this study to assess concordance between the Artificial Intelligence platform, Watson for Oncology (WFO) and their own multidisciplinary tumor board, which comprised of a group of 12 to 15 oncologists, who met weekly to review cases from their hospital system. The goal of the study was to understand how Watson for Oncology would impact oncologists day-to-day practice, and how Watson’s recommendations compared to the decisions of their team of experts.
The researchers studied 638 patients with breast cancer treated at Manipal Comprehensive Cancer Center in Bengaluru, India. Patient data was entered into the Watson for Oncology (WFO) computer system and the degree of concordance between WFO’s recommendations and those of the tumor board were analyzed, in addition to the time it took for each group to come up with their recommendations. In this study, WFO analyzed more than 100 patient attributes for breast cancer and provided treatment options ranked as follows - Recommended Standard Treatment (REC), For Consideration (FC) and Not Recommended (NREC). These recommendations provided by WFO were evidence based and the computer system allowed the treating physicians to learn more about the recommendations and the rationale behind those recommendations.
It was noted that 90% of WFO’s Recommendations for Standard Treatment (REC) and For Consideration (FC) were concordant with the recommendations of the tumor board. WFO recommendations were concordant nearly 80% of the time in non-metastatic breast cancer, but only 45% of the time in metastatic disease. In patients with triple-negative breast cancer, WFO agreed with the physicians 68% of the time, but in HER-2 negative cases, WFO’s recommendations matched the physician’s recommendations only 35% of the time. The authors attributed the difference in concordance to fewer treatment options for triple-negative breast cancer, compared to HER-2 negative breast cancer. Further, including HER-2 patients made more treatment options available and this would increase the demands on human thinking capacity. Additionally, more complicated cases lead to more divergent opinions on the recommended treatment.
This study also compared the amount of time it took to provide recommendations, after the data was captured and analyzed. It took an average of 20 minutes when done manually, but after gaining more familiarity with the cases, the time decreased to about 12 minutes. Watson for Oncology by comparison, took a median time of 40 seconds to capture and analyze data and give a treatment recommendation.
It was concluded that while Artificial Intelligence is a step towards personalized medicine, it should not be viewed as a replacement for a physician, but rather as a complement. In the end, the best treatment option for the patient should be determined together by the treating physician and the patient. Double blinded validation study to assess performance of IBM artificial intelligence platform Watson for oncology in comparison with Manipal multidisciplinary tumor board—first study of 638 breast cancer cases. Somashekhar SP, Kumar R, Rauthan A, et al. Presented at: San Antonio Breast Cancer Symposium, Friday, Dec. 9, 2016; San Antonio, TX. Abstract S6-07
ESR1 Mutations Predict Response to Endocrine Therapy in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop breast cancer during their life time. Approximately, 255,180 new cases of breast cancer will be diagnosed in 2017 and 41,070 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Approximately 60-65% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy which is the cornerstone of their treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. FASLODEX® (Fulvestrant) is an estrogen antagonist and like NOLVADEX®, binds to estrogen receptors (ERs) competitively, but unlike NOLVADEX® causes rapid degradation and loss of ER protein (ER downregulator), and is devoid of ER agonist activity.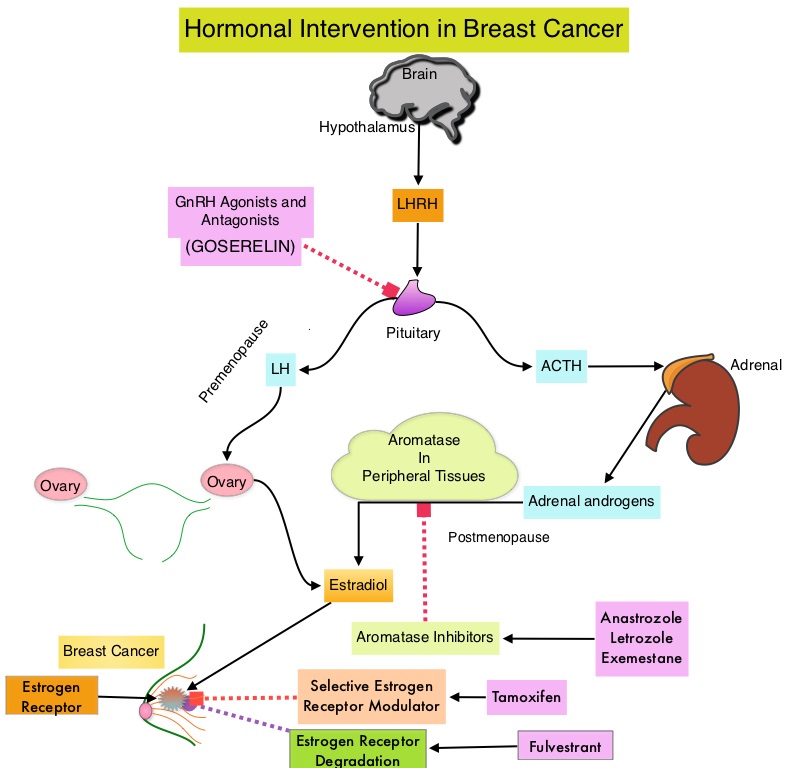
Upon development of metastatic disease, a subgroup of these patients, develop resistance to endocrine therapy. The most common acquired mutation noted in breast tumors as they progress from primary to metastatic setting are the ESR1 mutations. These mutations promote ligand independent estrogen receptor activation and have been shown to promote resistance to estrogen deprivation therapy. It appears that ESR1 mutations are harbored in metastatic ER-positive breast cancers with prior Aromatase Inhibitor (AI) therapy, but not in primary breast cancers, suggesting that ESR1 mutations may be selected by prior therapy with an AI, in advanced breast cancer. In a recently published study (JAMA Oncol.2016;2:1310-1315) ESR1 mutations Y537S and D538G mutations detected in baseline plasma samples from ER positive, HER negative advanced breast cancer patients, was associated with shorter Overall Survival. In this study it was noted that there was a three-fold increase in the prevalence of these mutations in patients who had failed first line hormonal therapy for metastatic disease, compared with those who were initiating first line therapy for advanced breast cancer (33% vs 11%).
Droplet digital Polymerase Chain Reaction (ddPCR) is a highly sensitive and specific technique and can detect ESR1 mutations in the plasma. Retrospective studies have shown that ESR1 mutations detected in plasma cfDNA (cell free DNA) by ddPCR were associated with a lack of response to subsequent AI therapy. The authors in this publication used baseline plasma samples and assessed the impact of ESR1 mutations on sensitivity to standard therapies in two phase III randomized trials, the SoFEA trial and PALOMA-3 trial, which are representative of the current standard of care for ER positive advanced breast cancer.
In the SoFEA trial (Study of Faslodex With or Without Concomitant Arimidex), AROMASIN® (Exemestane), a steroidal AI, was compared with FASLODEX® (Fulvestrant)-containing regimens, in patients with prior sensitivity to nonsteroidal AIs (Letrozole and Anastrozole). In the PALOMA3 trial (Palbociclib Combined With Fulvestrant in Hormone Receptor–Positive HER2-Negative Metastatic Breast Cancer After Endocrine Failure) trial, FASLODEX® plus placebo was compared with FASLODEX® plus IBRANCE® (Palbociclib), in patients with progression after receiving prior endocrine therapy. ESR1 mutations were analyzed by multiplex digital Polymerase Chain Reaction. (Multiplex PCR amplifies several different DNA sequences simultaneously and more information can be obtained from a single test run).
In the SoFEA trial, ESR1 mutations were found in 39.1% of patients of whom 49.1% were polyclonal. Polyclonal ESR1 mutations suggest that biopsy from a single metastatic site would fail to show capture these mutations. Patients with ESR1 mutations on FASLODEX® had improved Progression Free Survival (PFS) compared with AROMASIN® (HR=0.52; P=0.02). Patients with wild-type ESR1 had similar PFS after receiving either treatment (HR=1.07; P=0 .77). Ability to detect these mutations was not impacted by delays in processing of archival plasma. In the PALOMA3 trial, ESR1 mutations were found in the plasma of 25.3% of patients of whom 28.6% were polyclonal ESR1 mutations. The combination of FASLODEX® plus IBRANCE® improved PFS compared with FASLODEX® plus placebo in both ESR1 mutant (HR=0.43; P=0.002) and ESR1 wild-type patients (HR=0.49; P<0.001).
The authors concluded that plasma analysis for ESR1 mutations after progression on prior AI therapy may help direct choice of further endocrine-based therapy. Plasma ESR1 Mutations and the Treatment of Estrogen Receptor–Positive Advanced Breast Cancer. Fribbens C, O’Leary B, Kilburn L, et al. J Clin Oncol. 2016;34:2961-2968
ASCO Guideline - Adjuvant Systemic Therapy Decision Making for Early Stage Operable Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their life time. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant therapy. The ASCO Clinical Practice Guidelines Committee endorsed a set of Cancer Care Ontario guideline recommendations that addressed the role of a range of patient and disease characteristics, in selecting adjuvant therapy for women with early-stage breast cancer. This guideline does not address the selection of optimal adjuvant chemotherapy regimens.
Guideline Question: Which patient and disease factors should be considered in selecting adjuvant therapy for women with early-stage breast cancer?
Target Population: Female patients who are being considered for, or who are receiving systemic therapy for early-stage invasive breast cancer (Stages I–IIA, T1N0–1, T2N0).
RECOMMENDATIONS
-Decisions regarding adjuvant therapy should be based on relevant (either prognostic or predictive) information and consideration given to-
1) Lymph node status, T stage, Estrogen Receptor status, Progesterone Receptor status, HER2 status, tumor grade, and presence of tumor lymphovascular invasion.
2) Risk-stratification tools including Oncotype DX score (for hormone receptor-positive, N0 or N1mic or isolated tumor cell, and HER2-negative cancers) and Adjuvant! Online.
3) Patient age, menopausal status, and medical comorbidities.
-For patients in whom chemotherapy would likely be tolerated and for whom chemotherapy is acceptable, adjuvant chemotherapy should be considered if the following characteristics are present:
1) Lymph node-positive tumor (at least one node with macrometastatic deposit > 2 mm)
2) Estrogen receptor-negative tumor (> 5 mm)
3) HER2-positive tumor
4) High-risk node-negative tumors (> 5 mm) and another high-risk feature
5) Adjuvant! Online 10-year risk of death from breast cancer > 10%.
-Patients with node-negative early stage breast cancer with high risk features who should be considered candidates for chemotherapy include
1) Tumors > 5 mm
2) Grade III histology
3) Triple negative tumors
4) Lymphovascular invasion
5) Oncotype DX recurrence score associated with an estimated distant relapse risk ≥ 15% at 10 years
6) HER2-positive tumors
(The ASCO panel suggested an estimated distant relapse risk > 20% in this setting).
-Patients with tumor size < 5 mm, node-negative tumors, and no other high-risk features, may not benefit from adjuvant chemotherapy.
-Adjuvant chemotherapy may not be required in patients with HER2-negative, strongly ER-positive, and PR-positive breast cancer and any of the following additional characteristics: positive nodes with micrometastasis only (< 2 mm), or Tumor size < 5 mm, or Oncotype DX recurrence score with an estimated distant relapse risk < 15% at 10 years. (The ASCO panel suggested an estimated distant relapse risk < 10% at 10 years in this setting)
ASCO Panel Discussion Points
Areas that warrant further consideration include-
1) Tumor histology and adjuvant therapy recommendations
2) Risk-stratification tools and proposed Oncotype DX recurrence score thresholds to guide decisions about chemotherapy
3) Patient factors in decision-making.
The panel noted that some uncommon breast cancer subtypes (eg, tubular, mucinous) may have a favorable prognosis and that such histologic information may be relevant for making decisions regarding adjuvant chemotherapy. Additionally, factors such as Grade III disease and lymphovascular invasion generally should not be used in isolation in decision-making but considered within the overall clinical context.
Role of patient and disease factors in adjuvant systemic therapy decision making for early-stage, operable breast cancer: Henry NL, Somerfield MR, Abramson VG, et al. American Society of Clinical Oncology endorsement of Cancer Care Ontario guideline recommendations. J Clin Oncol 34:2303-2311, 2016
Axillary Lymph Node Dissection Can Be Avoided in Some High Risk Breast Cancer Patients
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Axillary lymph node evaluation is an important part of breast cancer staging and the presence of axillary lymph metastases decreases the 5-year survival rate by 28-40%. Axillary lymph node status remains the most powerful predictor of breast cancer recurrence and survival. Axillary Lymph Node Dissection (ALND) was first advocated in the 18th century as part of the treatment of invasive breast cancer and has been standard practice until 2 decades back. ALND can be associated with significant morbidities such as upper limb lymphedema, pain, and sensitivity disorders and this can have a major psychological impact on breast cancer patients. Sentinel Lymph Node Biopsy (SLNB) which was introduced into clinical practice in the mid 1990’s, however has now become a standard method of treatment for stage I and II breast cancer. This therapeutic surgical modality facilitates selective histopathological evaluation of the sentinel lymph nodes rather than routine lymphadenectomy, thereby sparing the patient from the morbidities associated with ALND. Several studies have shown no statistically significant difference in the axillary recurrence risk and survival rates, between these two therapeutic surgical modalities.
Neoadjuvant chemotherapy is considered standard practice in women with locally advanced breast cancer. This intervention increases the possibility of breast conserving surgery in women with locally advanced breast cancer and for tumors more than 3 cm in diameter, with good cosmetic outcomes. Close to half of the patients treated with neoadjuvant chemotherapy have no axillary lymph node involvement at the time of surgery. GANEA 2 trial was conducted to assess the feasibility and safety of SLNB, a less invasive procedure, for patients treated with neoadjuvant chemotherapy.
The researchers enrolled 590 patients with large, operable breast tumors who had no cancer in the lymph nodes as determined by axillary sonography with fine needle cytology. All patients received neoadjuvant chemotherapy, and then underwent surgery and Sentinel Lymph Node Biopsy. Cancer cells were detected in the Sentinel Lymph Node Biopsy specimens of 139 patients and these patients underwent Axillary Lymph Node Dissection.
No cancer cells were detected in the Sentinel Lymph Node Biopsy samples from 432 patients. Follow-up data was available for 416 of these patients. The median follow-up was 35.8 months. The Disease Free Survival at 3 years in the patient group who had no cancer cells in the Sentinel Lymph Node Biopsy sample, and therefore did not receive Axillary Lymph Node Dissection, was 94.8%. The Overall Survival rate was 98.7%.
The authors concluded that Axillary Lymph Node Dissection could be avoided in patients who have no signs of cancer involvement in the axillary lymph nodes, based on sonographic axillary assessment, prior to neoadjuvant chemotherapy and Sentinel Lymph Node Biopsy findings after neoadjuvant chemotherapy. In this study, the Disease Free Survival and Overall Survival rates for the patients who underwent only a Sentinel Lymph Node Biopsy after neoadjuvant chemotherapy, were comparable with the historical survival rates for patients in this situation who had Axillary Lymph Node Dissection rather than Sentinel Lymph Node Biopsy. Classe JM, Loaec C, Alran S, et al. Sentinel node detection after neoadjuvant chemotherapy in patient without previous axillary node involvement (GANEA 2 trial): follow-up of a prospective multi-institutional cohort. Presented at: 2016 San Antonio Breast Cancer Symposium; December 6-10, 2016; San Antonio, TX. Abstract S2-07.
Late Breaking Abstract - ESMO 2016: FASLODEX® Superior to ARIMIDEX® as Initial Therapy in Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Approximately 60-65% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. FASLODEX® (Fulvestrant) is an estrogen antagonist and like NOLVADEX®, binds to estrogen receptors (ERs) competitively, but unlike NOLVADEX® causes rapid degradation and loss of ER protein (ER downregulator), and is devoid of ER agonist activity.
The superiority of ARIMIDEX® over NOLVADEX® was first established in the year 2000 following the publication of the results of a North American Multicenter Randomized Trial. In this study, ARIMIDEX® as first-line treatment in postmenopausal women with advanced breast cancer resulted in a significant increase in Time To Progression and a lower incidence of thromboembolic events and vaginal bleeding, compared to NOLVADEX®. In a previously reported phase II study (FIRST trial), first-line treatment with FASLODEX® significantly improved Time To disease Progression and Overall Survival compared with ARIMIDEX®, in patients with hormone receptor (HR) positive advanced breast cancer.
The FALCON trial is a phase III study conducted to confirm findings from the FIRST trial. This study included 462 postmenopausal women, with locally advanced or metastatic hormone receptor positive, HER2-negative, endocrine-therapy naive breast cancer. Patients were randomized to receive FASLODEX® IM at 500 mg on days 0, 1, and 28 and then every 28 days (N=230) or ARIMIDEX® 1 mg PO daily (N=232). Treatment was continued until disease progression or unacceptable toxicity. The primary endpoint was Progression Free Survival (PFS), and secondary endpoints included Overall Survival, Objective Response Rate, Clinical Benefit Rate, Duration of Response, health-related Quality of Life, and safety.
It was noted that FASLODEX® provided better disease control with a median PFS of 16.6 months compared to 13.8 months with ARIMIDEX® (P=0.048). Subgroup analysis showed that FASLODEX® was markedly superior to ARIMIDEX® in patients with non-visceral disease with a median Progression Free Survival of 22.3 months compared with 13.8 months for ARIMIDEX®. There was no significant improvement in the Overall Response Rate between the treatment groups. However, the median Duration of Response was 20.0 months with FASLODEX® compared to 13.2 months with ARIMIDEX®. Expected Duration of response and expected Duration of Clinical Benefit were in favor of FASLODEX® (11.4 vs 7.5 months; P=0.001) and (21.9 vs 17.5 months; P=0.001), respectively. There was no difference in Overall Survival at the time of this analysis. Rates of adverse events were similar in both treatment groups.
The authors concluded that FASLODEX® was superior to ARIMIDEX® as initial treatment of hormone receptor positive, endocrine therapy naive, advanced breast cancer. Patients with non-visceral and low volume disease, as well as elderly patients, may benefit the most with FASLODEX®, as this therapy is well tolerated with a low toxicity profile. FALCON: a phase III randomised trial of fulvestrant 500 mg vs. anastrozole for hormone receptor-positive advanced breast cancer. Ellis MJ, Bondarenko I, Trishkina E, et al. Presented at: 2016 ESMO Congress; October 7-11, 2016; Copenhagen, Denmark. Abstract LBA14.
IBRANCE® plus FEMARA® - A New Standard for Previously Untreated Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Approximately 60-65% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. However, resistance to hormonal therapy occurs in a majority of the patients.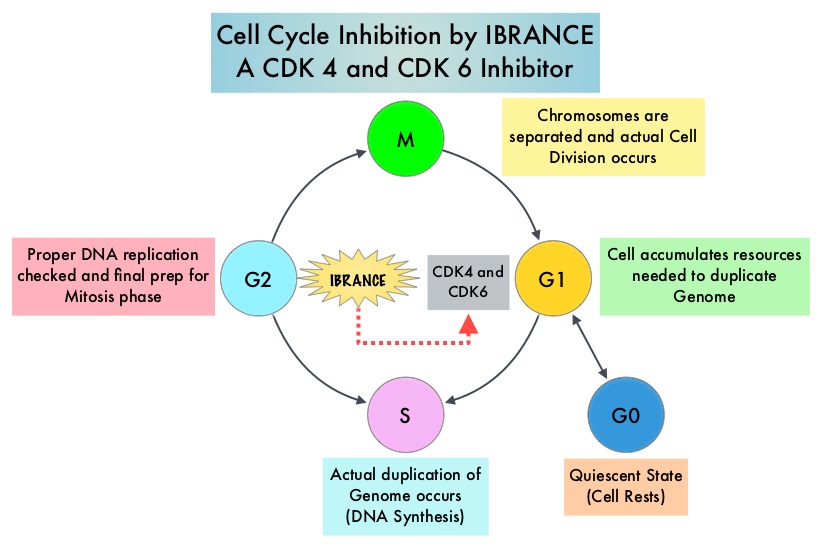
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. RetinoBlastoma protein has antiproliferative and tumor-suppressor activity and phosphorylation of RB protein cancels it beneficial activities. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. The understanding of the role of Cyclin Dependent Kinases in the cell cycle, has paved the way for the development of CDK inhibitors. IBRANCE® (Palbociclib) is a reversible, oral, selective, small molecule inhibitor of Cyclin Dependent Kinases, CDK4 and CDK6, and prevents RB1 phosphorylation. IBRANCE® is the first CDK inhibitor approved by the FDA. It exhibits synergy when combined with endocrine therapies. The FDA in February 2016, approved IBRANCE® in combination with FASLODEX® (Fulvestrant), for the treatment of women with Hormone Receptor (HR)-positive, Human Epidermal growth factor Receptor 2 (HER2) negative advanced or metastatic breast cancer, with disease progression following endocrine therapy. In a phase II study, a combination of IBRANCE® plus FEMARA® showed improved Progression Free Survival compared with FEMARA® alone, in the initial treatment of postmenopausal women with Estrogen-Receptor (ER) positive, HER2 negative advanced breast cancer. Based on this encouraging data, a phase III study was conducted, to confirm the efficacy and safety of IBRANCE® plus FEMARA® for this patient group.
In this double blind study (PALOMA-2), 666 postmenopausal women with ER positive, HER2 negative breast cancer, who had no prior therapy for advanced disease, were randomly assigned, in a 2:1 ratio to receive IBRANCE® plus FEMARA® (N=444) or placebo plus FEMARA® (N=222). IBRANCE® was administered at 125 mg PO daily, 3 weeks on and 1 week off, every 4 weeks and all patients received FEMARA® 2.5 mg PO daily . The median age was 62 years, 48% had visceral disease and 63% had prior systemic therapy for breast cancer. The primary end point was Progression Free Survival and secondary end points included Overall Survival, Objective Response Rate, Clinical Benefit Response and safety.
The median PFS was 24.8 months in the IBRANCE® plus FEMARA® group compared with 14.5 months in the placebo plus FEMARA® group (HR=0.58; P<0.001). This benefit was seen across all patient subgroups. The Objective Response Rate was 42% with the IBRANCE® plus FEMARA® combination and 35% with the placebo plus FEMARA® combination. The Clinical Benefit Response (complete response, partial response or stable disease for 24 weeks or more) was 84% in the IBRANCE® plus FEMARA® group and 71% in the placebo plus FEMARA® group (P<0.001). The most common grade 3 or 4 adverse events were neutropenia noted in 66% of the patients in the IBRANCE® group versus 1.4% in the placebo plus FEMARA® group. Approximately 10% of the patients in the IBRANCE® group permanently discontinued study treatment due to toxicities and 6% did so in the placebo plus FEMARA® group.
The authors concluded that a combination of IBRANCE® and FEMARA® significantly prolonged Progression Free Survival compared with FEMARA® alone, among patients with previously untreated ER-positive, HER2 negative advanced breast cancer and this combination should be the new standard of care for this patient group. Palbociclib and Letrozole in Advanced Breast Cancer. Finn RS, Martin M, Rugo HS, et al. N Engl J Med 2016; 375:1925-1936
Postmastectomy Radiotherapy: ASCO Guideline Update
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. The update of the American Society of Clinical Oncology guideline concerning use of PostMastectomy RadioTherapy (PMRT) published in 2001, was developed by an expert panel following review of relevant literature published between January 2001 and July 2015 and further included a meta-analysis of 22 clinical trials published in 2014. Even though the use of PMRT has been widely accepted for patients with four or more positive lymph nodes, the role of PMRT for those with one to three positive nodes still remains controversial. This update addresses the issues at large and provides guidelines to help Health Care Providers and patients make informed decisions.
Clinical Question 1
Is PMRT indicated in patients with T1-2 tumors with one to three positive axillary lymph nodes who undergo Axillary Lymph Node Dissection (ALND)?
Recommendation 1a: The panel unanimously agreed that the available evidence shows that PMRT reduces the risks of locoregional failure, any recurrence, and breast cancer mortality for patients with T1-2 breast cancer and one to three positive lymph nodes. However, one has to weigh the risk and benefit with PMRT and individualize therapy. Patients with low tumor burden, favorable tumor characteristics, comorbidities, or coexisting conditions and limited life expectancy, may not be appropriate candidates for PMRT, as its potential toxicities outweigh the absolute benefit of PMRT.
Recommendation 1b: The decision to use PMRT should be made in a multidisciplinary fashion through discussion among providers from all treating disciplines, early in a patient’s treatment course, soon after surgery or before or soon after the initiation of systemic therapy.
Recommendation 1c: Decision making must fully involve the patients, so that they are able to weigh the risk/benefits of PMRT, with the best information provided by the treating Health Care Provider.
Clinical Question 2
Is PMRT indicated in patients with T1-2 tumors and a positive Sentinel Node Biopsy (SNB) who do not undergo completion ALND?
Recommendation: Patients with T1-2 tumors with positive sentinel lymph node biopsy, who choose not to have Axillary Lymph Node Disection, should receive PMRT only if there is already sufficient information to justify its use, without needing to know that additional axillary nodes are involved. SNB is generally performed at the time of mastectomy for this patient group, with omission of ALND if the nodes are negative. If the sentinel nodes are positive, ALND is performed. There is increasing controversy about whether ALND is always necessary, if there is limited disease in the affected sentinel nodes. This practice is based on extrapolation of data from randomized trials of patients treated exclusively or predominantly with breast-conserving surgery and whole-breast irradiation or breast plus axillary irradiation.
Clinical Question 3
Is PMRT indicated in patients presenting with clinical stage I or II cancers who have received NeoAdjuvant Systemic Therapy (NAST)?
Recommendation: Patients with axillary nodal involvement that persists after NAST, such as less than a complete pathologic response, should receive PMRT. Observational data suggest a low risk of locoregional recurrence for patients who have clinically negative nodes and receive NAST or who have a complete pathologic response in the lymph nodes with NAST.
Clinical Question 4
Should Regional Nodal Irradiation (RNI) include both the Internal Mammary (IMNs) and supraclavicular-axillary apical nodes when PMRT is used in patients with T1-2 tumors with one to three positive axillary nodes?
Recommendation: Radiation Therapy should generally be administered to both the IMNs and the supraclavicular-axillary apical nodes in addition to the chest wall or reconstructed breast, when PMRT is used for patients with positive axillary lymph nodes.
Postmastectomy Radiotherapy: An American Society of Clinical Oncology, American Society for Radiation Oncology, and Society of Surgical Oncology Focused Guideline Update. Recht A, Comen EA, Fine RE, et al. Published online before print September 19, 2016, doi:10.1200/JCO.2016.69.1188
CDK4 and CDK6 Inhibitor ABEMACICLIB Highly Effective in Refractory Hormone Receptor Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Approximately 80% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation.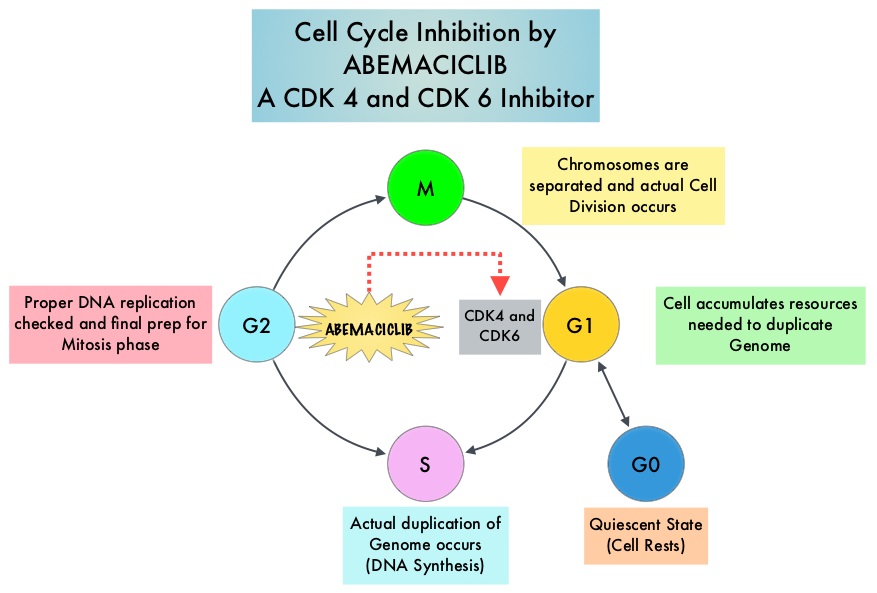
Abemaciclib is an oral, selective inhibitor of CDK4 and CDK6 kinase activity that prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell cycle arrest and inhibition of cell proliferation. The FDA granted breakthrough designation for Abemaciclib based on a phase I trial in which this agent demonstrated significant single agent activity in refractory Hormone Receptor (HR) positive metastatic breast cancer. MONARCH 1 is a single arm phase II study which evaluated the single-agent activity of Abemaciclib in heavily pretreated patients with HR-positive, HER2-negative metastatic breast cancer. This trial included 132 patients with HR-positive, HER2-negative metastatic breast cancer, whose disease progressed on or after endocrine therapy and chemotherapy. Patients had received a median of 3 lines of prior therapy for advanced disease and this included a median of 2 lines of chemotherapy. Approximately 50% of the patients had received FASLODEX® (Fulvestrant), 70% of patients had received Taxane based chemotherapy and 55 % of patients had received XELODA® (Capecitabine), in the metastatic setting. Abemaciclib was administered at 200 mg PO daily on a continuous schedule every 12 hours until disease progression. The median age was 58 yrs, 90% of the patients had visceral disease and 85% had at least 2 sites of metastatic disease. The primary endpoint was Objective Response Rate (ORR) and secondary endpoints included Duration of Response, Progression Free Survival (PFS), Overall Survival (OS), Clinical Benefit Rate (Complete Response plus Partial Response plus Stable Disease) and safety.
An interim analysis was performed at 8 months by when 35.6% of patients had received at least 8 cycles of Abemaciclib. The ORR was 17.4%, the Clinical Benefit Rate lasting for 6 months or more was 42.4%. The median time to response was 3.7 months and the median Duration of Response was 8.6 months. The median PFS was 5.7 months. The most common adverse events were diarrhea, fatigue, nausea, decreased appetite, abdominal pain and treatment discontinuation rate was infrequent at 6.8%.
It was concluded that CDK4 and CDK6 inhibitor, Abemaciclib, has significant antitumor activity in patients with refractory, HR-positive, HER2-negative metastatic breast cancer, for whom chemotherapy is the only option. Studies are underway combining Abemaciclib with FASLODEX®, for postmenopausal patients with HR-positive, HER2-negative, locally advanced or metastatic breast cancer who had progressed on 1 prior endocrine therapy (MONARCH-2 trial), as well as combining Abemaciclib with a nonsteroidal Aromatase Inhibitor in an earlier setting (MONARCH-3 trial), for patients with HR-positive, HER2-negative, locoregionally recurrent or metastatic breast cancer. MONARCH 1: Results from a phase II study of abemaciclib, a CDK4 and CDK6 inhibitor, as monotherapy, in patients with HR+/HER2- breast cancer, after chemotherapy for advanced disease. Dickler MN, Tolaney SM, Rugo HS, et al. J Clin Oncol 34, 2016 (suppl; abstr 510)
ASCO Guidelines on Adjuvant Targeted Therapy for HER2-Positive Breast Cancers
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their life time. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 20-25% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. HERCEPTIN® binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). Adjuvant chemotherapy in combination with HERCEPTIN® has been shown to reduce the relative risk of relapse by 52% and relative risk of death by 33%. ASCO has established a process for adapting clinical practice guidelines of other organizations and this summary of the practice recommendations were adapted from the Cancer Care Ontario evidence based clinical practice guidelines, for the adjuvant treatment of HER2-positive early breast cancers.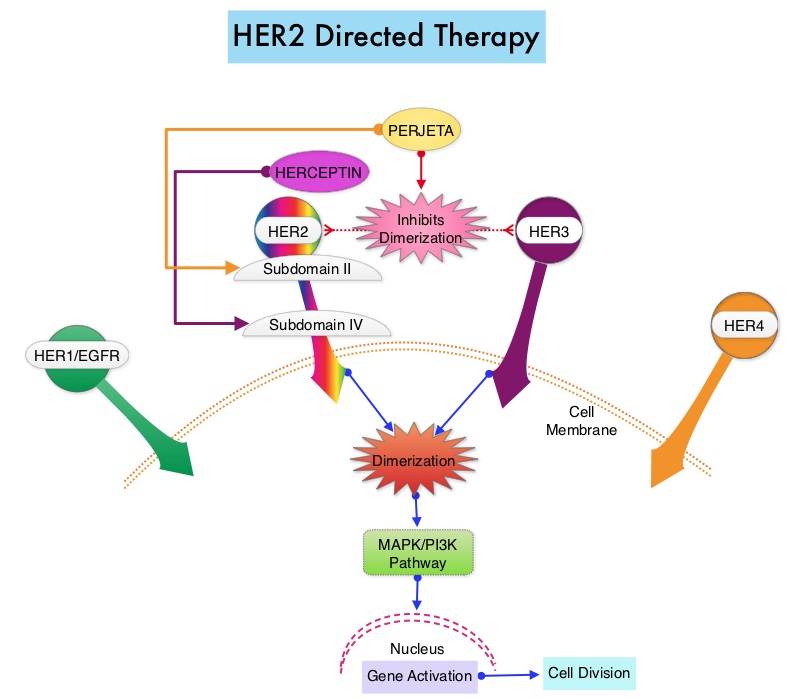
Guideline Question: What is the optimal use of cytotoxic chemotherapy and Human Epidermal growth factor Receptor 2 (HER2) - directed therapy?
Target Population: Female patients who are being considered for, or who are receiving, systemic therapy after definitive surgery for early invasive breast cancer, defined largely as invasive cancer stages I to IIA (T1N0-1, T2N0).
RECOMMENDATIONS
Use of an Anthracycline-Taxane Regimen
1) An adjuvant chemotherapy regimen containing Anthracycline-Taxane is considered the optimal strategy for high risk patients, if they are able to tolerate this regimen.
2) For patients with high-risk disease who will not receive a Taxane, an optimal-dose Anthracycline three-drug regimen (cumulative dose of doxorubicin ≥ 240 mg/m2 or epirubicin ≥ 600 but no higher than 720 mg/m2) that contains Cyclophosphamide, is recommended. The cumulative dose of Doxorubicin in two-drug regimens should not exceed 240 mg/m2.
3) The addition of Gemcitabine or Capecitabine to an Anthracycline-Taxane regimen is not recommended for adjuvant chemotherapy.
Capecitabine in Patients 65 Years of Age and Older
In patients age 65 years or older, Capecitabine is not recommended as an adjuvant chemotherapy option in lieu of standard regimens like Doxorubicin plus Cyclophosphamide (AC) or Cyclophosphamide, Methotrexate, and Fluorouracil (CMF with oral cyclophosphamide).
CMF as an Alternative to AC
For patients in whom Anthracycline-Taxane is contraindicated, CMF (with oral cyclophosphamide) is an acceptable chemotherapy alternative to AC. The ASCO panel recommends classic CMF (oral cyclophosphamide days 1 to 14 with IV Methotrexate-Fluorouracil days 1 and 8, repeated every 28 days for six cycles) as the default adjuvant CMF regimen. However, the panel also recognizes IV CMF regimen given every 21 days.
Acceptable Adjuvant Chemotherapy Regimens for Patients with Higher-Risk Early-Stage Breast Cancer
1) FEC (Fluorouracil, Epirubicin, and Cyclophosphamide) × 3 → T (Docetaxel) × 3 (superior to FEC × 6)
2) AC × 4 → T × 4 (superior to AC × 4)
3) Docetaxel, Doxorubicin, and Cyclophosphamide × 6 (superior to Fluorouracil, Doxorubicin, and Cyclophosphamide × 6)
4) AC × 4 → paclitaxel (P) administered weekly
5) Dose-Dense AC → P (every 2 weeks)
Adjuvant Regimen When an Anthracycline Is Not Preferred
1) Docetaxel plus Cyclophosphamide (TC) × 4 is recommended as an alternative to AC × 4
2) Classic CMF with oral Cyclophosphamide for six cycles. The panel also recognizes IV CMF regimen given every 21 days.
Patient Selection and Adjuvant Trastuzumab Therapy
Only patients with HER2-positive breast cancer (overexpressed based on ImmunoHistoChemistry (IHC 3+) or amplified based on in situ hybridization [ISH ratio ≥ 2.0 or average HER2 copy number ≥ 6.0]), should be offered adjuvant Trastuzumab.
Trastuzumab Plus Chemotherapy
1) Trastuzumab plus chemotherapy is recommended for all patients with HER2-positive, node-positive breast cancer and for patients with HER2-positive, node-negative breast cancer greater than 1 cm in size.
2) Trastuzumab therapy can be considered in small, node-negative tumors (1 cm or less).
3) Trastuzumab can be administered with any acceptable adjuvant chemotherapy regimen.
4) The administration of Trastuzumab concurrently with the Anthracycline component of a chemotherapy regimen is not recommended because of the potential for increased cardiotoxicity.
5) Trastuzumab should be preferentially administered concurrently (not sequentially) with a non-Anthracycline chemotherapy regimen.
6) Less cardiotoxicity is seen with TCH (Docetaxel, Carboplatin, and Trastuzumab) than with AC→TH (Doxorubicin and Cyclophosphamide→Docetaxel and Trastuzumab), and TCH is recommended for patients at higher risk for cardiotoxicity.
7) Even though there is no phase III evidence for the addition of Trastuzumab to some chemotherapy regimens, such as TC, those regimens might be in use and are reasonable options, particularly to mitigate cardiotoxicity in certain patients.
Duration of Trastuzumab Therapy and Cardiac Function Assessment
Patients should be offered 1 year total of adjuvant Trastuzumab, with regular assessments of cardiac function during that period.
Selection of Optimal Adjuvant Chemotherapy Regimens for Early Breast Cancer and Adjuvant Targeted Therapy for HER2-Positive Breast Cancers: An American Society of Clinical Oncology Guideline Adaptation of the Cancer Care Ontario Clinical Practice Guideline. Denduluri N, Somerfield MR, Eisen A, et al. J Clin Oncol 2016;34:2416-2427
MammaPrint® Identifies Women with Early Stage Breast Cancer Who Can Avoid Adjuvant Chemotherapy
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant chemotherapy. Chemotherapy recommendations for early stage breast cancer are often made based on tumor size, grade, hormone receptor and HER-2 status, immunohistochemical markers such as Ki-67, nodal status, patients age, menopausal and performance status. Adjuvant! Online is one of the tools that incorporates these features and assists in treatment decision making. This tool however does not take into account individual tumor molecular signatures which can better predict clinical outcomes. MammaPrint® is a 70-gene signature assay approved by the FDA and is able to distinguish low risk and high risk tumors based on the risk of distant recurrence at 5 and 10 years. The authors in this study selected patients for adjuvant chemotherapy utilizing 70-gene signature assay in addition to standard clinicopathological criteria and prospectively reported 5-year outcomes in the treatment groups.
MINDACT (Microarray in Node-Negative and 1 to 3 Positive Lymph Node Disease May Avoid Chemotherapy) study is a international, prospective, randomized, phase III study which enrolled 6693 women with early-stage breast cancer and determined their genomic risk using the 70-gene signature assay (MammaPrint®) and their clinical risk using a modified version of Adjuvant! Online. Enrolled patients were divided into four groups based on their clinical and genomic risk: low clinical risk and low genomic risk (N=2745, 41%); high clinical risk and high genomic risk (N=1806, 27%), high clinical risk and low genomic risk (N=1550, 23.2%); low clinical risk and high genomic risk (N=592, 8.8%). Patients in the first 2 concordant groups were treated according to their risk category, ie. low-risk group did not receive adjuvant chemotherapy whereas adjuvant chemotherapy was added to endocrine therapy following surgery, in the high-risk patient group. Patients in the last 2 discordant groups were randomly assigned to receive adjuvant chemotherapy or no chemotherapy. The median age of the patients was 55 yrs, 79% of the patients had node-negative disease and 21% had 1-3 positive nodes. A total of 88% of the tumors expressed ER, PR, or both, and 9.5% were HER-2 positive.
The primary endpoint of this study was to show noninferiority against a predefined benchmark of a 5-year metastasis-free survival rate of 92%, in patients at high clinical risk, for whom a discordant low genomic risk led to the omission of otherwise standard adjuvant chemotherapy. It was noted that in this group of patients at high clinical risk and low genomic risk (N=1550) at 5 years, the rate of survival without distant metastasis was 94.7% among those not receiving chemotherapy, and this met the study criterion for noninferiority, with similar outcomes noted in all sub groups of patients.. The absolute difference in the survival rate between those who did not receive adjuvant chemotherapy and those who received chemotherapy was 1.5%, with the survival rate slightly higher in those who received adjuvant chemotherapy. The 5-year metastasis-free survival rate for women who were low risk by both genomic and clinical criteria and who did not receive adjuvant chemotherapy was 97.6%, compared with 90.6% for those women who were high risk by both criteria and who did receive adjuvant chemotherapy.
It was concluded that in patients with early stage breast cancer, who were considered to be at high clinical risk for recurrence, 70-gene signature assay was able to identify those with low genomic risk, who had marginal benefit with adjuvant chemotherapy. With treatment guidance using 70-gene signature assay, approximately 46% of early stage breast cancer patients with high clinical risk and low genomic risk might not require adjuvant chemotherapy. 70-Gene Signature as an Aid to Treatment Decisions in Early-Stage Breast Cancer. Cardoso F, van’t Veer LJ, Bogaerts J, et al. N Engl J Med 2016; 375:717-729
Anthracycline Regimen Superior in High Risk Early Stage Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. The superiority of Anthracycline based chemotherapy regimens for the treatment of breast cancer was demonstrated in the mid 1980's. The Early Breast Cancer Trialists Collaborative Group (EBCTCG) overview analysis published in the Lancet in 1998 concluded that there was a 12% proportional reduction in the risk of recurrence and 11% proportional reduction in mortality with Anthracycline containing regimens versus non-Anthracycline containing chemotherapy regimens. There is however a small risk of cardiotoxicity even with cumulative doses of Doxorubicin of less than 550 mg/m2. Jones and colleagues in 2009 published the results of US Oncology Research Trial 9735 which compared TC with AC and concluded that TC is superior to AC chemotherapy regimen and would be a reasonable option for both younger and older patients requiring chemotherapy, who are hormone receptor positive or negative with either node negative disease or have 1-3 positive lymph nodes.
The ABC (Anthracyclines in early Breast Cancer) adjuvant phase III trials (USOR 06-090, NSABP B-46I/USOR 07132, NSABP B-49) done in sequence, were developed by USOR and NSABP to determine if a regimen of TC for 6 cycles was non-inferior to combination regimens of Doxorubicin/Cyclophosphamide with Docetaxel or Paclitaxel (TaxAC), in patients with resected, high risk, HER2-negative breast cancer. The final analysis set from these collective trials known as ABC included 4130 patients, of whom 2078 patients were randomized to TC and 2052 patients to TaxAC. The treatment groups were well balanced. Sixty nine percent (69%) were hormone receptor positive, 41% were node negative and 51% had high grade tumors. The Primary Endpoint was invasive Disease Free Survival (iDFS) and the median follow up was 3.2 years.
At the time of pre-planned analysis with 399 invasive Disease Free Survival events, the 4 year DFS was significantly higher with TaxAC (90.7%) compared to 88.2% with TC (P=0.04). TaxAC provided little or no added benefit in hormone receptor positive and node negative patients. There was some benefit for patients with hormone receptor positive disease with 1-3 positive lymph nodes and those with hormone receptor negative disease with negative nodes. The most benefit was seen with TaxAC in patients with hormone receptor positive disease with 4 or more positive lymph nodes and in those with hormone receptor negative disease with positive nodes. The 4 year Overall Survival was comparable in both treatment groups although longer follow up is needed.
It can be concluded based on these findings that in early stage breast cancer, Anthracycline containing regimens are superior to non-Anthracycline regimens in patients with triple negative breast cancer and for those hormone receptor positive patients with 4 or more positive lymph nodes. There may be some benefit in select group of hormone receptor positive patients with 1-3 positive lymph nodes and in some patients with node negative, hormone receptor negative disease. Non-Anthracycline regimen such as TC is appropriate in node negative, hormone receptor positive patients. Interim joint analysis of the ABC (anthracyclines in early breast cancer) phase III trials (USOR 06-090, NSABP B-46I/USOR 07132, NSABP B-49 [NRG Oncology]) comparing docetaxel + cyclophosphamide (TC) v anthracycline/taxane-based chemotherapy regimens (TaxAC) in women with high-risk, HER2-negative breast cancer. Blum JL, Flynn PJ, Yothers G, et al. J Clin Oncol 34, 2016 (suppl; abstr 1000)
Late Breaking Abstract - ASCO 2016: Biosimilar of HERCEPTIN® Equally Effective and Safe
SUMMARY: A Biosimilar product is a biological product that is approved based on its high similarity to an already approved biological product (also known as reference product). Biological products are made from living organisms including humans, animals and microorganisms such as bacteria or yeast and are manufactured through biotechnology, derived from natural sources or produced synthetically. Biological products have larger molecules with a complex structure than conventional drugs (also known as small molecule drugs). Unlike biological products, conventional drugs are made of pure chemical substances and their structures can be identified. A generic drug is a copy of brand name drug and has the same active ingredient and is the same as brand name drug in dosage form, safety and strength, route of administration, quality, performance characteristics and intended use. Therefore, brand name and the generic drugs are bioequivalent.  The Affordable Care Act in 2010 created an abbreviated licensure pathway for biological products that are demonstrated to be “Biosimilar” to, or “interchangeable” with an FDA-licensed (FDA approved) biological product (reference product). The Biosimilar must show that it has no clinically meaningful differences in terms of safety and effectiveness from the reference product. A Biosimilar product can only be approved by the FDA if it has the same mechanism of action, route of administration, dosage form and strength as the reference product, and only for the indications and conditions of use that have been approved for the reference product. Biosimilars are not as easy to manufacture as generics (copies of brand name drugs) because of the complexity of the structure of the biologic product and the process used to make a biologic product. The facilities where Biosimilars are manufactured must also meet the FDA’s standards.
The Affordable Care Act in 2010 created an abbreviated licensure pathway for biological products that are demonstrated to be “Biosimilar” to, or “interchangeable” with an FDA-licensed (FDA approved) biological product (reference product). The Biosimilar must show that it has no clinically meaningful differences in terms of safety and effectiveness from the reference product. A Biosimilar product can only be approved by the FDA if it has the same mechanism of action, route of administration, dosage form and strength as the reference product, and only for the indications and conditions of use that have been approved for the reference product. Biosimilars are not as easy to manufacture as generics (copies of brand name drugs) because of the complexity of the structure of the biologic product and the process used to make a biologic product. The facilities where Biosimilars are manufactured must also meet the FDA’s standards.
Heritage is a double-blind, randomized phase III trial in which the efficacy and safety of Myl-1401O, a Biosimilar, was compared with HERCEPTIN®. The randomization included 500 patients treated at 95 sites worldwide, with centrally confirmed, measurable HER2 positive metastatic breast cancer, who had not received prior chemotherapy or HERCEPTIN® for their metastatic disease. Patients received either Myl-1401O or HERCEPTIN® along with TAXOTERE® (Docetaxel) or TAXOL® (Paclitaxel) administered every 3 weeks for a minimum of 8 cycles (24 weeks), with the antibody therapy continued, until disease progression. Both antibodies were administered with a loading dose of 8 mg/kg and a maintenance dose of 6 mg/kg every 3 weeks. Approximately 44% of the enrolled patients had hormone receptor positive disease and 84% received TAXOTERE®. The final analysis included 458 patients of whom 230 were in the Myl-1401O group and 228 were in the HERCEPTIN® group. The Primary endpoint was Overall Response Rate (ORR) at 24 weeks and Secondary endpoints include Progression Free Survival (PFS), Overall Survival (OS) and Safety.
The ORR after 24 weeks of treatment was 69.6% for the Myl-1401O group and 64% for the HERCEPTIN® group and this was not statistically significant. The median PFS had not yet been reached. Safety data in both treatment groups were comparable and there was no significant change in cardiac function from baseline to Week 24 in either group. The dose-normalized maximum concentration and Area Under the Curve, were similar for both antibodies.
The authors concluded that this study is one of the first trials of Biosimilars in oncology to demonstrate similar results and they added that MYL-1401O is equivalent to HERCEPTIN®, when given in combination with a Taxane, as first line therapy, for patients with HER2 positive metastatic breast cancer. Heritage: A phase III safety and efficacy trial of the proposed trastuzumab biosimilar Myl-1401O versus Herceptin. Rugo HS, Barve A, Waller CF, et al. J Clin Oncol 34, 2016 (suppl; abstr LBA503)
Ovarian Suppression in Premenopausal Women with ER Positive Breast Cancer - ASCO Clinical Practice Guideline Update
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. 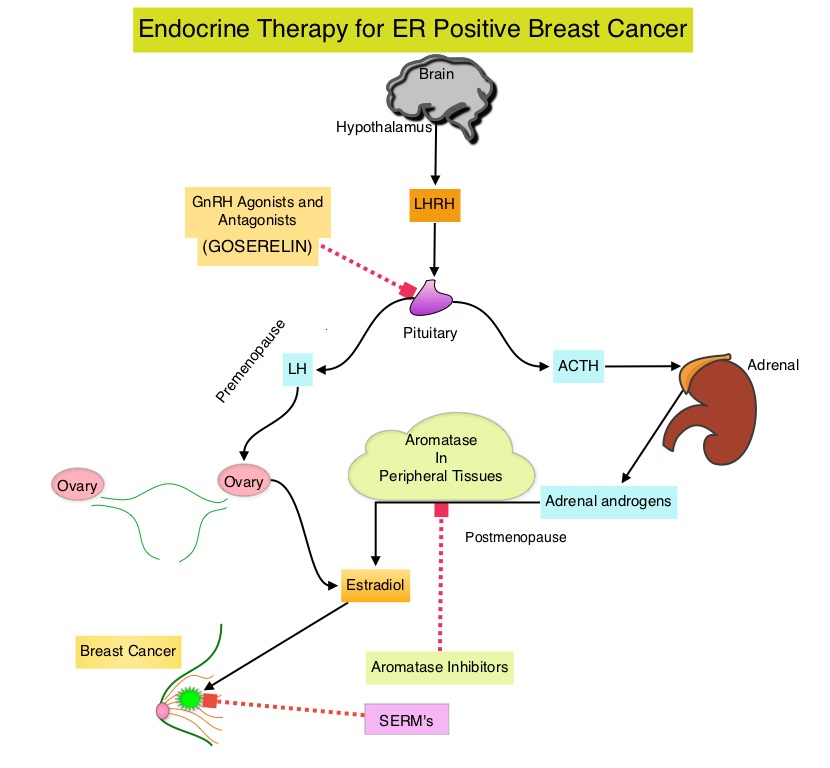 These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues.
These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues.
An Update Panel of the ASCO conducted a systematic review of randomized clinical trials investigating ovarian suppression and published this update. This information is an update of the ASCO adjuvant endocrine therapy guideline, regarding the risks and benefits of ovarian suppression in addition to standard adjuvant therapy, in premenopausal women with Estrogen Receptor positive breast cancer. The ASCO Update Panel addressed the following questions with regard to premenopausal women with stage I-III hormone receptor-positive Breast Cancer:
1) Should premenopausal women with ER positive tumors receive adjuvant ovarian suppression in addition to standard adjuvant therapy, and, if so, in which subsets of patients?
2) If ovarian suppression is recommended, should ovarian suppression be administered in combination with Tamoxifen or an Aromatase Inhibitor?
Recommendation 1
1.1 The panel recommends that higher risk patients should receive ovarian suppression in addition to adjuvant endocrine therapy, whereas lower-risk patients should not. (For women with higher risk cancers who receive chemotherapy but remain premenopausal, ovarian suppression added to Tamoxifen reduces the risk of breast cancer recurrence).
1.2 Women with stage II or III Breast Cancers who would ordinarily be advised to receive adjuvant chemotherapy should receive ovarian suppression in addition to endocrine therapy.
1.3 Women with stage I or II breast cancers at higher risk of recurrence, who might consider chemotherapy, may also be offered ovarian suppression in addition to endocrine therapy.
1.4 Women with stage I breast cancers not warranting chemotherapy should receive endocrine therapy but not ovarian suppression.
1.5 Women with node-negative cancers 1 cm or less (T1a, T1b) should receive endocrine therapy but not ovarian suppression.
Qualifying Statements
• The standard duration of ovarian suppression in the included trials was 5 years. The panel therefore supports ovarian suppression for 5 years as there is no comparative data available on alternative durations.
• To date, there is no adequate evidence for assessing the benefit of adjuvant ovarian suppression in women at sufficient risk to warrant chemotherapy compared with 10 years of Tamoxifen.
• There is no current role for ovarian suppression as adjuvant therapy in ER negative breast cancers.
• There are substantial adverse effects related to ovarian suppression. Clinicians and patients should take this into consideration when choosing ovarian suppression.
• The long-term effects of ovarian suppression on breast cancer risk and survival are not yet established.
Recommendation 2
Ovarian suppression may be administered with either Tamoxifen or an Aromatase Inhibitor.
Qualifying Statements
• Tamoxifen and Aromatase Inhibitor therapy differ in their side effect profiles, which may affect patient preferences.
• Clinicians should be alert to the possibility of incomplete ovarian suppression with Gonadotropin-Releasing Hormone agonist therapy and evaluate patients for whom there is concern about residual ovarian function.
Burstein HJ, Lacchetti C, Anderson H, et al: Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American Society of Clinical Oncology clinical practice guideline update on ovarian suppression. J Clin Oncol 2016;34:1689-1701
ASCO Guidelines on Use of Biomarkers in Early Stage Breast Cancer - Part II
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant therapy. Tumor biomarker assays have become an integral part of the treatment decision making process along with clinical and histologic tumor characteristics, further enabling customized care for patients with early-stage invasive breast cancer. Developed by an expert panel based on systematic reviews, meta-analyses, randomized controlled trials, prospective-retrospective studies and prospective comparative observational studies published from 2006 through 2014, these recommendations are meant to provide guidance to the Health Care Provider, as appropriate treatment is considered for patients with newly diagnosed, early-stage invasive breast cancer.
Two important questions were addressed by these guidelines - The Part I edition last week (www.oncoprescribe.com) addressed the first clinical question. This week’s edition (Part II) addresses the second clinical question.
Clinical Question 2: For women with early-stage invasive breast cancer and with known estrogen receptor/progesterone receptor and HER2 status, which additional biomarkers have demonstrated clinical utility to guide the choice of specific drugs or regimens for adjuvant systemic therapy?
Tamoxifen
CYP2D6 polymorphisms should not be used to guide adjuvant endocrine therapy selection. The expression of p27 by IHC should not be used to guide adjuvant endocrine therapy selection.
Aromatase Inhibitors
Protein encoded by the MKI67 gene labeling index by IHC should not be used to guide adjuvant endocrine therapy.
Taxanes
Microtubule-associated protein Tau mRNA expression or mRNA expression by IHC should not be used to guide adjuvant chemotherapy selection. HER1/Epidermal Growth Factor Receptor expression by IHC should not be used to guide adjuvant chemotherapy selection.
Anthracyclines
TOP2A gene amplification or TOP2A protein expression by IHC should not be used to guide adjuvant chemotherapy selection. HER2 and TOP2A gene coamplification, CEP17 duplication, TIMP-1, FOXP3, or p53 should not be used to guide adjuvant chemotherapy selection.
Trastuzumab
If a patient has HER2 positive breast cancer, PTEN should not be used to guide adjuvant therapy selection. If a patient has HER2 positive breast cancer, soluble HER2 levels should not be used to guide the selection of the type of adjuvant therapy.
Harris LN, Ismaila N, McShane LM, et al: Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2016;34:1134-1150.
ASCO Guidelines on Use of Biomarkers in Early Stage Breast Cancer – Part 1
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant therapy. Tumor biomarker assays have become an integral part of the treatment decision making process along with clinical and histologic tumor characteristics, further enabling customized care for patients with early-stage invasive breast cancer. A multitude of biomarker assays are presently available for the practicing Health Care Provider. Choosing the appropriate biomarker assay for a given patient can be a daunting task and the ASCO guidelines set forth herein, were developed by an expert panel based on systematic reviews, meta-analyses, randomized controlled trials, prospective-retrospective studies, and prospective comparative observational studies, published from 2006 through 2014. These guidelines are only applicable for patients with newly diagnosed, non-metastatic, primary breast cancer, to prognosticate and predict outcomes but they do not however comment on the choice of specific treatment or regimens based on recurrence score. Treatment decisions should take into consideration disease stage, comorbidities and patient preferences. Even though several tests are now recommended in the guidelines, only one test should be used to guide therapy for an individual patient.
Two important questions were addressed by these guidelines - This edition (Part 1) addresses the first Clinical Question
Clinical Question 1: For women with early-stage invasive breast cancer and with known Estrogen receptor/Progesterone receptor and HER2 status, which other biomarkers have demonstrated clinical utility to guide decisions on the need for adjuvant systemic therapy?
Oncotype DX
If a patient has ER/PR positive, HER2 negative, node negative breast cancer, the Oncotype DX 21-gene recurrence score may be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with ER/PR positive, HER2 negative, node positive disease. It should not be used in patients with HER2 positive or triple negative disease.
PAM50 Risk of Recurrence Score
If a patient has ER/PR positive, HER2 negative, node-negative breast cancer, the PAM50 Risk of Recurrence score may be used in conjunction with other clinicopathologic variables to guide decisions on adjuvant systemic therapy. It should not be used in patients with ER/PR positive, HER2 negative, node-positive disease. It should not be used in patients with HER2 positive breast cancer and those with triple-negative breast cancer to guide decisions on adjuvant systemic therapy.
EndoPredict
If a patient has ER/PR positive, HER2 negative, node-negative breast cancer, EndoPredict 12-gene risk score may be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with ER/PR positive, HER2 negative, node-positive disease. It should not be used in patients with HER2 positive or triple-negative disease.
Breast Cancer Index
If a patient has ER/PR positive, HER2 negative, node-negative breast cancer, the Breast Cancer Index may be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with ER/PR positive, HER2 negative, node-positive disease. It should not be used in patients with HER2 positive or triple negative breast cancer to guide decisions on adjuvant systemic therapy.
Urokinase Plasminogen Activator and Plasminogen Activator Inhibitor Type 1
If a patient has ER/PR positive, HER2 negative, node negative breast cancer, Urokinase Plasminogen Activator and Plasminogen Activator Inhibitor Type 1 may be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with HER2 positive or triple negative breast cancer.
MammaPrint
If a patient has ER/PR positive, HER2 negative (node-positive or node-negative) breast cancer, the MammaPrint 70-gene assay should not be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with HER2 positive disease. It should not be used in patients with triple negative breast cancer.
Mammostrat
If a patient has ER/PR positive, HER2 negative (node-positive or node-negative) breast cancer, the Mammostrat 5-protein assay should not be used to guide decisions on adjuvant systemic therapy. It should not be used in patients with HER2 positive or triple negative breast cancer.
Immunohistochemistry 4
If a patient has ER/PR positive, HER2 negative (node-positive or node-negative) breast cancer, Immunohistochemistry 4 (IHC4) should not be used to guide decisions on adjuvant systemic chemotherapy. It should not be used in patients with HER2 positive or triple negative breast cancer.
Circulating Tumor Cells
The clinician should not use circulating tumor cells to guide decisions on adjuvant systemic therapy.
Tumor-Infiltrating Lymphocytes
If a patient has ER/PR positive, HER2 negative (node-positive or node-negative) breast cancer, Tumor-Infiltrating Lymphocytes should not be used for decision making. It should not be used in patients with HER2 positive or triple negative breast cancer.
Protein Encoded by MKI67 Gene
Protein encoded by the MKI67 gene labeling index by IHC should not be used to guide choice on adjuvant chemotherapy.
Extended Endocrine Therapy
If a patient has ER/PR positive, HER2 negative (node-negative) breast cancer and has had 5 years of endocrine therapy without evidence of recurrence, multiparameter gene expression or protein assays (Oncotype DX, EndoPredict, PAM50, Breast Cancer Index, or IHC4) should not be used to guide decisions on extended endocrine therapy.
The Clinical Question 2 will be addressed in the eNL edition (Part 2) next week.
Harris LN, Ismaila N, McShane LM, et al: Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2016;34:1134-1150.
Late Breaking Abstract - ASCO 2016: Extended Adjuvant AI Therapy Improves DFS in Postmenopausal Hormone Receptor Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. Postmenopausal women with hormone receptor positive early breast cancer are often treated with 5 years of Aromatase Inhibitor (AI) therapy either as up-front treatment or after 2-5 years of Tamoxifen. The benefit of extending treatment with an AI to 10 years may further reduce the risk of breast cancer recurrence, but this benefit was not previously known.
The Canadian Cancer Trials Group MA.17R is a double blind, placebo controlled, phase III trial, which tested the benefit of extending AI treatment, using FEMARA®, for an additional 5 years. This study involved 1,918 postmenopausal women with early stage breast cancer and included three patient groups – one group had no prior treatment with adjuvant Tamoxifen whereas the other two groups had adjuvant Tamoxifen for some duration of time. All patients however had recently received 4.5 to 6 years of adjuvant AI therapy. These patients were then randomly assigned to receive either extended adjuvant treatment with FEMARA® or placebo for an additional five years. The primary endpoint was Disease Free Survival (DFS).
After a median follow up of 6.3 years, the 5 year DFS rate for the extended FEMARA® group was 95% compared with 91% for the placebo group (HR=0.66; P=0.01). The improvement in DFS was significant among patients with node-positive disease, but not for those with node-negative disease. There was no difference in the 5 year Overall Survival between the two groups - 93% with FEMARA® versus 94% with placebo (HR 0.97; P=NS). The annual incidence rate of contralateral breast cancer was however significantly better in the FEMARA® group at 0.21%, compared with 0.49% with placebo (HR=0.42; P=0.007). Patients receiving extended treatment with FEMARA® had more frequent adverse events such as bone pain, elevation of alkaline phosphatase, and elevation of alanine transaminase. There was also a greater incidence of osteoporosis with FEMARA® than with placebo (11% vs 6%; P<0.0001) and fracture risk was higher in the FEMARA® group (14%) compared with 9% in the placebo group (P=0.001).
The authors concluded that this is the first study to show added benefit of improved Disease Free Survival, by extending an adjuvant AI beyond 5 years to 10 years, when compared with 5 years of AI treatment as initial therapy or preceded by 2-5 years of Tamoxifen. A randomized trial (MA.17R) of extending adjuvant letrozole for 5 years after completing an initial 5 years of aromatase inhibitor therapy alone or preceded by tamoxifen in postmenopausal women with early-stage breast cancer. Goss PE, Ingle JN, Pritchard KI, et al. J Clin Oncol 34, 2016 (suppl; abstr LBA1)
Adjuvant AROMASIN® Most Beneficial for Premenopausal Women with High Risk Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. Presently available therapies include Tamoxifen and other Selective Estrogen Receptor (ER) Modulators, which modulate ER alpha activity, Aromatase Inhibitors (AIs) and Ovarian ablation that decrease estrogen production and FASLODEX® (Fulvestrant) that down regulates Estrogen Receptor. Aromatase Inhibitors are often prescribed due to their superiority over Tamoxifen, for postmenopausal women with Hormone Receptor positive breast tumors, in adjuvant as well as metastatic settings. Aromatase Inhibitors by themselves however, are not effective in premenopausal women, as these individuals derive their estrogen mainly from ovaries and not extragonadal tissue.
The TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) are two phase III randomized trials, conducted at the same time and included premenopausal women (average age was 43 years) with hormone receptor positive, early breast cancer. In the joint analysis of these two trials which included 4,891 women, the authors set out to answer 2 important questions - whether adjuvant AI treatment improves outcomes in this patient group, when their Ovarian Function is suppressed and whether there is any benefit with Ovarian Function suppression in premenopausal women suitable for adjuvant Tamoxifen. TEXT randomized patients within 3 months of surgery to 5 years of AROMASIN® (Exemestane) plus Ovarian Function Suppression (OFS) or 5 years of Tamoxifen plus OFS. The SOFT study randomized patients to 5 years of AROMASIN® plus OFS or 5 years of Tamoxifen plus OFS or 5 years of Tamoxifen alone. OFS choices included oophorectomy, ovarian irradiation or 5 years of TRELSTAR® (Triptorelin), a GnRH (Gonadotropin Releasing Hormone) agonist. The 5 year Disease Free Survival was 91.1% in the AROMASIN® plus OFS group and 87.3% in the Tamoxifen plus OFS group (HR=0.72, P<0.0002). Compared to patients receiving Tamoxifen plus OFS, AROMASIN® plus OFS reduced the relative risk of premenopausal women developing a subsequent invasive breast cancer by 28% and the relative risk of breast cancer recurrence by 34%.
The authors in this analysis examined the absolute treatment effect in the TEXT and SOFT trials across a continuum of recurrence risk, to help individualize decision making for endocrine therapy, in premenopausal women with Human Epidermal growth factor Receptor 2 (HER2) -negative disease. Incorporating age, nodal status, tumor size, grade, Ki-67 expression levels and hormone receptor status, a composite recurrence risk for each patient was determined, from a Cox model.
It was noted that patients in the SOFT trial who remained premenopausal after chemotherapy experienced absolute improvement of 5% or more in 5-year Breast Cancer-Free Interval with AROMASIN® plus OFS compared with Tamoxifen plus OFS or Tamoxifen alone, and this benefit was even higher, reaching 10% to 15% for the intermediate to high composite recurrence risk group of patients. Patients in the SOFT trial whose composite recurrence risk was low did not receive chemotherapy and did well with all endocrine therapies. For patients in the TEXT trial, the benefit of AROMASIN® plus OFS compared with Tamoxifen plus OFS was similar to the SOFT trial, with the 5-year Breast Cancer-Free Interval ranging from 5-15%. Again, patients not receiving chemotherapy and with lowest composite recurrence risk did well with both endocrine therapies.
The authors concluded that premenopausal women with hormone receptor-positive, HER2-negative disease, with high risk for recurrence based on clinicopathologic features, may experience a 10% to 15% improvement in the 5-year Breast Cancer-Free Interval with AROMASIN® plus OFS compared with Tamoxifen alone. Absolute Benefit of Adjuvant Endocrine Therapies for Premenopausal Women with Hormone Receptor–Positive, Human Epidermal Growth Factor Receptor 2–Negative Early Breast Cancer: TEXT and SOFT Trials.Regan MM, Francis PA, Pagani O, et al. Published online before print April 4, 2016, doi: 10.1200/JCO.2015.64.3171 JCO April 4, 2016 JCO643171
Five Year Follow up Data without Adjuvant Chemotherapy, Utilizing Oncotype DX 21-gene Recurrence Score Assay
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant chemotherapy. The Oncotype DX breast cancer assay, is a multigene genomic test that analyzes the activity of a group of 21 genes and is able to predict the risk of breast cancer recurrence and likelihood of benefit from systemic chemotherapy, following surgery, in women with early stage breast cancer. Chemotherapy recommendations for early stage, hormone receptor positive, HER negative breast cancer patients, are often made based on tumor size, grade, immunohistochemical markers such as Ki-67, nodal status and 21-gene Recurrence Score (RS) assay.
The West German Study Group (WSG) PlanB phase III trial, used Recurrence Score assay prospectively, to define a low risk subset of patients with node negative disease with high risk traditional parameters and patients with node positive disease (HR+, HER2 negative), who could be treated with adjuvant endocrine therapy alone, sparing chemotherapy. In this study, patients with a Recurrence Score of 11 or less were defined as having low risk for recurrence, even in those considered to have tumor with high risk features such as large tumor size, high tumor grade and lymph node involvement. Patients were considered to be at intermediate or high risk if they had a Recurrence Score of 12 or more and 25 or more, respectively. Gluz and colleagues had previously reported 3-year follow up results of a planned interim analysis of this study, and were able to show significant difference between tumor grade, Ki-67 and Oncotype DX Recurrence Score.
In this current analysis, the authors reported the 5-year Disease Free Survival (DFS) outcomes of this large prospective trial. This analysis included data from 3,198 patients with early stage hormone receptor positive or HER2 negative breast cancer. The median age was 56 years and 32.5% of the patients had grade 3 tumors and 41% of the patients had node positive disease. Patients with a Recurrence Score of 11 or less received hormonal therapy and adjuvant chemotherapy was omitted. The intermediate and high risk patients were randomized to receive six cycles of TAXOTERE® (Docetaxel)/CYTOXAN® (Cyclophosphamide) or four cycles of ELLENCE® (Epirubicin)/CYTOXAN® followed by four cycles of TAXOTERE®. The primary endpoint was Disease Free Survival (DFS) defined as invasive or noninvasive relapse.
It was noted that the 5-year DFS in the low risk group was 94%, 84% in the high risk group and 94% in the intermediate risk group (P<0.001). It should be noted that approximately 15% of the patients in the clinically determined intermediate or high risk group, with 0-3 lymph node involvement, fell in the low genomic risk group (Recurrence Score of 11 or less) and received hormonal therapy alone.
The authors concluded that West German Study Group (WSG) PlanB study is the first trial that has reported five year survival data using 21-gene Recurrence Score assay, which can identify patients with early breast cancer, who would benefit from hormonal therapy alone and could be spared chemotherapy. Based on these data, 21-gene Recurrence Score has stronger prognostic value compared to immunohistochemical studies such as Ki-67 and hormone-receptor expression and should therefore be routinely incorporated in clinical practice as a decision making tool for this patient population, in addition tumor size, grade and nodal status. Prospective WSG Phase III PlanB trial: Clinical outcome at 5 year follow up and impact of 21 Gene Recurrence Score result, central/local-pathological review of grade, ER, PR and Ki67 in HR+/HER2- high risk node-negative and –positive breast cancer. Gluz O, Nitz U, Christgen M, et al. Abstract 8LBA. Presented at: 10th European Breast Cancer Conference; March 9-11, 2016; Amsterdam.
Impact of Treatment Delay on Clinical Outcomes in Breast Cancer Patients
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Patients with early stage breast cancer often receive adjuvant chemotherapy and this is even more so true for HER positive and triple negative (ER, PR and HER negative) breast cancer patients, who are at an increased risk to develop recurrent disease. Even though majority of the patients start their adjuvant chemotherapy within 4-6 weeks following surgery, the impact of delay in the initiation of adjuvant therapy on outcomes, has remained unclear. Preclinical models have suggested that there is phase of increased angiogenesis and accelerated growth of micrometastases, as well as development of drug resistant clones, following removal of the primary tumor. Previously published data from a large meta-analysis had suggested that a four week delay in the initiation of adjuvant chemotherapy resulted in a 6% increase in the risk of death and an 8% increase in the risk of relapse. Nonetheless, over the past 2 decades, there has been increasing delay for both surgery and adjuvant chemotherapy treatment intervention, following diagnosis of breast cancer. These delays have been attributed to the increasing use of prognostic tools prior to treatment intervention, in order to optimize breast cancer care, germ-line genetic testing to plan appropriate surgical intervention, as well as patients seeking immediate reconstructive surgical options. Two studies addressed the impact of delay in treatment intervention following diagnosis of breast cancer, by investigating outcomes, in a very large group of patients with breast cancer.
In the study by Bleicher, et al., the relationship between the time from diagnosis to breast cancer surgery and survival was investigated, by collecting data from the Surveillance, Epidemiology, and End Results (SEER)-Medicare linked database and the National Cancer Database (NCDB). The SEER-Medicare cohort had 94,544 patients 66 years or older, diagnosed between 1992 and 2009 and the NCDB cohort included 115,790 patients 18 years or older, diagnosed between 2003 and 2005. Patients included in this analysis underwent surgery as initial treatment and had a diagnosis of non-inflammatory, non-metastatic, invasive breast cancer. The primary outcome was Overall and Disease-Specific Survival, as a function of time between diagnosis and surgery, measured in 30 day increments. They noted that with each interval of treatment delay increase, Overall Survival was lower (HR=1.09; P<0.001 in the SEER-Medicare cohort and HR=1.10; P<0.001 in the NCDB cohort). This relationship was statistically significant only in stages I and II breast cancer. The authors in this study concluded that longer time to surgery is associated with lower Overall and Disease-Specific Survival.
Chavez-MacGregor et al. analyzed the outcomes of 24, 843 patients in the California Cancer Registry with stage I-III invasive breast cancer, diagnosed between January 2005 and December 2010, and treated with adjuvant chemotherapy. Time to chemotherapy was defined as the number of days between surgery and the first dose of chemotherapy, and delayed treatment was defined as 91 or more days from surgery to the first dose of adjuvant chemotherapy. Median age at the time of diagnosis was 53 years, and median time to adjuvant chemotherapy was 46 days. Patients were evaluated for Overall Survival and Breast Cancer-Specific Survival. They noted that patients receiving adjuvant chemotherapy 91 or more days following surgery experienced worse Overall Survival (HR=1.34) and worse Breast Cancer-Specific Survival (HR=1.27) compared with patients receiving adjuvant chemotherapy within 31 days from surgery and these adverse outcomes were even more so, among patients with triple negative breast cancer (HR=1.53). Factors associated with adjuvant therapy delays included, low socioeconomic status, breast reconstruction, non-private insurance, and Hispanic or African American ethnicity. The authors in this study concluded that delaying initiation of adjuvant chemotherapy 91 days or more, results in adverse outcomes and this may be even more detrimental, in patients with triple negative breast cancer.
These two studies strongly suggest that treatment delays should be avoided in patients with early stage breast cancer and if surgery is to be delayed, neoadjuvant treatment approach may be reasonable, to avoid adverse outcomes.
Time to Surgery and Breast Cancer Survival in the United States. Bleicher RJ, Ruth K, Sigurdson ER, et al. JAMA Oncol. 2016;2:330-339.
Delayed Initiation of Adjuvant Chemotherapy Among Patients With Breast Cancer. Chavez-MacGregor M, Clarke CA, Lichtensztajn DY, et al. JAMA Oncol. 2016;2:322-329.
Aromatase Inhibitors-Induced Carpal Tunnel Syndrome
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their life time. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and 40,450 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol, in extragonadal/peripheral tissues. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole), FEMARA® (Letrozole) and AROMASIN® (Exemestane) are Aromatase Inhibitors (AIs) that binds to the Aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. The use of Aromatase Inhibitors (AIs) has been long associated with musculoskeletal symptoms, as well as accelerated bone loss, leading to a decrease in Bone Mineral Density (BMD). Approximately 25% of the patients on AIs are non-compliant during the first year of therapy and this has been attributed to musculoskeletal symptoms. Increased risk of Carpal Tunnel Syndrome (CTS) has also been reported with AIs.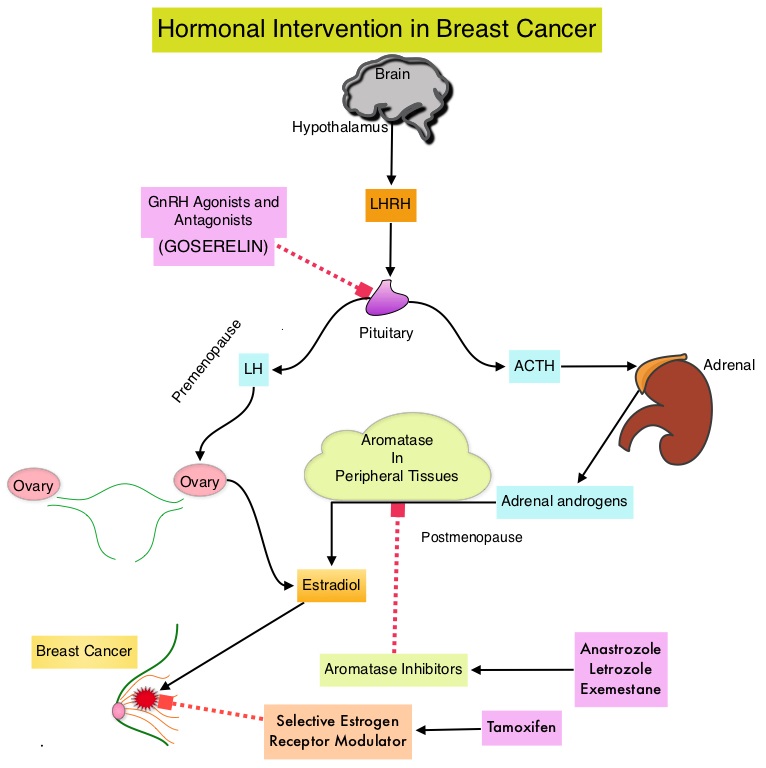
Carpal tunnel syndrome (CTS), also called median nerve compression, is the most common entrapment neuropathy and results from compression of the median nerve, as it runs from the forearm into the palm of the hand, through the carpal tunnel. The carpal tunnel is a narrow and rigid passage at the base of the hand and houses the median nerve and tendons. In most cases, CTS is due to a congenital predisposition, with the carpal tunnel being simply smaller in some individuals than others. Secondary CTS is caused by any condition that further narrows this osteofibrous passage such as arthritis, acromegaly and mechanical problems in the wrist joint or effects the contents of this passage such as tenosynovitis, synovial hypertrophy, hypothyroidism, fluid retention during pregnancy or menopause, or the development of a cyst or tumor in the passage. Bilateral oophorectomy and use of the combined oral contraceptive have also been identified as risk factors for CTS. Patients often experience nocturnal paraesthesias in median nerve distribution (thumb, index and middle fingers), such as burning sensation, tingling, or heaviness, with the pain radiating to the forearm or elbow. It has been postulated that estrogen has antinociceptive properties and estrogen deprivation with AIs decreases the threshold for pain stimuli. Estrogen deprivation may also impact the metabolism of transverse carpal ligament on which estrogen and progesterone receptors are expressed and lack of estrogen may additionally result in morphological changes in the contents of carpal tunnel including, thickening of the tendon sheaths and fluid accumulation. This may directly induce CTS.
To address the risk factors and incidence of CTS in women taking AIs, the authors conducted an exploratory analysis of the International Breast Cancer Intervention Study II, a double-blind randomized clinical trial, in which women at increased risk of breast cancer were randomly assigned to receive ARIMIDEX® or placebo for 5 years. In this study, a total of 3,864 women were randomly assigned to receive either ARIMIDEX® (N=1920) or placebo (N=1944). The median age was 60 years and majority of the women (69%) had a BMI of greater than 25 kg/m2.
After a median follow up of 6.4 years, 96 patients had symptoms of Carpal Tunnel Syndrome (CTS). Patients receiving ARIMIDEX® were more likely to have CTS related symptoms than those receiving placebo (3.4% versus 1.6%; P<0.001). Eight of the 10 participants reported as having severe CTS were taking ARIMIDEX® (P =0.08). Eighteen women (0.9%) in the ARIMIDEX® group required surgical intervention for CTS compared to six women (0.3%) in the placebo group and this was significantly different (P=0.018). Participants experiencing CTS symptoms did so early in the course of treatment and only 6 women discontinued the allocated treatment. In addition to taking AIs, high Body Mass Index and prior complaints of musculoskeletal symptoms, were the only other risk factors for developing CTS.
The authors concluded that the use of ARIMIDEX® was associated with a higher incidence of Carpal Tunnel Syndrome (CTS), although few participants required surgery. Given the association between CTS and other musculoskeletal symptoms induced by AIs (Aromatase Inhibitors), the authors suggested that these findings induced by AIs, may share the same pathobiology. Anastrozole-Induced Carpal Tunnel Syndrome: Results from the International Breast Cancer Intervention Study II Prevention Trial. Spagnolo F, Sestak I, Howell A, et al. J Clin Oncol 2016;34:139-143
FDA Approves IBRANCE® in Combination with FASLODEX® for Advanced Breast Cancer
SUMMARY: The FDA on February 19, 2016, approved IBRANCE® (Palbociclib) in combination with FASLODEX® (Fulvestrant), for the treatment of women with Hormone Receptor (HR)-positive, Human Epidermal growth factor Receptor 2 (HER2)-negative advanced or metastatic breast cancer, with disease progression following endocrine therapy. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 246,660 new cases of invasive breast cancer will be diagnosed in 2016 and over 40,450 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. NOLVADEX® (Tamoxifen) is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. ARIMIDEX® (Anastrozole) and FEMARA® (Letrozole) are nonsteroidal Aromatase Inhibitors that binds reversibly to the aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. Approximately 80% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment.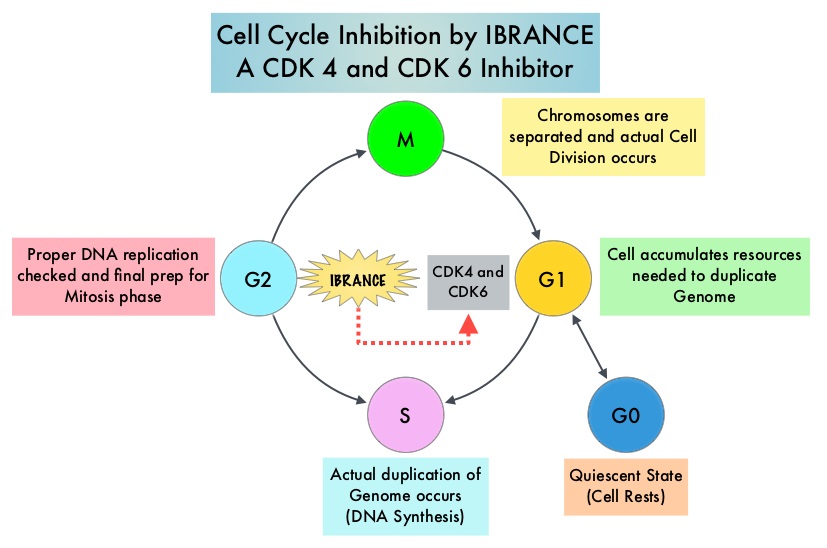
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6), phosphorylate RetinoBlastoma protein (RB), and initiate transition from the G1 phase to the S phase of the cell cycle. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. IBRANCE® (Palbociclib) is a reversible, oral, selective, small molecule inhibitor of Cyclin Dependent Kinases, CDK4 and CDK6, and prevents RB1 phosphorylation. IBRANCE® is the first CDK inhibitor approved by the FDA. It exhibits synergy when combined with endocrine therapies. In an open-label, randomized, phase II study, which included treatment naïve postmenopausal women with ER-positive, HER2-negative, advanced breast cancer, IBRANCE® given along with Aromatase Inhibitor FEMARA® (Letrozole), significantly prolonged Progression Free Survival, Overall Response rate and median duration of response, compared to FEMARA® alone. Based on this data, the U. S. Food and Drug Administration in February 2015, granted accelerated approval to IBRANCE® (Palbociclib), for use in combination with FEMARA®, in this patient population. FASLODEX® (Fulvestrant) is a selective estrogen receptor down-regulator presently indicated for the treatment of hormone receptor positive metastatic breast cancer patients, with disease progression following antiestrogen therapy.
The PALOMA3 is double-blind, phase 3 study in which the efficacy and safety of the combination of IBRANCE® and FASLODEX® was evaluated, in premenopausal or postmenopausal women, with hormone receptor positive, HER-2 negative, advanced breast cancer, who had disease progression during prior endocrine therapy. Five hundred and twenty one (N=521) patients were randomly assigned in a 2:1 ratio to receive either FASLODEX® 500 mg IM on days 1 and 15 during cycle 1, of a 28 day cycle, and then on day 1 of each cycle thereafter, along with IBRANCE® 125 mg PO daily for 3 weeks, followed by 1 week off (N=347) or FASLODEX® and placebo (N=174). ZOLADEX® (Goserelin) was administered to premenopausal or perimenopausal patients for the duration of study treatment, starting at least 4 weeks before randomization and continuing every 28 days. The median age was 57 years. One previous line of chemotherapy for metastatic disease was allowed and 79% were post-menopausal, 60% had visceral disease and 75% of the patients had received a previous chemotherapy regimen.
The primary endpoint was Progression Free Survival (PFS) and secondary endpoints included Overall Survival (OS), Response Rates, safety and tolerability. At the time of the preplanned interim analysis, the median Progression Free Survival was 9.2 months in the FASLODEX® / IBRANCE® group and 3.8 months in the FASLODEX® /placebo group (HR=0.42; P<0.001). This PFS benefit was observed across all prespecified patient subgroups, regardless of menopausal status. The most common grade 3 or 4 adverse events in the FASLODEX® / IBRANCE® group were neutropenia (62.0%, vs. 0.6%) and fatigue (2.0% vs. 1.2%). The incidence of febrile neutropenia was very rare (0.6%) and similar in both treatment groups. Treatment discontinuation rate due to adverse events was 2.6% in the IBRANCE® group and 1.7% in the placebo group.
The authors concluded that IBRANCE® in combination with FASLODEX® more than doubled the Progression Free Survival in advanced breast cancer patients, with hormone receptor positive and HER-2 negative disease, who had progressed on prior endocrine therapy. This study has reinforced the importance of CDK4 and CDK6, as key targets for hormone receptor positive breast cancer. Palbociclib in Hormone-Receptor–Positive Advanced Breast Cancer. Turner NC, Ro J, Andre F, et al. N Engl J Med 2015; 373:209-219
Cardiac Outcomes of Patients Receiving Adjuvant Weekly TAXOL® and HERCEPTIN®
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their life time. Approximately, 246,660 new cases of invasive breast cancer were diagnosed in 2016 and 40,450 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 20-25% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. HERCEPTIN® binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Adjuvant chemotherapy in combination with HERCEPTIN® has been shown to reduce the relative risk of relapse by 52% and relative risk of death by 33%. The National Comprehensive Cancer Network (NCCN) has recommended adjuvant chemotherapy with HERCEPTIN® for patients with small, HER positive, node-negative tumors, including those with T1bN0 tumors, even though there are little or no data supporting this recommendation, because these patients are generally not included in adjuvant therapy studies. Further, the chemotherapy regimens often recommended (ACTH, TCH) along with HERCEPTIN® are relatively toxic.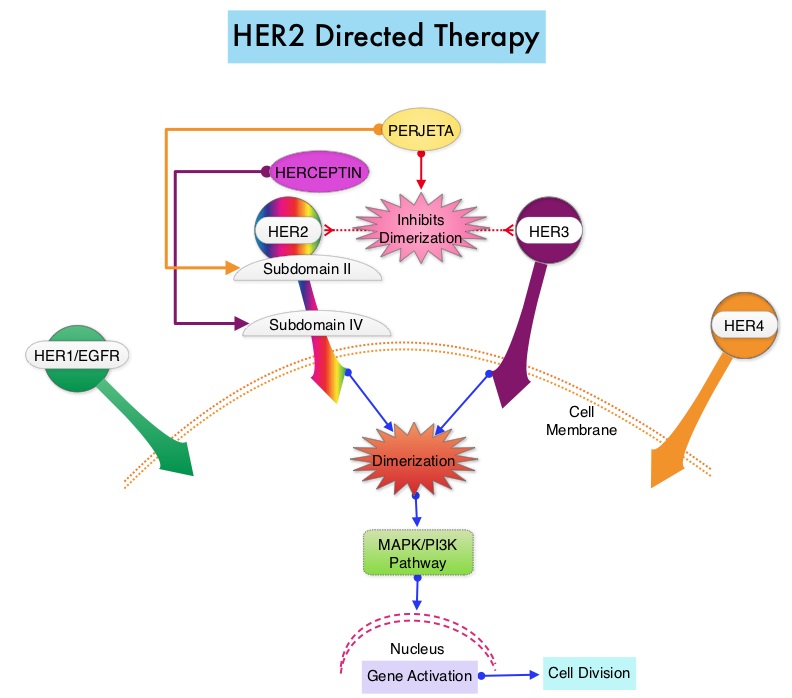
In a previously published study, it was noted that a less toxic regimen such as HERCEPTIN®, given along with weekly TAXOL® (Paclitaxel), had significant efficacy in patients with node negative patients with tumors measuring up to 3 cm in greatest dimension, decreasing the risk of recurrence in this patient group, most notable during the first three years after diagnosis. (Tolaney SM, Barry WT, Dang CT, et al. N Engl J Med 2015;372:134-141). Risk risk factors associated with HERCEPTIN® induced cardiotoxicity include, age over 50 years, borderline LVEF (Left Ventricular Ejection Fraction) before HERCEPTIN® treatment, history of cardiovascular disease, cardiovascular risk factors such as diabetes, dislipidemia or elevated body mass index (>30), sequence in which chemotherapy is administered and prior treatment with Anthracyclines (cumulative doses more than 300 mg/m2). However, unlike Anthracycline induced cardiotoxicity, HERCEPTIN® induced cardiotoxicity is reversible and there are no ultrastructural changes noted in cardiomyocytes in HERCEPTIN® induced cardiotoxicity.
This publication is a secondary analysis of the above mentioned previously published study and the authors here reported the cardiac safety data of a HERCEPIN® based nonanthracycline treatment, (TAXOL® with HERCEPTIN®), for patients with early-stage, node negative, HER2 positive breast cancer and the utility of monitoring LVEF in this patient group. This clinical trial enrolled 406 patients with node-negative, HER2 positive breast cancer 3 cm, or smaller with a baseline LVEF of 50% or greater. Treatment consisted of TAXOL® 80 mg/m2 IV weekly administered concurrently with HERCEPTIN® IV for 12 weeks, followed by HERCEPTIN® monotherapy for 39 weeks. HERCEPTIN® could be administered 2 mg/kg weekly or 6 mg/kg every 3 weeks during the monotherapy phase. Radiation and hormone therapy were administered as planned, following completion of the 12 weeks of chemotherapy. Patient LVEF was assessed at baseline, 12 weeks, 6 months, and 1 year. Median age was 55 years and 29% of the patients had hypertension, and 7% had diabetes. The median follow up was 4 years.
It was noted that a significant, asymptomatic LVEF decline was seen in 3.2% of the patients and 0.5% developed grade 3 Left Ventricular Systolic Dysfunction. The median LVEF at baseline was 65%, at 12 weeks was 64%, at 6 months was 64%; and at 1 year was 64%. The authors concluded that cardiotoxicity from a combination of TAXOL® and HERCEPTIN® is low and a baseline LVEF assessment may be adequate for the majority of patients although serial LVEF assessments could be considered for patients considered at a higher risk for cardiotoxicity. Cardiac Outcomes of Patients Receiving Adjuvant Weekly Paclitaxel and Trastuzumab for Node-Negative, ERBB2-Positive Breast Cancer. Dang C, Guo H, Najita J, et al. JAMA Oncol. 2016;2:29-36
Use of Biomarkers to Guide Decisions on Systemic Therapy for Women with Metastatic Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. The American Society of Clinical Oncology (ASCO) guidelines on the use of tumor markers in breast cancer are meant to provide evidence-based recommendations and guidance to practicing oncologists, on the appropriate use of breast tumor biomarker assays, for management of patients with metastatic breast cancer. These recommendations are for women with metastatic breast cancer being considered for systemic therapy or for changes in the drug or regimen they are receiving.
These guidelines were compiled after reviewing 17 clinical publications following an extensive literature search between 2006 and 2014. They included 11 studies that reported discordance in expression of hormone receptors or HER-2 between primary tumors and metastases, one randomized controlled study that addressed the use of a biomarker to decide whether to continue or change a treatment regimen and 5 prospective and retrospective studies that evaluated the clinical utility of biomarkers.
Should metastases be biopsied or otherwise sampled to test for changes from the primary tumor with respect to ER, PR, or HER2 status?
Patients with accessible, newly diagnosed metastases from primary breast cancer should be offered biopsy for confirmation of disease process and testing of ER, PR, and HER2 status. They should also be informed that if discordances are found, evidence is lacking to determine whether outcomes are better with treatment regimens based on receptor status in the metastases or the primary tumor. With discordance of results between primary and metastatic tissues, the Panel consensus is to preferentially use the ER, PR, and HER2 status from the metastasis to direct therapy, if supported by the clinical scenario and the patient's goals for care.
For women with metastatic breast cancer and with known ER, PR, and HER2 status, which additional tumor markers have demonstrated clinical utility to initiate systemic therapy or direct selection of a new systemic therapy regimen?
Decisions on initiating systemic therapy for metastatic breast cancer should be based on clinical evaluation, judgment, and patient preferences. There is no evidence at this time that initiating therapy solely on the basis of biomarker results beyond those of ER, PR, and HER2 improves health outcomes.
For women with metastatic breast cancer and with known ER, PR, and HER2 status, which additional tumor markers have demonstrated clinical utility to guide decisions on switching to a different drug or regimen or discontinuing treatment?
Recommendations for tissue biomarkers: In patients already receiving systemic therapy for metastatic breast cancer, decisions on changing to a new drug or regimen or discontinuing treatment should be based on clinical evaluation, judgment of disease progression or response, and the patient's goals for care. There is no evidence at this time that changing therapy based solely on biomarker results beyond ER, PR, and HER2 improves health outcomes, quality of life, or cost effectiveness.
Recommendations for circulating tumor markers: In patients already receiving systemic therapy for metastatic breast cancer, decisions on changing to a new drug or regimen or discontinuing treatment should be based on clinical evaluation, judgment of disease progression or response, and the patient's goals for care. There is no evidence at this time that changing therapy based solely on circulating biomarker results improves health outcomes, quality of life, or cost effectiveness. CEA, CA 15-3, and CA 27-29 may be used as adjunctive assessments to contribute to decisions regarding therapy for metastatic breast cancer. Data are insufficient to recommend use of CEA, CA 15-3, and CA 27-29 alone for monitoring response to treatment. As such, it is also reasonable for clinicians to not use these markers as adjunctive assessments.
For biomarkers shown to have clinical utility to guide decisions on systemic therapy for metastatic disease in questions 2 and 3, what are the appropriate assays, timing, and frequency of measurement?
Decisions for systemic therapy should be influenced by ER, PR, and HER2. ASCO recently updated the guideline addressing optimization of HER2 assays. To date, clinical utility has not been demonstrated for any additional biomarkers.
Poznak CV, Somerfield MR, Bast RC, et al. J Clin Oncol 2015;33:2695-2704
Adjuvant PROLIA® Significantly Reduces Fracture Risk for Breast Cancer Patients on Aromatase Inhibitors
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. These patients are often treated with anti-estrogen therapy as first line treatment. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. Tamoxifen is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. Anastrozole,Letrozole and Exemestane are Aromatase Inhibitors that bind reversibly to the aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues. Aromatase inhibitors are associated with accelerated bone loss, leading to a decrease in Bone Mineral Density (BMD) and can thus cause osteopenia and osteoporosis, thereby increasing fracture risk. According to the WHO definitions, a healthy 30 year old adult (young adult) with the ideal Bone Mineral Density (BMD) is given a T-score of 0. A normal BMD is within 1 Standard Deviation-SD (+1 or −1) of the young adult mean. Osteopenia is between 1 and 2.5 SD below the young adult mean (−1 to −2.5 SD). Osteoporosis is 2.5 SD or more below the young adult mean (−2.5 SD or lower).
PROLIA® (Denosumab) is a monoclonal antibody that inhibits osteoclast formation, function and survival by selectively targeting the RANK ligand. In this randomized, double-blind, phase III trial, the authors evaluated the benefits of the anti-RANK ligand antibody PROLIA® (Denosumab) on bone health, in postmenopausal patients, with early stage hormone receptor-positive breast cancer, treated with Aromatase Inhibitors. Of the 3425 enrolled patients, 3420 patients were randomly assigned to receive PROLIA® 60 mg (N=1711) or placebo (N=1709) subcutaneously every 6 months. Majority of the patients participating in this study had breast cancer with good prognosis and only 25% of the patients required adjuvant chemotherapy. Patient received a median of 7 doses of PROLIA® . The primary endpoint was time from randomization to first clinical fracture.
Compared with placebo, PROLIA® significantly delayed time to first clinical fracture (HR=0.50; P<0•0001), with the PROLIA® group, half as likely to have a first clinical fracture as the placebo group. This benefit of lowering fracture risk was seen in subgroups of patients with BMD T-score of less than –1 as well as those with BMD T-score of -1 or greater. Further, patients in the PROLIA® group had improvements in BMD from baseline, of the lumbar spine, total hip, and femoral neck, compared to the placebo group, which showed worsening at all sites (P<0.0001). At 3 years, patients in the PROLIA® group had significantly lower risk of both new, or worsening vertebral fractures. No cases of osteonecrosis of the jaw bone were reported.
The authors concluded that PROLIA® administered in an adjuvant setting, significantly reduces the risk of fractures in postmenopausal women with breast cancer receiving Aromatase Inhibitors, without added toxicity. Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Gnant M, Pfeiler G, Dubsky PC, et al. The Lancet 2015;386:433-443
FDA Approves DigniCap Cooling System for Prevention of Chemotherapy Induced Alopecia
SUMMARY: The FDA on December 8, 2015 allowed the marketing of the first cooling cap in the US, Dignitana DigniCap Cooling System, to reduce hair loss, in female breast cancer patients undergoing chemotherapy. Alopecia (hair loss) is a common side effect of several chemotherapeutic agents and can be emotionally traumatic. Even though temporary, minimizing or alleviating hair loss, can have a significant impact on patients psychological well being and willingness to pursue necessary treatment. Presently available non-FDA approved cooling devices include Penguin Cold Caps and Paxman Scalp Cooling System, although the later is not available in the US. One of the major concerns with cold caps use has been the risk for scalp metastasis due to decreased chemotherapy access to the scalp tissue from vasoconstriction associated with cooling devices. It is clear now that that the risk of metastases to the scalp is extremely rare and low (1.2%) and even lower as an initial event for advanced disease.
The Dignitana DigniCap computer-controlled cooling system pumps liquid coolant through a head-worn silicone cooling cap during chemotherapy treatment. This cooling cap is covered by an outer insulating cap which holds the cooling cap in place. The circulating coolant inside the cap gradually gets colder. The cold and near freezing temperature constricts the blood vessels in the scalp, which, in turn reduces chemotherapy access in the hair follicles, as well as metabolic activity of the hair follicle cells, thus slowing cell division. This combined action impairs the effect of chemotherapy on hair follicles and reduces chemotherapy induced hair loss.
The FDA approval was based on a multicenter prospective open-label, nonrandomized study in which the efficacy of the cooling system was studied in 122 women with Stage I and Stage II breast cancer who were receiving chemotherapy regimens associated with hair loss. The primary endpoint was patient self-assessment of hair loss using standardized photographs at three to six weeks after the last chemotherapy cycle. A score of 0-2 (50% or less hair loss) was defined as treatment success. Patients who chose not to undergo scalp cooling were enrolled in a control group. It was noted that more than 66 percent of patients treated with the DigniCap reported losing less than half their hair whereas 94% had more than 75% hair loss in the control group. The most common side effects with the scalp cooling system included cold-induced headaches and neck and shoulder discomfort, chills and pain associated with wearing the cooling cap for prolonged period of time.
The authors concluded that the DigniCap System is highly effective in reducing chemotherapy-induced alopecia and the FDA approval of this scientifically proven option will provide a major relief for cancer patients receiving chemotherapy. Clinical performance of the DigniCap system, a scalp hypothermia system, in preventing chemotherapy-induced alopecia. Rugo HS, Klein P, Melin SA, et al. J Clin Oncol 33, 2015 (suppl; abstr 9518)
Preserving Fertility with ZOLADEX® in Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. Premature Ovarian Failure (POF) is a common unintended consequence of chemotherapy in premenopausal women. Besides of loss of fertility, which can influence treatment decisions in young women, ovarian failure can lead to menopausal symptoms, sexual dysfunction and loss of bone density.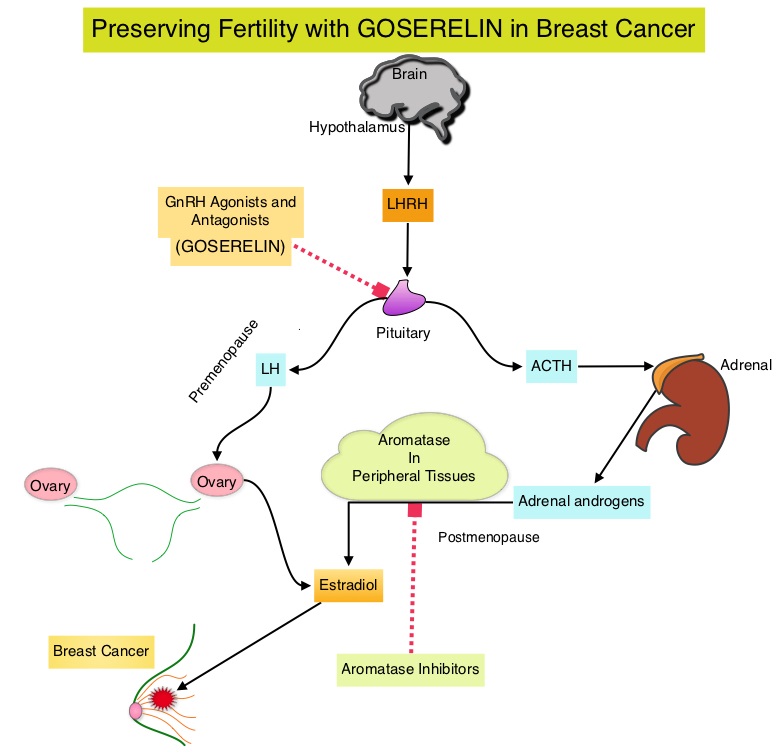
POEMS (Prevention of Early Menopause Study) is a randomized phase III trial designed to evaluate whether the addition of LHRH (Luteinizing Hormone-Releasing Hormone) analog Goserelin (ZOLADEX®), which suppresses the production of estrogens, to Cyclophosphamide based chemotherapy, would reduce POF in breast cancer patients, when compared to chemotherapy alone. Premenopausal patients less than 50 years of age, with hormone receptor negative (ER/PR negative ), Stage I-IIIA breast cancer, scheduled to receive chemotherapy, were randomly assigned to receive standard Cyclophosphamide based chemotherapy with or without monthly ZOLADEX® . Patients in the ZOLADEX® group received 3.6 mg SQ starting 1 week prior to the first dose of chemotherapy.
The primary endpoint was ovarian failure at two years (defined as amenorrhea for the prior 6 months AND post-menopausal FSH level). Other endpoints included pregnancy and survival rates. The median age of the patients was 38 years and median follow up was 4.1 years. Of the 218 evaluable patients, 135 premenopausal women were evaluable for the primary end point. POF rates were 22% in the chemotherapy alone group and 8% in the ZOLADEX® group (P=0.04). When the definition of POF was more liberal to include EITHER amenorrhea or elevated FSH but not both, POF rates were 45% in the chemotherapy alone group and 20% in the ZOLADEX® group (P=0.006). Among the 218 evaluable patients, more women in the ZOLADEX® group achieved at least one pregnancy (21%) compared to 11% in the chemotherapy alone group (P=0.03). Secondary outcomes also favored the ZOLADEX® group with a Disease free Survival (DFS) rate of 78% in the chemotherapy alone group compared with 89% in the ZOLADEX® group (P=0.04) and Overall Survival (OS) rate of 82% in the chemotherapy alone group compared with 92% in the ZOLADEX® group (P=0.05).
The authors concluded that the addition of ZOLADEX® to chemotherapy improved fertility prospects with a lower incidence of Premature Ovarian Failure and more pregnancies. Further, the improved Disease Free Survival and Overall Survival are important additional perks and prevention of Premature Ovarian Failure with ZOLADEX® may be a consideration not only in premenopausal breast cancer patients but also in other malignancies such as lymphomas, when treated with similar chemotherapeutic agents. Goserelin for Ovarian Protection during Adjuvant Chemotherapy for Breast Cancer. Moore HC, Unger JM, Phillips K, et al. N Engl J Med 2015; 372:923-932
Breast Cancer Screening Imaging Modalities - A Primer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Screening mammography complemented by breast self exam and clinical breast exam has resulted in early detection of breast cancer and successful outcomes. Even though mammography is a sensitive screening test, a small percentage of breast cancers may not show up on mammograms but may be palpable on examination by the patient or the clinician. Further, mammograms are less likely to find breast tumors in younger women with dense breast tissue. The following is a brief overview of the imaging techniques for breast cancer screening
Mammography
Mammography is performed by compressing the breast firmly between two plates and utilizes ionizing radiation to image breast tissue. During routine screening, breast tissue from the nipple to the pectoral muscle in the mediolateral oblique and craniocaudal views, is included. The radiation exposure is 4 to 24 mSv per standard two view screening examination. Two view screening is associated with a lower recall rate and lower interval cancer rates than are single-view exams. Breast Imaging Reporting and Data System (BI-RADS) categories are used for reporting mammographic results as follows:
0: Incomplete—needs additional image evaluation and/or prior mammograms for comparison.
1: Negative.
2: Benign.
3: Probably benign.
4: Suspicious.
5: Highly suggestive of malignancy.
6: Known biopsy—proven malignancy.
A digital mammogram is more expensive than screen-film mammography (SFM) and the data can be stored and shared. Compared with film mammography, sensitivity is higher for digital mammography, particularly in women younger than 50 years, but the specificity is either the same or lower than film mammography.
Computer-Aided Detection (CAD) systems increase detection of ductal carcinoma in situ (DCIS) by highlighting suspicious regions in the breast such as clustered microcalcifications and masses in mammograms. There is however no improvement in invasive cancer detection rate and there is an increase in recall rate.
Tomosynthesis
Tomosynthesis, or 3-Dimensional (3-D) mammography involves multiple short-exposure x-rays, from different angles and a three dimensional image is created for better visualization. A combination of 2-D and 3-D mammography has been reported to be more accurate than 2-D mammography alone, with the caveat that the radiation exposure to the patient is essentially doubled. Tomosynthesis in the diagnostic setting is at least as effective as spot compression views, for workup of non-calcified abnormalities, including asymmetries and distortions and may decrease the need for ultrasound testing.
Ultrasonography
Primarily utilized for the diagnostic evaluation of palpable or mammographically detected masses and distinguish solid tumors from cysts. It is a helpful adjunct modality in women with dense breast tissue. Images are created using high frequency sound waves with no radiation exposure. Evidence is lacking to support the use of ultrasound instead of mammography, at any age, in population based breast cancer screening.
Thermography
Thermography uses infrared imaging techniques and identifies temperature changes in the skin as an indicator of an underlying tumor. These changes are displayed in color patterns. The impact of thermography on breast cancer detection or mortality, has not been evaluated in randomized clinical trials and there appears to be no additional benefit for the use of thermography as an adjunct modality, for breast cancer screening.
Magnetic Resonance Imaging
Magnetic Resonance Imaging (MRI) is more sensitive than mammography although the specificity of a breast MRI is lower, resulting in a higher rate of false-positive findings and potentially unnecessary biopsies. Microcalcifications in the breast can be missed by a breast MRI. The American Cancer Society (ACS) recommends an annual MRI as an adjunct to screening mammogram and clinical breast exam in certain groups with increased risk of breast cancer. They include individuals with deleterious genetic mutations such as BRCA1/2 mutation carriers, a strong family history of breast cancer, or several genetic syndromes such as Li-Fraumeni or Cowden disease. MRI may also be used to evaluate the integrity of silicone breast implants, assess palpable masses following surgery or radiation therapy, detect mammographically and sonographically occult breast cancer in patients with axillary nodal metastasis and preoperative planning for some patients with known breast cancer. Breast MRI is performed preferably between days 7-15 of menstrual cycle for premenopausal women, using a dedicated breast coil, with the ability to perform a biopsy under MRI guidance by experienced radiologists, during the same visit.
Molecular Breast Imaging
Molecular Breast Imaging (MBI) involves the injection of technetium-99m (Tc-99m) sestamibi, a radioactive substance, which then allows tumor visualization with a gamma camera. MBI along with mammography significantly increased the cancer detection rate in women with mammographically dense breasts, compared to mammography alone, in a recent study. This new technology is not yet widely available.
References: 1) American Cancer Society recommendations for early breast cancer detection in women without breast symptoms. http://www.cancer.org/cancer/breastcancer 2) National Cancer Institute: PDQ® Breast Cancer Screening. Bethesda, MD
Genetics of Breast Cancer – A Primer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Approximately 5% to 10% of breast cancers are hereditary. The discovery of several new genetic mutations which can increase the risk of breast cancer, helped better understand inherited breast cancer susceptibility.
Genetic mutations in Breast cancer can be grouped into three categories:
1) High penetrance genes such as BRCA1 and BRCA2 gene mutations which account for 15–25% of the inherited breast cancers, TP53 gene mutations which cause the Li–Fraumeni syndrome, PTEN gene mutations which cause Cowden syndrome, STK11 gene mutations which cause Peutz–Jeghers syndrome and CDH1 gene mutations which cause hereditary diffuse gastric cancer. Under normal circumstances, the proteins produced from these genes act as tumor suppressors and are involved in repairing damaged DNA, which in turn helps to maintain the stability of a cell's genetic information. These gene mutations are rare and can result in a 10 fold increase in breast cancer risk.
2) Intermediate penetrance gene mutations increase the risk of breast cancer two to four fold. They include ATM, CHEK2, BRIP1, BARD1, and PALB2 gene mutations. Some of these genes provide instructions for making proteins that interact with the proteins produced from the BRCA1 or BRCA2 genes.
3) Low penetrance gene mutations such as FGFR2 gene mutations
MULTIGENE PANEL TESTING
Multigene panel testing can detect several more than the abnormal genes mentioned above and may also incidentally pick up an unexpected abnormal gene which may be associated with a low risk for cancer. This information can be emotionally stressful for patients, as there is little or no guidance regarding management. Further, multigene panel testing may find “Variants of Uncertain Significance” (VUS) which are genetic mutations that may or may not be linked to a disease. It is therefore imperative to counsel patients and families before and after genetic testing and adequate resources should be allocated to properly interpret the test results to these individuals.
WHICH GENES TO TEST
Actionable information from multigene panel testing can significantly benefit patients and family members. The testing panel should include BRCA1 and BRCA2 gene mutations as close to 10% of breast cancer patients with a strong family history who undergo multigene panel testing will have a deleterious mutation. Amongst them, about 6% will have BRCA1 or BRCA2 mutation and 4% will have gene mutations other than BRCA1and BRCA2. The panel testing should also include PALB2 gene mutations which carries a lifetime breast cancer risk of 33% to 58%, as well as CHEK2, ATM, and TP53 gene mutations for estrogen receptor positive breast cancer patients.
WHO SHOULD BE TESTED
1) A gene mutation linked to breast cancer is more likely if
2) Family is of Ashkenazi (Eastern European) Jewish descent.
3) Two or more first-degree (parent, sibling, or child) or second-degree (grandmother, granddaughter, aunt, niece, half-sibling) relatives were diagnosed with breast or ovarian cancer.
4) Breast cancer diagnosed before the age of 50 (premenopausal) in a close relative.
5) There is a family history of both breast and ovarian cancer.
6) Bilateral breast cancer was diagnosed in a close relative.
7) Male relatives were diagnosed with breast cancer.
8) Breast cancer is diagnosed in family and either male relatives on the same side of the family have had prostate cancer at a young age, or male or female relatives on the same side of the family have had gastrointestinal cancers, such as cancer of the pancreas, gall bladder or stomach.
Antoniou AC, Casadei S, Heikkinen T, et al. N Engl J Med 2014; 371:497-506.
National Comprehensive Cancer Network: Genetic/Familial High-Risk Assessment, Version 2.2015
Late Breaking Abstract – ASCO 2015: IBRANCE® More Than Doubles Progression Free Survival in Hormone Receptor Positive Advanced Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Tamoxifen is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. Anastrozole and Letrozole are nonsteroidal Aromatase Inhibitors that binds reversibly to the aromatase enzyme and inhibit the conversion of androgens to estrogens in the extra-gonadal tissues.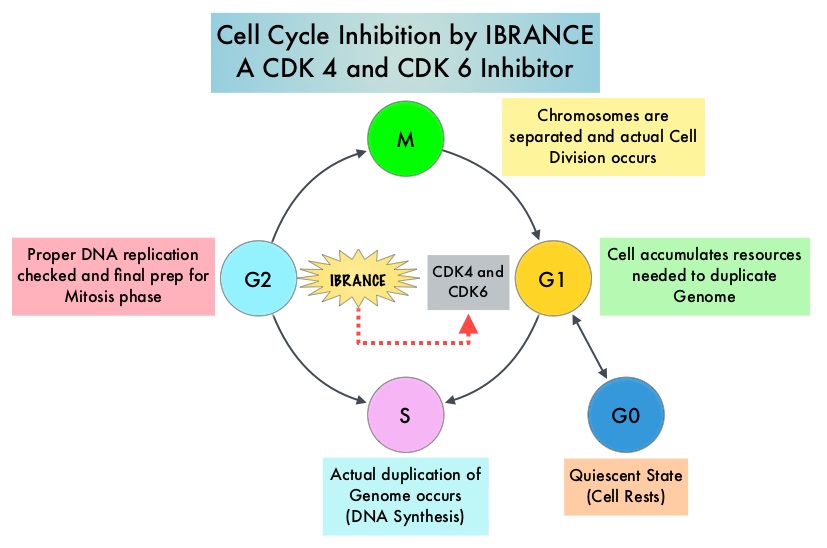 Approximately 80% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB) and initiate transition from the G1 phase to the S phase of the cell cycle. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. IBRANCE® (Palbociclib) is a reversible, oral, selective, small molecule inhibitor of Cyclin Dependent Kinases, CDK4 and CDK6, and prevent RB1 phosphorylation. IBRANCE® is the first CDK inhibitor approved by the FDA. It exhibits synergy when combined with endocrine therapies. In an open-label, randomized, phase II study, which included treatment naïve postmenopausal women with ER-positive, HER2-negative, advanced breast cancer, IBRANCE® given along with Aromatase Inhibitor FEMARA® (Letrozole) significantly prolonged Progression Free Survival, Overall Response rate and median duration of response, compared to FEMARA® alone. Based on this data, the U. S. Food and Drug Administration on February 3, 2015 granted accelerated approval to IBRANCE® (Palbociclib), for use in combination with FEMARA® (Letrozole) in this patient population. FASLODEX® (Fulvestrant) is a selective estrogen receptor down-regulator presently indicated for the treatment of hormone receptor positive metastatic breast cancer patients, with disease progression following antiestrogen therapy.
Approximately 80% of breast tumors express Estrogen Receptors and/or Progesterone Receptors and these patients are often treated with anti-estrogen therapy as first line treatment. Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. Cyclin Dependent Kinases 4 and 6 (CDK4 and CDK6) phosphorylate RetinoBlastoma protein (RB) and initiate transition from the G1 phase to the S phase of the cell cycle. CDK4 and CDK6 are activated in hormone receptor positive breast cancer, promoting breast cancer cell proliferation. Further, there is evidence to suggest that endocrine resistant breast cancer cell lines depend on CDK4 for cell proliferation. IBRANCE® (Palbociclib) is a reversible, oral, selective, small molecule inhibitor of Cyclin Dependent Kinases, CDK4 and CDK6, and prevent RB1 phosphorylation. IBRANCE® is the first CDK inhibitor approved by the FDA. It exhibits synergy when combined with endocrine therapies. In an open-label, randomized, phase II study, which included treatment naïve postmenopausal women with ER-positive, HER2-negative, advanced breast cancer, IBRANCE® given along with Aromatase Inhibitor FEMARA® (Letrozole) significantly prolonged Progression Free Survival, Overall Response rate and median duration of response, compared to FEMARA® alone. Based on this data, the U. S. Food and Drug Administration on February 3, 2015 granted accelerated approval to IBRANCE® (Palbociclib), for use in combination with FEMARA® (Letrozole) in this patient population. FASLODEX® (Fulvestrant) is a selective estrogen receptor down-regulator presently indicated for the treatment of hormone receptor positive metastatic breast cancer patients, with disease progression following antiestrogen therapy.
The PALOMA3 is double-blind, phase 3 study in which the efficacy and safety of the combination of IBRANCE® and FASLODEX® was evaluated, in premenopausal or postmenopausal women, with hormone receptor positive, HER-2 negative, advanced breast cancer, who had disease progression during prior endocrine therapy. Five hundred and twenty one (N=521) patients were randomly assigned in a 2:1 ratio to receive either FASLODEX® 500 mg IM on days 1 and 15 during cycle 1, of a 28 day cycle, and then on day 1 of each cycle thereafter, along with IBRANCE® 125 mg PO daily for 3 weeks, followed by 1 week off (N=347) or FASLODEX® and placebo (N=174). ZOLADEX® (Goserelin) was administered to premenopausal or perimenopausal patients for the duration of study treatment, starting at least 4 weeks before randomization and continuing every 28 days. The median age was 57 years. One previous line of chemotherapy for metastatic disease was allowed and 79% were post-menopausal, 60% had visceral disease and 33% of the patients had prior chemotherapy for advanced disease. The primary endpoint was Progression Free Survival (PFS) and secondary endpoints included Overall Survival (OS), Response Rates, safety and tolerability. At the time of the preplanned interim analysis, the median Progression Free Survival was 9.2 months in the FASLODEX® / IBRANCE® group and 3.8 months in the FASLODEX® /placebo group (HR=0.422; P<0.000001). This PFS benefit was observed across all prespecified patient subgroups, regardless of menopausal status. The most common adverse events in the IBRANCE® group were neutropenia (78.8% vs 3.5%) and fatigue (38.0% vs 26.7%). The incidence of febrile neutropenia was very rare (0.6%) and similar in both treatment groups. Treatment discontinuation rate due to adverse events was 2% in the IBRANCE® group and 1.7% in the placebo group. The authors concluded that IBRANCE® in combination with FASLODEX® more than doubled the Progression Free Survival in advanced breast cancer patients, with hormone receptor positive and HER-2 negative disease, who had progressed on prior endocrine therapy. This study has reinforced the importance of CDK4 and CDK6, as key targets for hormone receptor positive breast cancer. Palbociclib in Hormone Receptor Positive Advanced Breast Cancer. Turner NC, Ro J, Andre F, et al. June 1, 2015DOI: 10.1056/NEJMoa1505270
Late Breaking Abstract – ASCO 2015: ARIMIDEX® may be a Better Treatment Option Compared to Tamoxifen in Postmenopausal Women with DCIS
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease.Estrogen Receptor (ER) positive breast cancer cells are driven by estrogens. Tamoxifen is a nonsteroidal Selective Estrogen Receptor Modulator (SERM) and works mainly by binding to the Estrogen Receptor and thus blocks the proliferative actions of estrogen on the mammary tissue. Anastrozole is a non-steroidal Aromatase Inhibitor that binds reversibly to the aromatase enzyme and inhibits the conversion of androgens to estrogens in the extra-gonadal tissues. Aromatase Inhibitors such as ARIMIDEX® have been proven to be superior to Tamoxifen, as adjuvant therapy, in postmenopausal patients with hormone receptor positive breast cancer. It was however not known whether ARIMIDEX® was superior to Tamoxifen in preventing the recurrence of breast cancer.
NSABP B-35 is a randomized phase III trial which compared ARIMIDEX® to Tamoxifen, in preventing the recurrence of breast cancer, in postmenopausal women with Ductal Carcinoma In Situ (DCIS), who underwent lumpectomy and radiation therapy. Of the 3,104 patients who were enrolled and randomized, 1552 patients received ARIMIDEX® 1 mg PO daily and 1552 patients received Tamoxifen 20 mg PO daily. Treatment was continued for 5 years. Enrolled patients had tumors with no invasive component, Estrogen or Progesterone Receptors were positive and margins of resection were clear. The primary endpoint was Breast Cancer-Free Interval (BCFI), defined as the time from randomization to any breast cancer event including local, regional, or distant recurrence or contralateral invasive cancer or DCIS. With a median follow up of 8.6 years, the 10 year Breast Cancer–Free Interval rates were 93.5% with ARIMIDEX® versus 89.2% with Tamoxifen, and this was statistically significant (HR=0.73, P=0.03). This benefit was more so in women less than 60 years of age, with a Breast Cancer–Free Interval rates of 94.9% with ARIMIDEX® and 88.2% with Tamoxifen (HR=0.52, P=0.003). However, in women over 60 years of age, there were no significant differences in outcomes noted between the ARIMIDEX® and Tamoxifen groups. The incidence of invasive contralateral breast cancer was reduced by 45% (HR=0.55, P=0.03) in the ARIMIDEX® group, compared to Tamoxifen group. Adverse events were less common with ARIMIDEX® compared to Tamoxifen. The authors concluded that for postmenopausal women with DCIS treated with lumpectomy and radiation, the 10 year Breast Cancer–Free Interval rates are significantly higher with 5 years of ARIMIDEX® than Tamoxifen and ARIMIDEX® may also be a consideration for those women, who are concerned about the risk of thromboembolic events and uterine cancer with Tamoxifen treatment. Primary results, NRG Oncology/NSABP B-35: A clinical trial of anastrozole (A) versus tamoxifen (tam) in postmenopausal patients with DCIS undergoing lumpectomy plus radiotherapy. Margolese RG, Cecchini RS, Julian TB, et al. J Clin Oncol 33, 2015 (suppl; abstr LBA500)
Role of Radiotherapy in Elderly Patients with Low Risk Breast Cancer Following Breast Conserving Surgery
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. The National Surgical Adjuvant Breast and Bowel Project (NASBP) protocols B-04 and B-06 have clearly established, after more than a 2 decades of evaluation and follow up that, in Stage I and II breast cancer, there is no significant difference in either distant Disease Free or Overall Survival between the breast conserving surgery and breast removal surgery (Mastectomy). This data established breast-sparing surgery as the preferred local-regional procedure. Radiotherapy after breast-conserving surgery significantly decreases the risk of local recurrence and improves breast cancer–specific survival in certain subgroups of patients. Under ideal circumstances, approximately 70% of the patients can be treated with breast conserving surgery and the risk factors for local recurrences include younger age, high tumor grade, negative hormone receptor status, lymphovascular invasion, extensive intraductal component and positive surgical margins. According to the American Cancer Society, 42% of all invasive breast cancers in the US occur in women 65 years of age or older. These patients may have associated comorbidities and may therefore be appropriate candidates for breast-sparing surgery (rather than mastectomy), as post-op recovery time is shorter. Elderly patients often present with breast tumors that are hormone receptor positive and indolent. Whether women 70 years of age or older with early stage breast cancer need radiotherapy after breast conserving surgery, was addressed by the CALGB cooperative group ( CALGB 9343) and it was noted that the addition of radiation did not improve Overall Survival or distant Disease Free Survival, although there was some improvement in locoregional recurrence rate.
The authors in PRIME II study evaluated local control outcomes in elderly women with early stage breast cancer, at low risk of local recurrence at 5 years, by avoiding whole-breast radiotherapy, following breast conserving surgery. In this Phase III randomized trial, 1326 patients 65 years or older, with low risk early breast cancer, who had undergone breast conserving surgery and were receiving adjuvant hormonal treatment, were randomly assigned to receive whole-breast irradiation (N=658) or no further therapy (N=668). Low risk was defined as tumors T1–T2 up to 3 cm in greatest dimension, clear surgical margins, negative axillary nodes and positive hormone receptors. Either lymphovascular invasion or grade 3 tumor histology was permitted, but not both. The primary endpoint was ipsilateral breast tumor recurrence. After median follow up of 5 years, ipsilateral breast tumor recurrence was 1.3% in women assigned to whole-breast radiotherapy and 4.1% in those assigned no radiotherapy (P=0•0002). There was however no differences in regional recurrence, contralateral breast cancers, distant metastases or new breast cancers, noted between the two groups. The 5-year Overall Survival was similar in both groups 93•9% (P=0•34). The authors concluded that the 5-year ipsilateral breast tumor recurrence is probably low enough that radiotherapy could be avoided in a subset of elderly patients with low risk breast cancer following breast conserving surgery. Kunkler IH, Williams LJ, Jack WJ, et al: Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (PRIME II): A randomised controlled trial. Lancet -Oncol. 2015;16:266-273
ABRAXANE® Superior to TAXOL® in High Risk Breast Cancer Including Triple Negative Tumors
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Taxanes, which include TAXOL® (Paclitaxel) and TAXOTERE® (Docetaxel) have an important role in the treatment of breast cancer and have been shown in numerous clinical studies to improve survival in patients with early-stage breast cancer and select group of patients with metastatic breast cancer.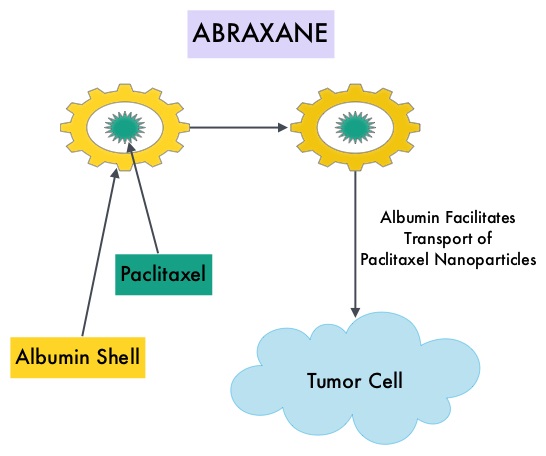 ABRAXANE® (Nab-paclitaxel) is a solvent free, albumin-encapsulated nanoparticle formulation of the taxane, Paclitaxel. By virtue of its formulation, unlike TAXOL®, hypersensitivity reactions are uncommon with ABRAXANE® and can therefore be rapidly infused and premedications are not needed. Further, higher tumor drug concentrations are achieved with ABRAXANE® compared to conventional Paclitaxel (TAXOL®). Previously published studies have shown that ABRAXANE® is superior to TAXOL® in metastatic breast cancer. GeparSepto trial is a randomized phase III study in which weekly ABRAXANE® was compared head to head with weekly TAXOL® in a neoadjuvant setting. This study enrolled 1204 treatment naïve, high risk patients, with clinical T2-T4d invasive breast carcinoma. Eligible patients included those with unilateral, bilateral, operable or inoperable breast cancer. The median age was 49 years, median tumor size was 3 cm, 23% of the patients had triple-negative disease and 33% had HER-2 positive tumors. Patients were randomized 1:1 to receive either ABRAXANE® 125 mg/m2 IV or TAXOL® 80 mg/m2 IV, weekly for 12 weeks followed by 4 cycles of Epirubicin 90 mg/m2 IV and Cyclophosphamide 600 mg/m2 IV. Patients with HER-2 positive tumors also received HERCEPTIN® (Trastuzumab) and PERJETA® (Pertuzumab). The primary endpoint was pathologic Complete Response (pCR), defined as absence of microscopic residual invasive or noninvasive viable tumor cells in all resected specimens of the breast and axilla. This endpoint was chosen because pathologic Complete Response serves as a surrogate marker for long-term efficacy and outcomes. In this trial, the researchers noted a pathologic Complete Response rate of 38% with ABRAXANE® compared to 29% with TAXOL® (P<0.01). On subgroup analysis, this benefit was even more evident in patients with triple negative breast cancer (N=275), with a pCR rate of 48.2% in the ABRAXANE® group compared with 25.7% in the TAXOL® group (P <0.001). The incidence of peripheral neuropathy was higher in the ABRAXANE® group compared to TAXOL® group and this was attributed to higher weekly doses of ABRAXANE® administered. It is felt that a lower dose of weekly ABRAXANE® (100 mg/m2) would result in a decrease in the incidence of peripheral neuropathy, without compromising efficacy. The authors concluded that ABRAXANE® is superior to TAXOL® in early stage, high risk patients with breast cancer and this benefit is even more evident in those patients with triple negative disease, which comprises about 15% of all breast cancers. Untch M, Jackisch C, Schneeweiss A, et al. A randomized phase III trial comparing neoadjuvant chemotherapy with weekly nanoparticle-based paclitaxel with solvent-based paclitaxel followed by anthracyline/cyclophosphamide for patients with early breast cancer (GeparSepto); GBG 69. Paper presented at: 2014 San Antonio Breast Cancer Symposium; December 9-13, 2014; San Antonio, TX. Abstract S2-07.
ABRAXANE® (Nab-paclitaxel) is a solvent free, albumin-encapsulated nanoparticle formulation of the taxane, Paclitaxel. By virtue of its formulation, unlike TAXOL®, hypersensitivity reactions are uncommon with ABRAXANE® and can therefore be rapidly infused and premedications are not needed. Further, higher tumor drug concentrations are achieved with ABRAXANE® compared to conventional Paclitaxel (TAXOL®). Previously published studies have shown that ABRAXANE® is superior to TAXOL® in metastatic breast cancer. GeparSepto trial is a randomized phase III study in which weekly ABRAXANE® was compared head to head with weekly TAXOL® in a neoadjuvant setting. This study enrolled 1204 treatment naïve, high risk patients, with clinical T2-T4d invasive breast carcinoma. Eligible patients included those with unilateral, bilateral, operable or inoperable breast cancer. The median age was 49 years, median tumor size was 3 cm, 23% of the patients had triple-negative disease and 33% had HER-2 positive tumors. Patients were randomized 1:1 to receive either ABRAXANE® 125 mg/m2 IV or TAXOL® 80 mg/m2 IV, weekly for 12 weeks followed by 4 cycles of Epirubicin 90 mg/m2 IV and Cyclophosphamide 600 mg/m2 IV. Patients with HER-2 positive tumors also received HERCEPTIN® (Trastuzumab) and PERJETA® (Pertuzumab). The primary endpoint was pathologic Complete Response (pCR), defined as absence of microscopic residual invasive or noninvasive viable tumor cells in all resected specimens of the breast and axilla. This endpoint was chosen because pathologic Complete Response serves as a surrogate marker for long-term efficacy and outcomes. In this trial, the researchers noted a pathologic Complete Response rate of 38% with ABRAXANE® compared to 29% with TAXOL® (P<0.01). On subgroup analysis, this benefit was even more evident in patients with triple negative breast cancer (N=275), with a pCR rate of 48.2% in the ABRAXANE® group compared with 25.7% in the TAXOL® group (P <0.001). The incidence of peripheral neuropathy was higher in the ABRAXANE® group compared to TAXOL® group and this was attributed to higher weekly doses of ABRAXANE® administered. It is felt that a lower dose of weekly ABRAXANE® (100 mg/m2) would result in a decrease in the incidence of peripheral neuropathy, without compromising efficacy. The authors concluded that ABRAXANE® is superior to TAXOL® in early stage, high risk patients with breast cancer and this benefit is even more evident in those patients with triple negative disease, which comprises about 15% of all breast cancers. Untch M, Jackisch C, Schneeweiss A, et al. A randomized phase III trial comparing neoadjuvant chemotherapy with weekly nanoparticle-based paclitaxel with solvent-based paclitaxel followed by anthracyline/cyclophosphamide for patients with early breast cancer (GeparSepto); GBG 69. Paper presented at: 2014 San Antonio Breast Cancer Symposium; December 9-13, 2014; San Antonio, TX. Abstract S2-07.
Goserelin for Ovarian Protection during Adjuvant Chemotherapy for Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 231,840 new cases of invasive breast cancer will be diagnosed in 2015 and over 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues.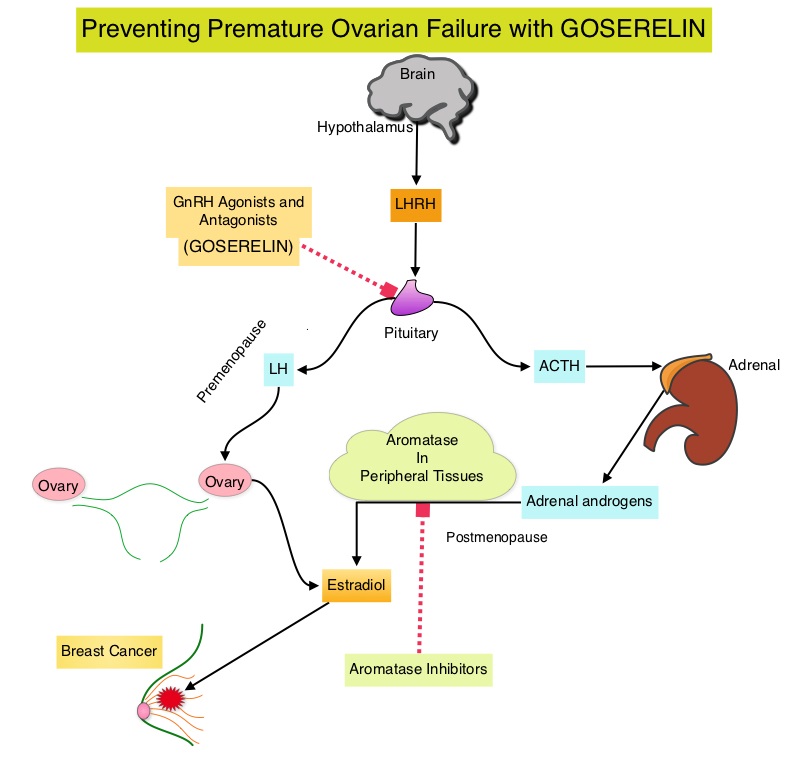 Premature Ovarian Failure (POF) is a common unintended consequence of chemotherapy in premenopausal women. Besides of loss of fertility, which can influence treatment decisions in young women, ovarian failure can lead to menopausal symptoms, sexual dysfunction and loss of bone density. POEMS (Prevention of Early Menopause Study) is a randomized phase III trial designed to evaluate whether the addition of LHRH (Luteinizing Hormone-Releasing Hormone) analog Goserelin (ZOLADEX®), which suppresses the production of estrogens, to Cyclophosphamide based chemotherapy, would reduce POF in breast cancer patients, when compared to chemotherapy alone. Premenopausal patients less than 50 years of age, with hormone receptor negative (ER/PR negative ), Stage I-IIIA breast cancer, scheduled to receive chemotherapy, were randomly assigned to receive standard Cyclophosphamide based chemotherapy with or without monthly ZOLADEX® . Patients in the ZOLADEX® group received 3.6 mg SQ starting 1 week prior to the first dose of chemotherapy. The primary endpoint was ovarian failure at two years (defined as amenorrhea for the prior 6 months AND post-menopausal FSH level). Other endpoints included pregnancy and survival rates. The median age of the patients was 38 years and median follow up was 4.1 years. Of the 218 evaluable patients, 135 premenopausal women were evaluable for the primary end point. POF rates were 22% in the chemotherapy alone group and 8% in the ZOLADEX® group (P=0.04). When the definition of POF was more liberal to include EITHER amenorrhea or elevated FSH but not both, POF rates were 45% in the chemotherapy alone group and 20% in the ZOLADEX® group (P=0.006). Among the 218 evaluable patients, more women in the ZOLADEX® group achieved at least one pregnancy (21%) compared to 11% in the chemotherapy alone group (P=0.03). Secondary outcomes also favored the ZOLADEX® group with a Disease free Survival (DFS) rate of 78% in the chemotherapy alone group compared with 89% in the ZOLADEX® group (P=0.04) and Overall Survival (OS) rate of 82% in the chemotherapy alone group compared with 92% in the ZOLADEX® group (P=0.05). The authors concluded that the addition of ZOLADEX® to chemotherapy improved fertility prospects with a lower incidence of Premature Ovarian Failure and more pregnancies. Further, the improved Disease Free Survival and Overall Survival is an important additional perk and prevention of Premature Ovarian Failure with ZOLADEX® may be a consideration not only in premenopausal patients with hormone receptor positive breast cancer but also in other malignancies such as lymphomas, when treated with similar chemotherapeutic agents. Goserelin for Ovarian Protection during Breast-Cancer Adjuvant Chemotherapy. Moore HC, Unger JM, Phillips K, et al. N Engl J Med 2015; 372:923-932
Premature Ovarian Failure (POF) is a common unintended consequence of chemotherapy in premenopausal women. Besides of loss of fertility, which can influence treatment decisions in young women, ovarian failure can lead to menopausal symptoms, sexual dysfunction and loss of bone density. POEMS (Prevention of Early Menopause Study) is a randomized phase III trial designed to evaluate whether the addition of LHRH (Luteinizing Hormone-Releasing Hormone) analog Goserelin (ZOLADEX®), which suppresses the production of estrogens, to Cyclophosphamide based chemotherapy, would reduce POF in breast cancer patients, when compared to chemotherapy alone. Premenopausal patients less than 50 years of age, with hormone receptor negative (ER/PR negative ), Stage I-IIIA breast cancer, scheduled to receive chemotherapy, were randomly assigned to receive standard Cyclophosphamide based chemotherapy with or without monthly ZOLADEX® . Patients in the ZOLADEX® group received 3.6 mg SQ starting 1 week prior to the first dose of chemotherapy. The primary endpoint was ovarian failure at two years (defined as amenorrhea for the prior 6 months AND post-menopausal FSH level). Other endpoints included pregnancy and survival rates. The median age of the patients was 38 years and median follow up was 4.1 years. Of the 218 evaluable patients, 135 premenopausal women were evaluable for the primary end point. POF rates were 22% in the chemotherapy alone group and 8% in the ZOLADEX® group (P=0.04). When the definition of POF was more liberal to include EITHER amenorrhea or elevated FSH but not both, POF rates were 45% in the chemotherapy alone group and 20% in the ZOLADEX® group (P=0.006). Among the 218 evaluable patients, more women in the ZOLADEX® group achieved at least one pregnancy (21%) compared to 11% in the chemotherapy alone group (P=0.03). Secondary outcomes also favored the ZOLADEX® group with a Disease free Survival (DFS) rate of 78% in the chemotherapy alone group compared with 89% in the ZOLADEX® group (P=0.04) and Overall Survival (OS) rate of 82% in the chemotherapy alone group compared with 92% in the ZOLADEX® group (P=0.05). The authors concluded that the addition of ZOLADEX® to chemotherapy improved fertility prospects with a lower incidence of Premature Ovarian Failure and more pregnancies. Further, the improved Disease Free Survival and Overall Survival is an important additional perk and prevention of Premature Ovarian Failure with ZOLADEX® may be a consideration not only in premenopausal patients with hormone receptor positive breast cancer but also in other malignancies such as lymphomas, when treated with similar chemotherapeutic agents. Goserelin for Ovarian Protection during Breast-Cancer Adjuvant Chemotherapy. Moore HC, Unger JM, Phillips K, et al. N Engl J Med 2015; 372:923-932
Pertuzumab, Trastuzumab, and Docetaxel in HER2-Positive Metastatic Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 232,000 new cases of invasive breast cancer will be diagnosed in 2015 and about 40,000 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. Trastuzumab binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Despite this benefit, majority of these patients develop progressive disease within 18 months. The tumors in these patients continue to express HER2 although the lower sensitivity to HER2 targeted agents has been attributed to HER2 independent escape mechanisms. Treatment strategies for this patient population have included switching chemotherapy in subsequent lines of treatment and continuing HERCEPTIN®, combining another HER2 targeted agent, Lapatinib (TYKERB®) with Capecitabine (XELODA®) and dual HER2 inhibition with a combination of HERCEPTIN® and TYKERB®.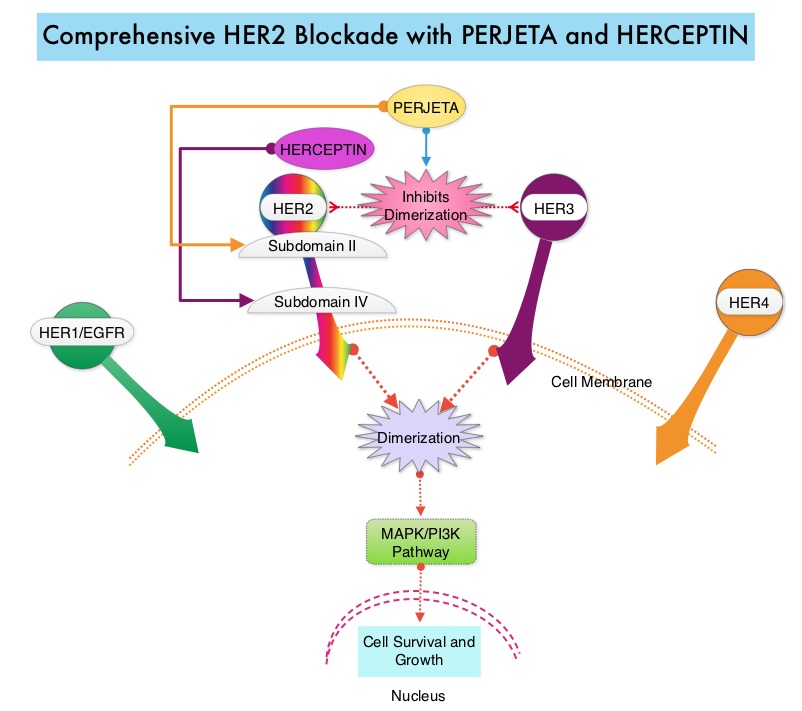 PERJETA® (Pertuzumab) is a recombinant humanized monoclonal antibody that binds to the HER2 at a different epitope of the HER2 extracellular domain (subdomain II) compared to HERCEPTIN® and prevents the dimerization of HER2 with HER3 receptor. PERJETA® stimulates antibody-dependent, cell-mediated cytotoxicity similar to HERCEPTIN®. By combining HERCEPTIN® and PERJETA®, a more comprehensive blockade of HER2 signaling can be accomplished, as these two agents bind to different HER2 epitopes and may complement each other and improve efficacy, as was demonstrated in early phase trials. The CLEOPATRA trial is a phase III study in which 808 treatment naive HER positive metastatic breast cancer patients, were randomly assigned to receive either HERCEPTIN® plus Docetaxel or this two drug combination given along with PERJETA®. PERJETA® was given as an 840 mg loading dose followed by a 420 mg maintenance dose, HERCEPTIN® was given as an 8 mg/kg loading dose followed by a 6 mg/kg maintenance dose and Docetaxel was given at 75 mg/m2 for at least 6 cycles. Treatment was administered every 3 weeks and continued until disease progression. The primary end point of this study was Progression Free Survival and secondary end points included Overall Survival, Objective Response Rate and safety. A previous analysis performed in May 2012 showed that the addition of PERJETA® to the combination of HERCEPTIN® and Docetaxel significantly prolonged Progression Free Survival compared to HERCEPTIN® plus Docetaxel alone (18.5 months vs 12.4 months) but the median overall survival had not been reached then. In this final Overall Survival analysis, at a median follow up of 50 months, median Overall Survival was 56.5 months with the PERJETA® combination compared to 40.8 months in the non-PERJETA® group (hazard ratio [HR] = 0.68; P<0.001). This meant that adding PERJETA® to HERCEPTIN® and Docetaxel increased the median Overall Survival by 15.7 months. The increase in Progression Free Survival by 6.3 months with the PERJETA® combination, was again maintained (HR = 0.68, P < 0.0001), at the time of the final analysis. The median duration of response was 20.2 months with the PERJETA® combination compared to 12.5 months in the non-PERJETA® group. The incidence of symptomatic left ventricular dysfunction as well as declines in left ventricular ejection fraction, were rare and similar between the two treatment groups. Based on the CLEOPATRA study, women with HER positive metastatic breast cancer should be considered candidates, for treatment with a combination of PERJETA®, HERCEPTIN® and Docetaxel. Swain S, Baselga J, Kim S, et al. N Engl J Med 2015; 372:724-734
PERJETA® (Pertuzumab) is a recombinant humanized monoclonal antibody that binds to the HER2 at a different epitope of the HER2 extracellular domain (subdomain II) compared to HERCEPTIN® and prevents the dimerization of HER2 with HER3 receptor. PERJETA® stimulates antibody-dependent, cell-mediated cytotoxicity similar to HERCEPTIN®. By combining HERCEPTIN® and PERJETA®, a more comprehensive blockade of HER2 signaling can be accomplished, as these two agents bind to different HER2 epitopes and may complement each other and improve efficacy, as was demonstrated in early phase trials. The CLEOPATRA trial is a phase III study in which 808 treatment naive HER positive metastatic breast cancer patients, were randomly assigned to receive either HERCEPTIN® plus Docetaxel or this two drug combination given along with PERJETA®. PERJETA® was given as an 840 mg loading dose followed by a 420 mg maintenance dose, HERCEPTIN® was given as an 8 mg/kg loading dose followed by a 6 mg/kg maintenance dose and Docetaxel was given at 75 mg/m2 for at least 6 cycles. Treatment was administered every 3 weeks and continued until disease progression. The primary end point of this study was Progression Free Survival and secondary end points included Overall Survival, Objective Response Rate and safety. A previous analysis performed in May 2012 showed that the addition of PERJETA® to the combination of HERCEPTIN® and Docetaxel significantly prolonged Progression Free Survival compared to HERCEPTIN® plus Docetaxel alone (18.5 months vs 12.4 months) but the median overall survival had not been reached then. In this final Overall Survival analysis, at a median follow up of 50 months, median Overall Survival was 56.5 months with the PERJETA® combination compared to 40.8 months in the non-PERJETA® group (hazard ratio [HR] = 0.68; P<0.001). This meant that adding PERJETA® to HERCEPTIN® and Docetaxel increased the median Overall Survival by 15.7 months. The increase in Progression Free Survival by 6.3 months with the PERJETA® combination, was again maintained (HR = 0.68, P < 0.0001), at the time of the final analysis. The median duration of response was 20.2 months with the PERJETA® combination compared to 12.5 months in the non-PERJETA® group. The incidence of symptomatic left ventricular dysfunction as well as declines in left ventricular ejection fraction, were rare and similar between the two treatment groups. Based on the CLEOPATRA study, women with HER positive metastatic breast cancer should be considered candidates, for treatment with a combination of PERJETA®, HERCEPTIN® and Docetaxel. Swain S, Baselga J, Kim S, et al. N Engl J Med 2015; 372:724-734
The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study
SUMMARY: The U. S. Food and Drug Administration on February 3, 2015 granted accelerated approval to IBRANCE® (Palbociclib), for use in combination with FEMARA® (Letrozole), for the treatment of postmenopausal women with Estrogen Receptor (ER)-positive, Human Epidermal growth factor Receptor 2 (HER2) negative advanced Breast Cancer, as initial endocrine-based therapy for their metastatic disease. Approximately 60% of the breast tumors express Estrogen Receptors and these patients are often treated with anti-estrogen therapy as first line treatment.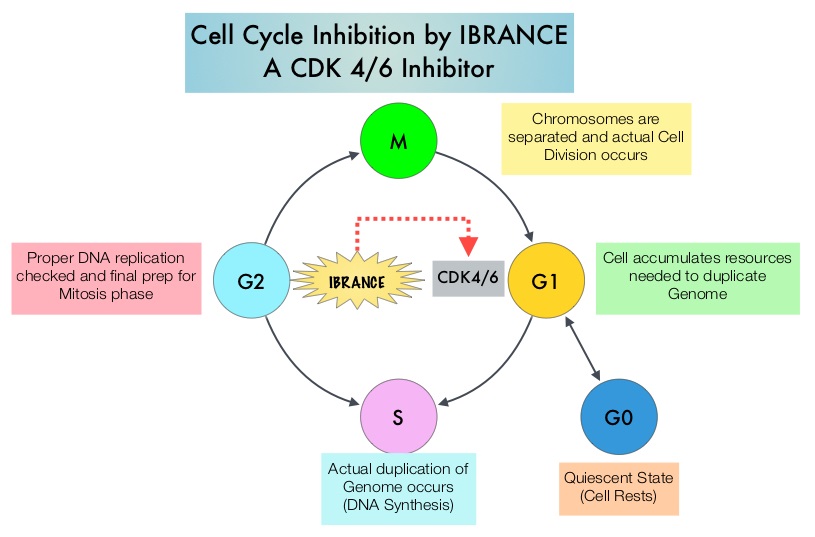 Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. IBRANCE® is a reversible, oral, small molecule inhibitor of Cyclin Dependent Kinases (CDK) 4/6 and is the first CDK inhibitor approved by the FDA. This agent in pre-clinical studies was noted to reduce cellular proliferation of Estrogen Receptor positive Breast Cancer cell lines by blocking progression of cells from G1 into S phase of the cell cycle and was also noted to be synergistic with anti-estrogens. The approval of IBRANCE® was based on an open-label, randomized, multicenter, phase II study which included postmenopausal women with ER-positive, HER2-negative, advanced (locally advanced or metastatic) Breast Cancer who had not received previous systemic treatment for advanced disease. This trial enrolled and randomly assigned 165 patients, to receive either IBRANCE® 125 mg PO daily for 21 consecutive days, followed by 7 days off treatment) plus FEMARA® (2.5 mg PO daily continuously throughout the 28-day cycle (N=84) or FEMARA® alone (N=81). Among the 165 patients, 43% had received chemotherapy and 33% had received anti-hormonal therapy as a neoadjuvant or adjuvant treatment. Fortynine percent (49%) of patients had no prior systemic therapy in the neoadjuvant or adjuvant setting. The majority of patients (98%) had stage IV disease, 48% had visceral disease and 17% had bone only disease. The primary endpoint was investigator-assessed Progression Free Survival. The median PFS was 20.2 months in the IBRANCE® plus FEMARA® group and 10.2 months in the FEMARA® alone group (HR=0.488, P=0•0004). In patients with measurable disease, the Overall Response rate was higher in the IBRANCE® plus FEMARA® group compared to the FEMARA® alone group (55% versus 39%, P=0.04). The median duration of response for those who had a partial or complete response was 20.3 months with a combination of IBRANCE® plus FEMARA® vs 11.1 months with FEMARA® alone. The most common Grade 3–4 adverse events in the IBRANCE® plus FEMARA® group were neutropenia and leucopenia but without any cases of neutropenic fever. Unlike chemotherapy, the duration of neutropenia was brief with rapid recovery of blood counts. The authors concluded that the addition of IBRANCE® to FEMARA® significantly improved Progression Free Survival in women with advanced Estrogen Receptor positive and HER2-negative breast cancer. This combination may therefore be a reasonable treatment option in this patient group, soon after metastatic disease is diagnosed. Biomarkers expression (cyclinD1 amplification and/or loss of p16) had no impact on outcomes suggesting that the biomarker for IBRANCE® may be the Estrogen Receptor itself, rather than CDK4/6 kinases. Finn RS, Crown JP, Lang I, et al. Lancet Oncol 2015; 16:25-35
Cyclin Dependent Kinases (CDK) play a very important role to facilitate orderly and controlled progression of the cell cycle. Genetic alterations in these kinases and their regulatory proteins have been implicated in various malignancies. IBRANCE® is a reversible, oral, small molecule inhibitor of Cyclin Dependent Kinases (CDK) 4/6 and is the first CDK inhibitor approved by the FDA. This agent in pre-clinical studies was noted to reduce cellular proliferation of Estrogen Receptor positive Breast Cancer cell lines by blocking progression of cells from G1 into S phase of the cell cycle and was also noted to be synergistic with anti-estrogens. The approval of IBRANCE® was based on an open-label, randomized, multicenter, phase II study which included postmenopausal women with ER-positive, HER2-negative, advanced (locally advanced or metastatic) Breast Cancer who had not received previous systemic treatment for advanced disease. This trial enrolled and randomly assigned 165 patients, to receive either IBRANCE® 125 mg PO daily for 21 consecutive days, followed by 7 days off treatment) plus FEMARA® (2.5 mg PO daily continuously throughout the 28-day cycle (N=84) or FEMARA® alone (N=81). Among the 165 patients, 43% had received chemotherapy and 33% had received anti-hormonal therapy as a neoadjuvant or adjuvant treatment. Fortynine percent (49%) of patients had no prior systemic therapy in the neoadjuvant or adjuvant setting. The majority of patients (98%) had stage IV disease, 48% had visceral disease and 17% had bone only disease. The primary endpoint was investigator-assessed Progression Free Survival. The median PFS was 20.2 months in the IBRANCE® plus FEMARA® group and 10.2 months in the FEMARA® alone group (HR=0.488, P=0•0004). In patients with measurable disease, the Overall Response rate was higher in the IBRANCE® plus FEMARA® group compared to the FEMARA® alone group (55% versus 39%, P=0.04). The median duration of response for those who had a partial or complete response was 20.3 months with a combination of IBRANCE® plus FEMARA® vs 11.1 months with FEMARA® alone. The most common Grade 3–4 adverse events in the IBRANCE® plus FEMARA® group were neutropenia and leucopenia but without any cases of neutropenic fever. Unlike chemotherapy, the duration of neutropenia was brief with rapid recovery of blood counts. The authors concluded that the addition of IBRANCE® to FEMARA® significantly improved Progression Free Survival in women with advanced Estrogen Receptor positive and HER2-negative breast cancer. This combination may therefore be a reasonable treatment option in this patient group, soon after metastatic disease is diagnosed. Biomarkers expression (cyclinD1 amplification and/or loss of p16) had no impact on outcomes suggesting that the biomarker for IBRANCE® may be the Estrogen Receptor itself, rather than CDK4/6 kinases. Finn RS, Crown JP, Lang I, et al. Lancet Oncol 2015; 16:25-35
Adjuvant Paclitaxel and Trastuzumab for Node-Negative, HER2-Positive Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their life time. Approximately, 233,000 new cases of invasive breast cancer were diagnosed in 2014 and 40,000 women died of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2.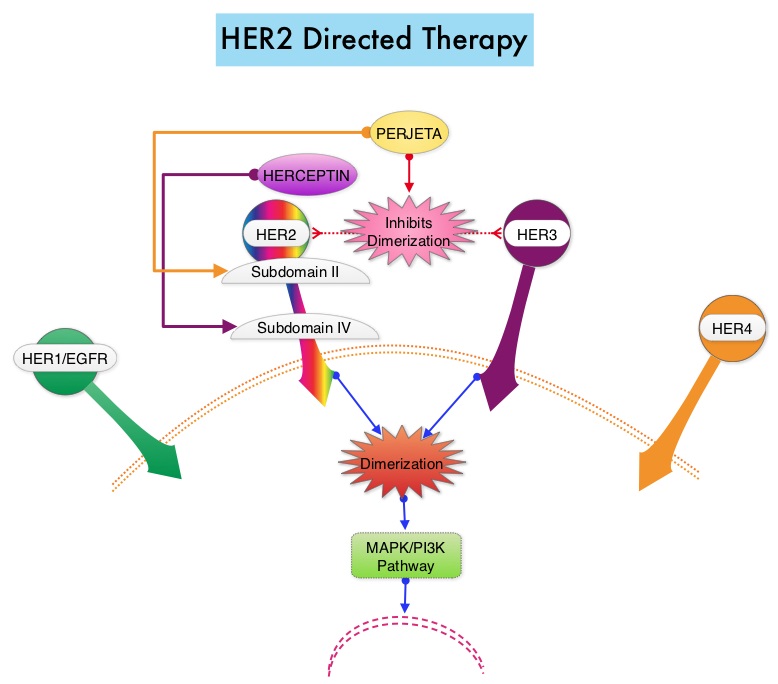 HERCEPTIN® binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Adjuvant chemotherapy in combination with HERCEPTIN® has been shown to reduce the relative risk of relapse by 52% and relative risk of death by 33%. The National Comprehensive Cancer Network (NCCN) has recommended adjuvant chemotherapy with HERCEPTIN® for patients with small, HER positive, node-negative tumors, including those with T1bN0 tumors, even though there are little or no data supporting this recommendation, because these patients are generally not included in adjuvant therapy studies. Further, the chemotherapy regimens often recommended (ACTH, TCH) along with HERCEPTIN® are relatively toxic. The authors in this study chose a less toxic chemotherapy regimen than the regimens often recommended for those patients with high risk disease. In this multicenter, investigator initiated study, 406 patients with tumors measuring up to 3 cm in greatest dimension received weekly treatment with TAXOL® (Paclitaxel) and HERCEPTIN (Trastuzumab) for 12 weeks, followed by 9 months of HERCEPTIN® monotherapy. Close to 50% of the patients had tumors 1 cm in diameter or less, about 40% of the patients had tumors 1-2 cm in diameter and majority of the tumors (56%) were high grade. Treatment regimen consisted of TAXOL® 80 mg/m2 IV weekly, for 12 weeks and HERCEPTIN® 4 mg/kg loading dose IV on day 1, followed by 2 mg/kg weekly, for a total of 12 doses followed by HERCEPTIN® 6 mg/kg every 3 weeks for an additional 40 weeks, for a total of 52 weeks of treatment with HERCEPTIN®. Patients who underwent lumpectomy received either partial breast radiation before the initiation of the therapy, or radiation of the whole breast, following completion of treatment with TAXOL®. Treatment with HERCEPTIN® was continued during the time patient was receiving radiation therapy. Adjuvant hormonal therapy was recommended for women with hormone-receptor positive tumors after the completion of TAXOL® treatment. The primary end point was survival free from invasive disease. The median follow up period was 4 years. The 3-year rate of survival free from invasive disease was 98.7%. Treatment was very well tolerated with a low incidence of heart failure (0.5%) and neuropathy. The authors concluded that a less toxic regimen such as HERCEPTIN® given along with weekly TAXOL® has significant efficacy, decreasing the risk of recurrence in this patient group, most notable during the first three years after diagnosis. They also point out that the risk of recurrence of breast cancer is greatest during the first 3-5 years after diagnosis and it would seem unlikely that a different chemotherapy regimen administered with HERCEPTIN® would impact the risk of late recurrences. Tolaney SM, Barry WT, Dang CT, et al. N Engl J Med 2015;372:134-141
HERCEPTIN® binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Adjuvant chemotherapy in combination with HERCEPTIN® has been shown to reduce the relative risk of relapse by 52% and relative risk of death by 33%. The National Comprehensive Cancer Network (NCCN) has recommended adjuvant chemotherapy with HERCEPTIN® for patients with small, HER positive, node-negative tumors, including those with T1bN0 tumors, even though there are little or no data supporting this recommendation, because these patients are generally not included in adjuvant therapy studies. Further, the chemotherapy regimens often recommended (ACTH, TCH) along with HERCEPTIN® are relatively toxic. The authors in this study chose a less toxic chemotherapy regimen than the regimens often recommended for those patients with high risk disease. In this multicenter, investigator initiated study, 406 patients with tumors measuring up to 3 cm in greatest dimension received weekly treatment with TAXOL® (Paclitaxel) and HERCEPTIN (Trastuzumab) for 12 weeks, followed by 9 months of HERCEPTIN® monotherapy. Close to 50% of the patients had tumors 1 cm in diameter or less, about 40% of the patients had tumors 1-2 cm in diameter and majority of the tumors (56%) were high grade. Treatment regimen consisted of TAXOL® 80 mg/m2 IV weekly, for 12 weeks and HERCEPTIN® 4 mg/kg loading dose IV on day 1, followed by 2 mg/kg weekly, for a total of 12 doses followed by HERCEPTIN® 6 mg/kg every 3 weeks for an additional 40 weeks, for a total of 52 weeks of treatment with HERCEPTIN®. Patients who underwent lumpectomy received either partial breast radiation before the initiation of the therapy, or radiation of the whole breast, following completion of treatment with TAXOL®. Treatment with HERCEPTIN® was continued during the time patient was receiving radiation therapy. Adjuvant hormonal therapy was recommended for women with hormone-receptor positive tumors after the completion of TAXOL® treatment. The primary end point was survival free from invasive disease. The median follow up period was 4 years. The 3-year rate of survival free from invasive disease was 98.7%. Treatment was very well tolerated with a low incidence of heart failure (0.5%) and neuropathy. The authors concluded that a less toxic regimen such as HERCEPTIN® given along with weekly TAXOL® has significant efficacy, decreasing the risk of recurrence in this patient group, most notable during the first three years after diagnosis. They also point out that the risk of recurrence of breast cancer is greatest during the first 3-5 years after diagnosis and it would seem unlikely that a different chemotherapy regimen administered with HERCEPTIN® would impact the risk of late recurrences. Tolaney SM, Barry WT, Dang CT, et al. N Engl J Med 2015;372:134-141
Sentinel Lymph Node Biopsy for Patients with Early-Stage Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. Surgical resection of the axillary lymph nodes in addition to potentially removing cancer that may have spread, also facilitates staging of breast cancer.  The sentinel node is the first lymph node(s) to which cancer cells are most likely to metastasize from a primary tumor. With the introduction of intraoperative lymphatic mapping in the 1990s, Sentinel Lymph Node Biopsy (SLNB) has gained general acceptance and is the preferred procedure in appropriate circumstances. Unlike Axillary Lymph Node Dissection (ALND), SLNB is associated with a lower incidence of Lymphedema, seroma at the surgery site, paresthesias and restriction of joint movement. Nine randomized clinical trials have not shown any difference in mortality among patients who underwent ALND or SLNB for either lymph node metastases or negative sentinel lymph nodes, validating Sentinel Lymph Node Biopsy (SLNB). The American Society of Clinical Oncology (ASCO) first published guidelines on the use of SLNB for patients with early stage breast cancer in 2005, based on one randomized clinical trial. Since then, additional information from 9 randomized clinical trials and13 cohort studies pertinent to SLNB and ALND has resulted in this ASCO Clinical Practice Guideline Update.
The sentinel node is the first lymph node(s) to which cancer cells are most likely to metastasize from a primary tumor. With the introduction of intraoperative lymphatic mapping in the 1990s, Sentinel Lymph Node Biopsy (SLNB) has gained general acceptance and is the preferred procedure in appropriate circumstances. Unlike Axillary Lymph Node Dissection (ALND), SLNB is associated with a lower incidence of Lymphedema, seroma at the surgery site, paresthesias and restriction of joint movement. Nine randomized clinical trials have not shown any difference in mortality among patients who underwent ALND or SLNB for either lymph node metastases or negative sentinel lymph nodes, validating Sentinel Lymph Node Biopsy (SLNB). The American Society of Clinical Oncology (ASCO) first published guidelines on the use of SLNB for patients with early stage breast cancer in 2005, based on one randomized clinical trial. Since then, additional information from 9 randomized clinical trials and13 cohort studies pertinent to SLNB and ALND has resulted in this ASCO Clinical Practice Guideline Update.
The following recommendations were made by the American Society of Clinical Oncology panel of experts:
1) Women without sentinel lymph node (SLN) metastases should not undergo Axillary Lymph Node Dissection (ALND).
2) Women with one to two metastatic SLNs planning to undergo breast conserving surgery with whole breast radiotherapy should not undergo ALND (in most cases).
3) Women with SLN metastases who will undergo mastectomy should be offered ALND.
4) Women with operable breast cancer and multicentric tumors, those with ductal carcinoma in situ (DCIS) who will undergo mastectomy, those who previously underwent breast and/or axillary surgery and those who received preoperative/neoadjuvant systemic therapy, may be offered SLNB.
5) Women who have large or locally advanced invasive breast cancer (tumor size T3/T4), inflammatory breast cancer, or DCIS (when breast-conserving surgery is planned) or are pregnant, should not undergo SLNB.
Lyman GH, Temin S, Edge SB, et al. J Clin Oncol 2014;32:1365-1383
Final overall survival analysis from the Cleopatra study of first-line pertuzumab, trastuzumab and docetaxel in patients with HER2-positive metastatic breast cancer.
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. Trastuzumab binds to subdomain IV of the HER2 extracellular domain and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Despite this benefit, majority of these patients develop progressive disease within 18 months. The tumors in these patients continue to express HER2 although the lower sensitivity to HER2 targeted agents has been attributed to HER2 independent escape mechanisms. Treatment strategies for this patient population have included switching chemotherapy in subsequent lines of treatment and continuing HERCEPTIN®, combining another HER2 targeted agent, Lapatinib (TYKERB®) with Capecitabine (XELODA®) and dual HER2 inhibition with a combination of HERCEPTIN® and TYKERB®.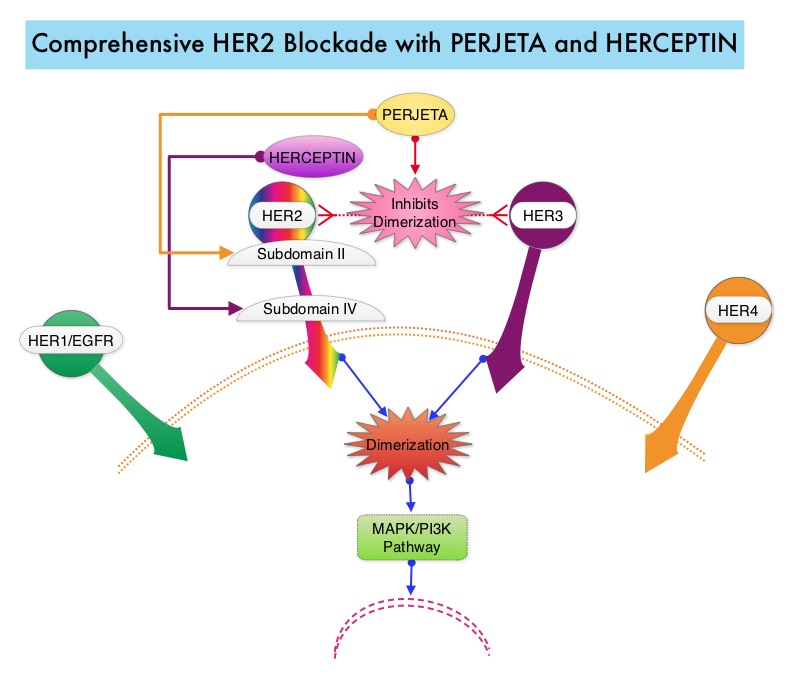 PERJETA® (Pertuzumab) is a recombinant humanized monoclonal antibody that binds to the HER2 at a different epitope of the HER2 extracellular domain (subdomain II) compared to HERCEPTIN® and prevents the dimerization of HER2 with HER3 receptor. PERJETA® stimulates antibody-dependent, cell-mediated cytotoxicity similar to HERCEPTIN®. By combining HERCEPTIN® and PERJETA®, a more comprehensive blockade of HER2 signaling can be accomplished, as these two agents bind to different HER2 epitopes and may complement each other and improve efficacy, as was demonstrated in early phase trials. The CLEOPATRA trial is a phase III study in which 808 treatment naive HER positive metastatic breast cancer patients, were randomly assigned to receive either HERCEPTIN® plus Docetaxel or this two drug combination given along with PERJETA®. PERJETA® was given as an 840 mg loading dose followed by a 420 mg maintenance dose, HERCEPTIN® was given as an 8 mg/kg loading dose followed by a 6 mg/kg maintenance dose and Docetaxel was given at 75 mg/m2 for at least 6 cycles. Treatment was administered every 3 weeks and continued until disease progression. The primary end point of this study was Progression Free Survival and secondary end points included Overall Survival, objective response rate and safety. A previous analysis performed in May 2012 showed that the addition of PERJETA® to the combination of HERCEPTIN® and Docetaxel significantly prolonged Progression Free Survival compared to HERCEPTIN® plus Docetaxel alone (18.5 months vs 12.4 months) but the median overall survival had not been reached then. In this final Overall Survival analysis, at a median follow up of 50 months, median Overall Survival was 56.5 months with the PERJETA® combination compared to 40.8 months in the non-PERJETA® group (hazard ratio [HR] = 0.68; P=0.0002). This meant that adding PERJETA® to HERCEPTIN® and Docetaxel increased the median Overall Survival by 15.7 months. The increase in Progression Free Survival by 6.3 months with the PERJETA® combination, was again maintained (HR = 0.68, P < 0.0001) at the time of the final analysis. The incidence of symptomatic left ventricular dysfunction as well as declines in left ventricular ejection fraction, were rare and similar between the two treatment groups. Based on the CLEOPATRA study, women with HER positive metastatic breast cancer, should be considered candidates, for treatment with a combination of PERJETA®, HERCEPTIN® and Docetaxel. Swain S, Kim S, Cortes J, et al. Presented at: the 2014 Congress of the European Society of Medical Oncology; September 26-30, 2014; Madrid, Spain. Abstract 350O
PERJETA® (Pertuzumab) is a recombinant humanized monoclonal antibody that binds to the HER2 at a different epitope of the HER2 extracellular domain (subdomain II) compared to HERCEPTIN® and prevents the dimerization of HER2 with HER3 receptor. PERJETA® stimulates antibody-dependent, cell-mediated cytotoxicity similar to HERCEPTIN®. By combining HERCEPTIN® and PERJETA®, a more comprehensive blockade of HER2 signaling can be accomplished, as these two agents bind to different HER2 epitopes and may complement each other and improve efficacy, as was demonstrated in early phase trials. The CLEOPATRA trial is a phase III study in which 808 treatment naive HER positive metastatic breast cancer patients, were randomly assigned to receive either HERCEPTIN® plus Docetaxel or this two drug combination given along with PERJETA®. PERJETA® was given as an 840 mg loading dose followed by a 420 mg maintenance dose, HERCEPTIN® was given as an 8 mg/kg loading dose followed by a 6 mg/kg maintenance dose and Docetaxel was given at 75 mg/m2 for at least 6 cycles. Treatment was administered every 3 weeks and continued until disease progression. The primary end point of this study was Progression Free Survival and secondary end points included Overall Survival, objective response rate and safety. A previous analysis performed in May 2012 showed that the addition of PERJETA® to the combination of HERCEPTIN® and Docetaxel significantly prolonged Progression Free Survival compared to HERCEPTIN® plus Docetaxel alone (18.5 months vs 12.4 months) but the median overall survival had not been reached then. In this final Overall Survival analysis, at a median follow up of 50 months, median Overall Survival was 56.5 months with the PERJETA® combination compared to 40.8 months in the non-PERJETA® group (hazard ratio [HR] = 0.68; P=0.0002). This meant that adding PERJETA® to HERCEPTIN® and Docetaxel increased the median Overall Survival by 15.7 months. The increase in Progression Free Survival by 6.3 months with the PERJETA® combination, was again maintained (HR = 0.68, P < 0.0001) at the time of the final analysis. The incidence of symptomatic left ventricular dysfunction as well as declines in left ventricular ejection fraction, were rare and similar between the two treatment groups. Based on the CLEOPATRA study, women with HER positive metastatic breast cancer, should be considered candidates, for treatment with a combination of PERJETA®, HERCEPTIN® and Docetaxel. Swain S, Kim S, Cortes J, et al. Presented at: the 2014 Congress of the European Society of Medical Oncology; September 26-30, 2014; Madrid, Spain. Abstract 350O
Circulating Tumor Cells and Response to Chemotherapy in Metastatic Breast Cancer: SWOG S0500
SUMMARY: Circulating tumor cells (CTCs) are epithelial cells that are shed into the circulation from a primary or metastatic tumor. After being shed, CTCs can remain in the circulation or undergo apoptosis. Evaluation of CTCs during the course of disease and treatment has prognostic value. Because of the very low concentrations of CTCs (1 CTC in the background of millions of normal hematopoietic cells) in the peripheral blood, different technologies have been developed that will allow enrichment and detection of these CTCs. One such technology is the CellSearch® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of Metastatic Breast Cancer (MBC) patients. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK and DAPI positive and CD45 negative. In essence, CTC assessment, is a real time, peripheral blood evaluation (“Liquid Biopsy”) in MBC patients. Previously published studies have concluded that in patients with MBC, increased levels of CTCs prior to administration of a new therapy was associated with poor outcomes and failure of CTCs to drop to below 5 CTCs per 7.5 mL of peripheral blood at 3- 5 weeks after systemic therapy initiation, predicted worse Progression Free Survival (PFS) and Overall Survival (OS), compared to those who did not have increased CTCs at baseline or had increased CTCs at baseline and but not at 3-5 weeks after therapy. With this background, a randomized study was conducted to assess whether changing treatment after one cycle of first line chemotherapy, in those with a persistent increase in CTCs, improved OS. Evaluable patients were initially divided into two groups – Group A (N=276) included patients who did not have increased CTCs at baseline and Group B (N=288) included patients who had 5 or more CTCs per 7.5 mL of peripheral blood. Eligible patients were chemotherapy naïve for MBC and were treated with single agent chemotherapy. The choice of chemotherapy was at the discretion of the attending physician. Patients in Group A remained on initial therapy until disease progression whereas patients in Group B had CTC evaluation at Day 22 and those with decreased CTCs remained on the initial therapy (N=165). Patients who had persistently increased CTCs at Day 22 (N=123), were then randomly assigned to either continue the initial therapy (Group C1) or switch to a different chemotherapy regimen (Group C2). The median Overall Survival for Groups A, B, and C (C1 and C2 combined) were 35 months, 23 months, and 13 months, respectively (P <0.001). There was no difference in median Overall Survival between Groups C1 and C2 (10.7 and 12.5 months, respectively (P = 0.98). The authors concluded that CTCs in patients with Metastatic Breast Cancer receiving first line chemotherapy has significant prognostic value and changing to a different chemotherapy regimen based on persistently increased CTCs after 3 weeks of first line chemotherapy, had no impact in prolonging Overall Survival. This group of patients (C1 and C2) should be encouraged to enroll in clinical trials as standard chemotherapy may not be as effective. CTC count can prognosticate Progression Free Survival and Overall Survival early in the treatment course thereby allowing customized care. Further, CTC enumeration, unlike mucin based serum biomarkers such as CEA and CA15-3, better correlates with clinical and pathological characteristics of the disease. Smerage JB, Barlow WE, Hortobagyi GN, et al. DOI: 10.1200/JCO.2014.56.2561
One such technology is the CellSearch® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of Metastatic Breast Cancer (MBC) patients. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK and DAPI positive and CD45 negative. In essence, CTC assessment, is a real time, peripheral blood evaluation (“Liquid Biopsy”) in MBC patients. Previously published studies have concluded that in patients with MBC, increased levels of CTCs prior to administration of a new therapy was associated with poor outcomes and failure of CTCs to drop to below 5 CTCs per 7.5 mL of peripheral blood at 3- 5 weeks after systemic therapy initiation, predicted worse Progression Free Survival (PFS) and Overall Survival (OS), compared to those who did not have increased CTCs at baseline or had increased CTCs at baseline and but not at 3-5 weeks after therapy. With this background, a randomized study was conducted to assess whether changing treatment after one cycle of first line chemotherapy, in those with a persistent increase in CTCs, improved OS. Evaluable patients were initially divided into two groups – Group A (N=276) included patients who did not have increased CTCs at baseline and Group B (N=288) included patients who had 5 or more CTCs per 7.5 mL of peripheral blood. Eligible patients were chemotherapy naïve for MBC and were treated with single agent chemotherapy. The choice of chemotherapy was at the discretion of the attending physician. Patients in Group A remained on initial therapy until disease progression whereas patients in Group B had CTC evaluation at Day 22 and those with decreased CTCs remained on the initial therapy (N=165). Patients who had persistently increased CTCs at Day 22 (N=123), were then randomly assigned to either continue the initial therapy (Group C1) or switch to a different chemotherapy regimen (Group C2). The median Overall Survival for Groups A, B, and C (C1 and C2 combined) were 35 months, 23 months, and 13 months, respectively (P <0.001). There was no difference in median Overall Survival between Groups C1 and C2 (10.7 and 12.5 months, respectively (P = 0.98). The authors concluded that CTCs in patients with Metastatic Breast Cancer receiving first line chemotherapy has significant prognostic value and changing to a different chemotherapy regimen based on persistently increased CTCs after 3 weeks of first line chemotherapy, had no impact in prolonging Overall Survival. This group of patients (C1 and C2) should be encouraged to enroll in clinical trials as standard chemotherapy may not be as effective. CTC count can prognosticate Progression Free Survival and Overall Survival early in the treatment course thereby allowing customized care. Further, CTC enumeration, unlike mucin based serum biomarkers such as CEA and CA15-3, better correlates with clinical and pathological characteristics of the disease. Smerage JB, Barlow WE, Hortobagyi GN, et al. DOI: 10.1200/JCO.2014.56.2561
Clinical Impact of Delaying Initiation of Adjuvant Chemotherapy in Patients with Breast Cancer
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes, without systemic therapy. Patients with early stage breast cancer often receive adjuvant chemotherapy and this is even more so true for HER positive and triple negative (ER, PR and HER negative) breast cancer patients, who are at an increased risk to develop recurrent disease. Even though majority of the patients start their adjuvant chemotherapy within 4-6 weeks following surgery, the impact of delay in the initiation of adjuvant therapy, on outcomes, has remained unclear. Preclinical models have suggested that there is phase of increased angiogenesis and accelerated growth of micrometastases, as well as development of drug resistant clones, following removal of the primary tumor. Previously published data from a large meta-analysis had suggested that a four week delay in the initiation of adjuvant chemotherapy resulted in a 6% increase in the risk of death and an 8% increase in the risk of relapse. Based on this background information, the authors in this study evaluated the impact of time to initiation of adjuvant chemotherapy, on survival, in patients with various stages and subtypes of early stage breast cancer. In this single institution study, 6,827 women diagnosed with stages I to III breast cancer between 1997 and 2011, were categorized into one of three groups - 30 days or less, 31 to 60 days and 61 days or more, according to the time from definitive surgery to adjuvant chemotherapy. Survival outcomes were then estimated in these three groups. The median follow up was 59.3 months and majority of the patients (84.5%) had stage I or II breast cancer and 15.5% of the patients had stage III disease. The authors noted that outcomes were inferior among patients with stage II and stage III disease when chemotherapy was initiated 61 days or more after surgery, with a 76% increase in the risk of death among patients with stage III disease. This disadvantage was however not noted in patients with stage I disease. Survival estimates based on the tumor sub types revealed that patients with triple negative breast cancer tumors and those with HER-2 positive (Human Epidermal growth factor Receptor- 2) tumors, treated 61 days or more after surgery with HERCEPTIN® (Trastuzumab) based chemotherapy, had the worse survival, compared with those who initiated adjuvant treatment within the first 30 days after surgery. Patients with hormone receptor positive tumors were however not impacted. The authors concluded that delaying the initiation of adjuvant chemotherapy in patients with high risk disease such as those with Stages II and III breast cancer and those with triple negative breast cancer and HER-2 positive tumors, can negatively impact survival outcomes. de Melo Gagliato D, Gonzalez-Angulo AM, Lei X, et al. J Clin Oncol 2014;32:735-744
Phase III trial (Prevention of Early Menopause Study [POEMS]-SWOG S0230) of LHRH analog during chemotherapy (CT) to reduce ovarian failure in early-stage, hormone receptor-negative breast cancer: An international Intergroup trial of SWOG, IBCSG, ECOG, and CALGB (Alliance).
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. Premature Ovarian Failure (POF) is a common unintended consequence of chemotherapy in premenopausal women. Besides of loss of fertility, which can influence treatment decisions in young women, ovarian failure can lead to menopausal symptoms, sexual dysfunction and loss of bone density.  POEMS (Prevention of Early Menopause Study) is a randomized phase III trial designed to evaluate whether the addition of LHRH (Luteinizing Hormone-Releasing Hormone) analog Goserelin (ZOLADEX®), which suppresses the production of estrogens, to Cyclophosphamide based chemotherapy, would reduce POF in breast cancer patients, when compared to chemotherapy alone. Premenopausal patients less than 50 years of age, with hormone negative (ER/PR negative ), Stage I-IIIA breast cancer, scheduled to receive chemotherapy, were randomly assigned to receive standard Cyclophosphamide based chemotherapy with or without monthly ZOLADEX® . Patients in the ZOLADEX® group received 3.6 mg SQ starting 1 week prior to the first dose of chemotherapy. The primary endpoint was ovarian failure at two years (defined as amenorrhea for the prior 6 months AND post-menopausal FSH level). Other endpoints included pregnancy and survival rates. Of the 218 evaluable patients, 135 premenopausal women were evaluable for the primary end point. POF rates were 22% in the chemotherapy alone group and 8% in the ZOLADEX® group (P=0.03). When the definition of POF was more liberal to include EITHER amenorrhea or elevated FSH but not both, POF rates were 45% in the chemotherapy alone group and 20% in the ZOLADEX® group (P=0.006). Among the 218 evaluable patients, more women in the ZOLADEX® group achieved at least one pregnancy (21%) compared to 11% in the chemotherapy alone group (P=0.03). Secondary outcomes also favored the ZOLADEX® group with a Disease free Survival (DFS) rate of 78% in the chemotherapy alone group compared with 89% in the ZOLADEX® group (P=0.04) and Overall Survival (OS) rate of 82% in the chemotherapy alone group compared with 92% in the ZOLADEX® group (P=0.05). The authors concluded that the addition of ZOLADEX® to chemotherapy improved fertility prospects with a lower incidence of Premature Ovarian Failure and more pregnancies. Further, the improved Disease Free Survival and Overall Survival is an important additional perk and prevention of POF with ZOLADEX® may be a consideration not only in premenopausal patients with hormone receptor positive breast cancer but also in other malignancies such as lymphomas, when treated with similar chemotherapeutic agents. Moore HC, Unger JM, Phillips K, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA505)
POEMS (Prevention of Early Menopause Study) is a randomized phase III trial designed to evaluate whether the addition of LHRH (Luteinizing Hormone-Releasing Hormone) analog Goserelin (ZOLADEX®), which suppresses the production of estrogens, to Cyclophosphamide based chemotherapy, would reduce POF in breast cancer patients, when compared to chemotherapy alone. Premenopausal patients less than 50 years of age, with hormone negative (ER/PR negative ), Stage I-IIIA breast cancer, scheduled to receive chemotherapy, were randomly assigned to receive standard Cyclophosphamide based chemotherapy with or without monthly ZOLADEX® . Patients in the ZOLADEX® group received 3.6 mg SQ starting 1 week prior to the first dose of chemotherapy. The primary endpoint was ovarian failure at two years (defined as amenorrhea for the prior 6 months AND post-menopausal FSH level). Other endpoints included pregnancy and survival rates. Of the 218 evaluable patients, 135 premenopausal women were evaluable for the primary end point. POF rates were 22% in the chemotherapy alone group and 8% in the ZOLADEX® group (P=0.03). When the definition of POF was more liberal to include EITHER amenorrhea or elevated FSH but not both, POF rates were 45% in the chemotherapy alone group and 20% in the ZOLADEX® group (P=0.006). Among the 218 evaluable patients, more women in the ZOLADEX® group achieved at least one pregnancy (21%) compared to 11% in the chemotherapy alone group (P=0.03). Secondary outcomes also favored the ZOLADEX® group with a Disease free Survival (DFS) rate of 78% in the chemotherapy alone group compared with 89% in the ZOLADEX® group (P=0.04) and Overall Survival (OS) rate of 82% in the chemotherapy alone group compared with 92% in the ZOLADEX® group (P=0.05). The authors concluded that the addition of ZOLADEX® to chemotherapy improved fertility prospects with a lower incidence of Premature Ovarian Failure and more pregnancies. Further, the improved Disease Free Survival and Overall Survival is an important additional perk and prevention of POF with ZOLADEX® may be a consideration not only in premenopausal patients with hormone receptor positive breast cancer but also in other malignancies such as lymphomas, when treated with similar chemotherapeutic agents. Moore HC, Unger JM, Phillips K, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA505)
Efficacy and safety of continued zoledronic acid every 4 weeks versus every 12 weeks in women with bone metastases from breast cancer: Results of the OPTIMIZE-2 trial.
SUMMARY: Bone is the most common site of metastatic disease, in patients with Breast Cancer. Bisphosphonates inhibit osteoclast-mediated bone resorption and both oral and IV bisphosphonates reduce the risk of developing Skeletal Related Events (SRE’s) and delay the time to SRE’s in patients with Breast Cancer with bone metastases. Bisphosphonates can also reduce bone pain and may improve Quality of life. Of the four bisphosphonates proven to be effective in patients with Breast Cancer with bone metastases, only intravenous Pamidronate (AREDIA®) and Zoledronic acid (ZOMETA®) have been approved in the USA, whereas intravenous and oral Ibandronate and oral Clodronate have been approved in Europe. Both AREDIA® and ZOMETA® are administered every 3 to 4 weeks during the first year, following diagnoses of bone metastases. However, the optimal treatment schedule following this initial phase of treatment has remained unclear. Further, renal toxicity, long bone fractures and OsteoNecrosis of the Jaw (ONJ) have been identified as potential problems with bisphosphonate use. OPTIMIZE-2 is a prospective, randomized, double-blind, multicenter trial, in which the authors evaluated the outcomes of a less intense schedule of ZOMETA® administered every 12 weeks, following one year of the standard initial phase of treatment with bisphosphonates. This study included 403 women with bone metastases from Breast Cancer, who had received 9 or more doses of either intravenous ZOMETA® or AREDIA®, during the first 10-15 months of therapy. The median age was 59 years and patients were randomized (1:1) to receive either ZOMETA® 4 mg IV every 4 weeks (N=200) or every 12 weeks (N=203), for one year. The primary endpoint was Skeletal Related Event (SRE) rate, defined as the proportion of patients with one or more SRE’s (pathologic fractures, spinal cord compression, need for radiotherapy or surgical stabilization of the bone). The primary analysis was non-inferiority, for the difference in SRE rates between the treatment groups. Secondary endpoints included time to first SRE, Skeletal Morbidity Rate (SMR), bone pain score, change in bone turnover markers, and safety. After a median follow up of 11.9 months, the SRE rate was 22% and 23.2% in the ZOMETA® every 4 weeks group and ZOMETA® every 12 weeks group respectively (P=0.724), suggesting that ZOMETA® given every 12 weeks was non-inferior to the q 4 week regimen. The secondary endpoints were comparable as well. More patients had renal toxicities in the ZOMETA® q 4 week group vs q 12 week group (9.6% vs 7.9%, respectively) and two cases (1.0%) of OsteoNecrosis of the Jaw (ONJ) were reported in the ZOMETA® q 4 week group. The authors concluded that the efficacy of continuing ZOMETA® for an additional year at the q 12 week schedule was non-inferior to ZOMETA® given q 4 weeks, among patients who had initially received IV bisphosphonates monthly, for one year or longer. Further the less frequent dosing of ZOMETA® compared with the standard monthly dosing, may be more convenient for the patients and result in less toxicities. Hortobagyi GN, Lipton A, Chew HK, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA9500)
OPTIMIZE-2 is a prospective, randomized, double-blind, multicenter trial, in which the authors evaluated the outcomes of a less intense schedule of ZOMETA® administered every 12 weeks, following one year of the standard initial phase of treatment with bisphosphonates. This study included 403 women with bone metastases from Breast Cancer, who had received 9 or more doses of either intravenous ZOMETA® or AREDIA®, during the first 10-15 months of therapy. The median age was 59 years and patients were randomized (1:1) to receive either ZOMETA® 4 mg IV every 4 weeks (N=200) or every 12 weeks (N=203), for one year. The primary endpoint was Skeletal Related Event (SRE) rate, defined as the proportion of patients with one or more SRE’s (pathologic fractures, spinal cord compression, need for radiotherapy or surgical stabilization of the bone). The primary analysis was non-inferiority, for the difference in SRE rates between the treatment groups. Secondary endpoints included time to first SRE, Skeletal Morbidity Rate (SMR), bone pain score, change in bone turnover markers, and safety. After a median follow up of 11.9 months, the SRE rate was 22% and 23.2% in the ZOMETA® every 4 weeks group and ZOMETA® every 12 weeks group respectively (P=0.724), suggesting that ZOMETA® given every 12 weeks was non-inferior to the q 4 week regimen. The secondary endpoints were comparable as well. More patients had renal toxicities in the ZOMETA® q 4 week group vs q 12 week group (9.6% vs 7.9%, respectively) and two cases (1.0%) of OsteoNecrosis of the Jaw (ONJ) were reported in the ZOMETA® q 4 week group. The authors concluded that the efficacy of continuing ZOMETA® for an additional year at the q 12 week schedule was non-inferior to ZOMETA® given q 4 weeks, among patients who had initially received IV bisphosphonates monthly, for one year or longer. Further the less frequent dosing of ZOMETA® compared with the standard monthly dosing, may be more convenient for the patients and result in less toxicities. Hortobagyi GN, Lipton A, Chew HK, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA9500)
Trastuzumab emtansine versus treatment of physician's choice for pretreated HER2-positive advanced breast cancer (TH3RESA): a randomised, open-label, phase 3 trial.
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. It binds to the extracellular domain of the receptor and blocks the downstream cell signaling pathways (PI3K-AKT pathway) and induces Antibody Dependent Cellular Cytotoxicity (ADCC). HERCEPTIN® in combination with chemotherapy has been proven to significantly improve Progression Free Survival and Overall Survival in patients with advanced breast cancer. Despite this benefit, majority of these patients develop progressive disease within 18 months. The tumors in these patients continue to express HER2 although the lower sensitivity to HER2 targeted agents has been attributed to HER2 independent escape mechanisms. Treatment strategies for this patient population have included switching chemotherapy in subsequent lines of treatment and continuing HERCEPTIN®, combining another HER2 targeted agent, Lapatinib (TYKERB®) with Capecitabine (XELODA®) and dual HER2 inhibition with a combination of HERCEPTIN® and TYKERB®. KADCYLA® (Ado-Trastuzumab Emtansine, T-DM1) is an antibody-drug conjugate (ADC) comprised of the antibody HERCEPTIN® and the chemotherapy agent Emtansine, linked together. Upon binding to the HER2 receptor, it not only inhibits the HER2 signaling pathways but also delivers a chemotherapy agent Emtansine, a microtubule inhibitor, directly inside the tumor cells. This agent is internalized by lysosomes and destroys the HER2-positive tumor cells upon intracellular release.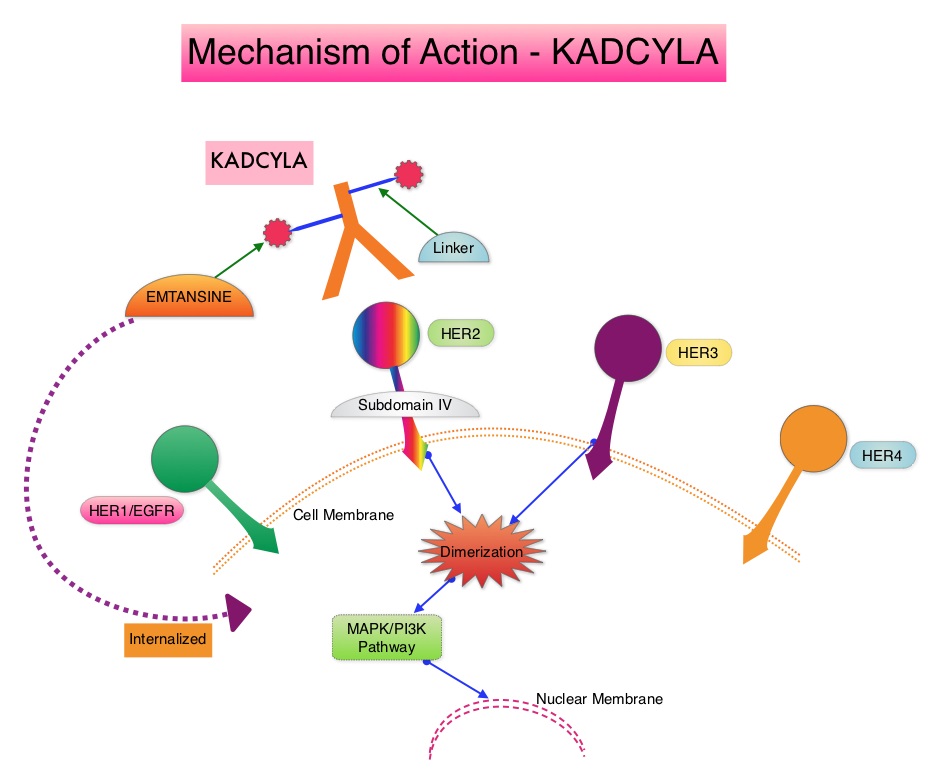 In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® and XELODA® in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a taxane. This study however excluded patients who had previously received TYKERB®. TH3RESA is an open label randomized phase III trial in which KADCYLA® was compared with treatment of physician’s choice, in patients with unresectable locally advanced, recurrent or metastatic breast cancer. Eligible patients had a left ventricular ejection fraction of 50% or more, ECOG performance status of 0-2 and had HER2-positive advanced breast cancer who had received two or more HER2-directed regimens in the advanced setting and had progressed on both HERCEPTIN® and TYKERB® containing regimens in metastatic setting and also had disease progression on a taxane, in any setting. Patients were randomized in a 2:1 ratio to receive either KADCYLA® 3•6 mg/kg intravenously every 21 days (N=404) or treatment of physician’s choice (N=198). Treatment was continued until disease progression or unmanageable toxicity. The Co-primary endpoints were Progression Free Survival (PFS) and Overall Survival. Secondary endpoints included Response Rates, duration of response, safety and quality of life. After a median follow up of 7•2 months in the KADCYLA® group and 6•5 months in the physician's treatment choice group, there was a significant improvement in Progression Free Survival with KADCYLA® compared with physician's treatment choice (6•2 months vs 3•3 months, HR= 0•528, P<0•0001). The interim Overall Survival analysis showed a trend favoring KADCYLA® (HR=0•552, P=0•0034). Patients in the KADCYLA® group had a lower incidence of grade 3 toxicities compared to the patients in the physician’s treatment choice group (32% vs 43%). Grade 3 thrombocytopenia however was more common in the KADCYLA® group compared to the physician’s choice group (5% vs 2%) and this has been attributed to the inhibition of megakaryocyte differentiation by KADCYLA®. The authors concluded that KADCYLA® should be considered the treatment of choice, for patients with HER2-positive advanced breast cancer, who have previously received HERCEPTIN® and TYKERB®. It remains to be seen however, if KADCYLA® is effective in patients who had progressed on Pertuzumab (PERJETA®) based therapies. Krop IE, Kim SB, González-Martín A, et al. Lancet Oncol. 2014;15:689-699
In the EMILIA trial, KADCYLA® was associated with significant increase in Overall Survival when compared with TYKERB® and XELODA® in HER2-positive metastatic breast cancer patients, who had previously received HERCEPTIN® and a taxane. This study however excluded patients who had previously received TYKERB®. TH3RESA is an open label randomized phase III trial in which KADCYLA® was compared with treatment of physician’s choice, in patients with unresectable locally advanced, recurrent or metastatic breast cancer. Eligible patients had a left ventricular ejection fraction of 50% or more, ECOG performance status of 0-2 and had HER2-positive advanced breast cancer who had received two or more HER2-directed regimens in the advanced setting and had progressed on both HERCEPTIN® and TYKERB® containing regimens in metastatic setting and also had disease progression on a taxane, in any setting. Patients were randomized in a 2:1 ratio to receive either KADCYLA® 3•6 mg/kg intravenously every 21 days (N=404) or treatment of physician’s choice (N=198). Treatment was continued until disease progression or unmanageable toxicity. The Co-primary endpoints were Progression Free Survival (PFS) and Overall Survival. Secondary endpoints included Response Rates, duration of response, safety and quality of life. After a median follow up of 7•2 months in the KADCYLA® group and 6•5 months in the physician's treatment choice group, there was a significant improvement in Progression Free Survival with KADCYLA® compared with physician's treatment choice (6•2 months vs 3•3 months, HR= 0•528, P<0•0001). The interim Overall Survival analysis showed a trend favoring KADCYLA® (HR=0•552, P=0•0034). Patients in the KADCYLA® group had a lower incidence of grade 3 toxicities compared to the patients in the physician’s treatment choice group (32% vs 43%). Grade 3 thrombocytopenia however was more common in the KADCYLA® group compared to the physician’s choice group (5% vs 2%) and this has been attributed to the inhibition of megakaryocyte differentiation by KADCYLA®. The authors concluded that KADCYLA® should be considered the treatment of choice, for patients with HER2-positive advanced breast cancer, who have previously received HERCEPTIN® and TYKERB®. It remains to be seen however, if KADCYLA® is effective in patients who had progressed on Pertuzumab (PERJETA®) based therapies. Krop IE, Kim SB, González-Martín A, et al. Lancet Oncol. 2014;15:689-699
Randomized comparison of adjuvant aromatase inhibitor (AI) exemestane (E) plus ovarian function suppression (OFS) vs tamoxifen (T) plus OFS in premenopausal women with hormone receptor-positive (HR+) early breast cancer (BC): Joint analysis of IBCSG TEXT and SOFT trials.
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive (Estrogen Receptor/Progesterone Receptor positive) and this is a predictor of response to endocrine therapy. In premenopausal woman, the ovary is the main source of estrogen production, whereas in postmenopausal women, the primary source of estrogen is the Aromatase enzyme mediated conversion of androstenedione and testosterone to estrone and estradiol in extragonadal/peripheral tissues. Presently available therapies include Tamoxifen and other Selective ER Modulators, which modulate ER alpha activity, Aromatase Inhibitors and Ovarian ablation that decrease estrogen production and FASLODEX® (Fulvestrant) that down regulates Estrogen Receptor. Aromatase Inhibitors (AI’s) are often prescribed, due to their superiority over Tamoxifen, for postmenopausal women with Hormone Receptor positive breast tumors, in adjuvant as well as metastatic settings. AI’s however, are not effective in premenopausal women, as these individuals derive their estrogen mainly from ovaries and not extragonadal tissues. The 2000 Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) overview, as well as subsequent studies comparing adjuvant ovarian ablation/suppression with adjuvant chemotherapy in premenopausal women with hormone positive breast tumors, have demonstrated similar magnitude of benefit. The TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) are two phase III randomized trials, conducted at the same time and included premenopausal women (average age was 43 years) with hormone receptor positive early breast cancer. In the joint analysis of these two trials in which 5738 women were enrolled, the authors set out to answer 2 important questions - whether adjuvant AI improves outcomes in this patient group when their Ovarian Function is suppressed and whether there is any benefit with Ovarian Function suppression in premenopausal women suitable for adjuvant Tamoxifen. TEXT randomized patients (N=2672) within 3 months of surgery to 5 years of AROMASIN® (Exemestane) plus Ovarian Function Suppression (OFS) or 5 years of Tamoxifen plus OFS. The SOFT study randomized patients (N=3066) to 5 years of AROMASIN® plus OFS or 5 years of Tamoxifen plus OFS or 5 years of Tamoxifen alone. OFS choices included oophorectomy, ovarian irradiation or 5 years of TRELSTAR® (Triptorelin), a GnRH (Gonadotropin Releasing Hormone) agonist. The primary endpoint of these two studies was Disease Free Survival (DFS). In this joint analysis the outcomes for 4690 women randomized to receive AROMASIN® plus OFS or Tamoxifen plus OFS for 5 years, were analyzed. The 5 year Disease Free Survival was 91.1% in the AROMASIN® plus OFS group and 87.3% in the Tamoxifen plus OFS group (HR=0.72, P<0.0002). Compared to patients receiving Tamoxifen plus OFS, AROMASIN® plus OFS reduced the relative risk of premenopausal women developing a subsequent invasive breast cancer by 28% and the relative risk of breast cancer recurrence by 34%. The authors concluded that this largest joint analysis, evaluating adjuvant AI therapy with OFS in premenopausal women with Hormone receptor positive breast cancer, has demonstrated that 5 years of highly effective adjuvant endocrine therapy without chemotherapy can result in excellent outcomes. Further, AROMASIN® may be better than tamoxifen, when given with Ovarian Function Suppression. Pagani O, Regan MM, Walley B, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA1)
Presently available therapies include Tamoxifen and other Selective ER Modulators, which modulate ER alpha activity, Aromatase Inhibitors and Ovarian ablation that decrease estrogen production and FASLODEX® (Fulvestrant) that down regulates Estrogen Receptor. Aromatase Inhibitors (AI’s) are often prescribed, due to their superiority over Tamoxifen, for postmenopausal women with Hormone Receptor positive breast tumors, in adjuvant as well as metastatic settings. AI’s however, are not effective in premenopausal women, as these individuals derive their estrogen mainly from ovaries and not extragonadal tissues. The 2000 Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) overview, as well as subsequent studies comparing adjuvant ovarian ablation/suppression with adjuvant chemotherapy in premenopausal women with hormone positive breast tumors, have demonstrated similar magnitude of benefit. The TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) are two phase III randomized trials, conducted at the same time and included premenopausal women (average age was 43 years) with hormone receptor positive early breast cancer. In the joint analysis of these two trials in which 5738 women were enrolled, the authors set out to answer 2 important questions - whether adjuvant AI improves outcomes in this patient group when their Ovarian Function is suppressed and whether there is any benefit with Ovarian Function suppression in premenopausal women suitable for adjuvant Tamoxifen. TEXT randomized patients (N=2672) within 3 months of surgery to 5 years of AROMASIN® (Exemestane) plus Ovarian Function Suppression (OFS) or 5 years of Tamoxifen plus OFS. The SOFT study randomized patients (N=3066) to 5 years of AROMASIN® plus OFS or 5 years of Tamoxifen plus OFS or 5 years of Tamoxifen alone. OFS choices included oophorectomy, ovarian irradiation or 5 years of TRELSTAR® (Triptorelin), a GnRH (Gonadotropin Releasing Hormone) agonist. The primary endpoint of these two studies was Disease Free Survival (DFS). In this joint analysis the outcomes for 4690 women randomized to receive AROMASIN® plus OFS or Tamoxifen plus OFS for 5 years, were analyzed. The 5 year Disease Free Survival was 91.1% in the AROMASIN® plus OFS group and 87.3% in the Tamoxifen plus OFS group (HR=0.72, P<0.0002). Compared to patients receiving Tamoxifen plus OFS, AROMASIN® plus OFS reduced the relative risk of premenopausal women developing a subsequent invasive breast cancer by 28% and the relative risk of breast cancer recurrence by 34%. The authors concluded that this largest joint analysis, evaluating adjuvant AI therapy with OFS in premenopausal women with Hormone receptor positive breast cancer, has demonstrated that 5 years of highly effective adjuvant endocrine therapy without chemotherapy can result in excellent outcomes. Further, AROMASIN® may be better than tamoxifen, when given with Ovarian Function Suppression. Pagani O, Regan MM, Walley B, et al. J Clin Oncol 32:5s, 2014 (suppl; abstr LBA1)
Clinical validity of circulating tumour cells in patients with metastatic breast cancer: a pooled analysis of individual patient data
SUMMARY: Circulating tumor cells (CTCs) are epithelial cells that are shed into the circulation from a primary or metastatic tumor. After being shed, CTCs can remain in the circulation or undergo apoptosis.  Evaluation of CTCs during the course of disease has prognostic value. Because of the very low concentrations of CTCs (1 CTC in the background of millions of normal hematopoietic cells) in the peripheral blood, different technologies have been developed that will allow enrichment and detection of these CTCs. One such technology is the CellSearch® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of Metastatic Breast Cancer (MBC) patients. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK positive, CD45 negative and DAPI positive. In essence, CTC assessment is a real time, peripheral blood evaluation (“Liquid Biopsy”) in MBC patients. The authors in this study conducted a pooled analysis to assess the clinical validity of CTCs in patients with MBC, as previously published studies reported contradictory results and were unable to ascertain whether enumeration of CTCs had better prognostic value than the traditional clinical and pathological features of the tumor and serum tumor markers. Data was gathered on 1944 patients with MBC, who had participated in clinical trials at 17 centers between 2003 and 2012. Participants in these studies were starting a new line of therapy (predominantly chemotherapy), and these studies had CTC quantification using CellSearch® platform, before start of new treatment (baseline), data for Progression Free Survival, Overall Survival or both. Using accepted statistical methodologies, the authors noted the following findings-
Evaluation of CTCs during the course of disease has prognostic value. Because of the very low concentrations of CTCs (1 CTC in the background of millions of normal hematopoietic cells) in the peripheral blood, different technologies have been developed that will allow enrichment and detection of these CTCs. One such technology is the CellSearch® system which is the first FDA-approved test for CTC assessment, in the peripheral blood of Metastatic Breast Cancer (MBC) patients. This automated system is able to enrich the peripheral blood sample with CTCs and the cells then are fluorescently stained for CytoKeratins (CK8,18 and 19), Common Leukocyte Antigen (CD45) and a nuclear dye (DAPI). CTCs are identified when they are CK positive, CD45 negative and DAPI positive. In essence, CTC assessment is a real time, peripheral blood evaluation (“Liquid Biopsy”) in MBC patients. The authors in this study conducted a pooled analysis to assess the clinical validity of CTCs in patients with MBC, as previously published studies reported contradictory results and were unable to ascertain whether enumeration of CTCs had better prognostic value than the traditional clinical and pathological features of the tumor and serum tumor markers. Data was gathered on 1944 patients with MBC, who had participated in clinical trials at 17 centers between 2003 and 2012. Participants in these studies were starting a new line of therapy (predominantly chemotherapy), and these studies had CTC quantification using CellSearch® platform, before start of new treatment (baseline), data for Progression Free Survival, Overall Survival or both. Using accepted statistical methodologies, the authors noted the following findings-
1) At baseline prior to starting treatment, 47% (N=911) patients had 5 or more CTCs per 7•5 mL of peripheral blood, suggesting more aggressive disease and this was associated with decreased Progression Free Survival (P<0•0001) and Overall Survival (P<0•0001), compared with patients with less than 5 CTCs per 7•5 mL at baseline.
2) At 3-5 weeks after start of treatment (1-2 cycles of treatment), when adjusted for baseline CTC count, 5 or more CTCs per 7•5 mL of peripheral blood was associated with shortened Progression Free Survival (P<0•0001) and Overall Survival (P<0•0001), suggesting that these patients were treatment resistant.
3) An early decrease in the CTC at week 3-5, from a high baseline of 5 or more CTCs per 7•5 mL to less than 5 CTCs per 7.5 ml was associated with significantly longer Progression Free Survival and Overall Survival.
4) Enumeration of CTCs was a better predictor of prognosis than mucin based serum biomarkers such as CEA and CA15-3.
The prognostic value of high CTC count at baseline and at 3-5 weeks of treatment, on Progression Free Survival and Overall Survival was maintained in all subgroups tested, regardless of breast cancer subtypes and type of therapy patients received. Based on this pooled analysis, with the largest assessment of CTC enumeration in MBC patients, the authors concluded that CTC count can prognosticate Progression Free Survival and Overall Survival early in the treatment course, allowing customized care. Further, CTC enumeration, unlike serum tumor markers, correlates with clinical and pathological characteristics. Bidard F, Peeters DJ, Fehm T, et al. The Lancet Oncology 2014;15:406-414
Everolimus plus exemestane as first-line therapy in HR+, HER2− advanced breast cancer in BOLERO-2
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. Approximately 75% of patients with breast cancer are hormone receptor positive and 15%-20% of breast cancers overexpress HER2/neu oncogene. As such a significant number of breast cancers are Hormone Receptor (HR) positive and HER2 negative. Aromatase Inhibitors (AI’s) are often prescribed, due to their superiority over Tamoxifen, for post menopausal women with HR+ and HER2-negative breast tumors, both in adjuvant as well as metastatic settings. These patients will eventually develop progressive disease on endocrine therapy with AI’s, attributed to endocrine resistance. The average Progression Free Survival for these patients is 4-6 months when treated with other hormonal interventions including higher doses of FASLODEX® (Fulvestrant). Further, FASLODEX® has not been shown to be superior to steroidal or non steroidal AI’s. The mechanism of endocrine resistance has been attributed to cross talk between Estrogen Receptor signaling and PI3K/AKT/mTOR pathway. The PI3K/AKT/mTOR is a complex pathway, essential for cell proliferation, survival and apoptosis (programmed cell death). This pathway is of interest because of its increased activity in malignant cells as a result of amplification or mutation of the genes associated with PI3-kinase (phosphatidylinositol-3 kinase) and AKT, as well as loss of function of PTEN. PTEN normally prevents activation of AKT and its downstream pathways. Therefore, elevated ER signaling and breast cancer progression has been associated with activation of this mTOR (mammalian Target Of Rapamycin) pathway. Everolimus (AFINITOR®) is a mTOR inhibitor that has been shown to inhibit ER signaling and restore sensitivity to anti-estrogen therapies. With this preclinical knowledge, the Breast cancer trial of OraL EveROlimus-2 (BOLERO-2), which is a randomized, multicenter phase III trial was conducted to evaluate the benefit of combining steroidal AI, AROMASIN® (Exemestane) and an mTOR inhibitor AFINITOR®, for treatment of postmenopausal patients with HR+ and HER2 negative advanced breast cancer, who had either recurrent or progressive disease after non steroidal AI’s. Seven hundred and twenty four (N=724) patients were randomly assigned in a 2:1 ratio to receive either a combination of a steroidal AI, AROMASIN® at 25 mg PO QD and AFINITOR® at 10 mg PO QD (N=485) or AROMASIN® along with a placebo (N=239). Both treatment groups were well balanced and patients were stratified according to sensitivity to previous hormonal therapy and presence of visceral metastases. The primary endpoint for this study was Progression Free Survival (PFS) and secondary endpoints included overall Response Rate (RR), Clinical Benefit Rate (CBR defined as complete response + partial response + stable disease for at least 6 months), Overall Survival, Quality of Life, changes in bone marker levels and patient safety. In this study, close to 60% of the patients had visceral disease and approximately 80% of the patients had prior therapies for metastatic disease, with only 20% receiving the study drugs as their first therapy for metastatic disease. The combination of AFINITOR® and AROMASIN® significantly prolonged PFS compared to AROMASIN® alone (11 vs 4.1 months, HR=0.38, P<0.0001) and the Clinical Benefit Rate was 51% in the combination group and 26% in the AROMASIN® alone group (P<0.0001). The combination of AFINITOR® and AROMASIN® benefitted all subgroups of patients including those who had disease recurrence during or after neoadjuvant/adjuvant non steroidal AI therapy, those with visceral and bone metastases, as well as those who had prior chemotherapy. The most common adverse events in all age groups were rash, stomatitis, fatigue, diarrhea and nausea and these toxicities were manageable. The authors concluded that AFINITOR® given along with AROMASIN® in the study population can decrease the risk of disease progression by 60% and can double the Clinical Benefit Rate, compared to AROMASIN® alone, even in patients 65 years of age or older, thereby delaying the need for chemotherapy. Beck JT, Hortobagyi GN, Campone M, et al. Breast Cancer Res Treat. 2014; 143: 459–467
The mechanism of endocrine resistance has been attributed to cross talk between Estrogen Receptor signaling and PI3K/AKT/mTOR pathway. The PI3K/AKT/mTOR is a complex pathway, essential for cell proliferation, survival and apoptosis (programmed cell death). This pathway is of interest because of its increased activity in malignant cells as a result of amplification or mutation of the genes associated with PI3-kinase (phosphatidylinositol-3 kinase) and AKT, as well as loss of function of PTEN. PTEN normally prevents activation of AKT and its downstream pathways. Therefore, elevated ER signaling and breast cancer progression has been associated with activation of this mTOR (mammalian Target Of Rapamycin) pathway. Everolimus (AFINITOR®) is a mTOR inhibitor that has been shown to inhibit ER signaling and restore sensitivity to anti-estrogen therapies. With this preclinical knowledge, the Breast cancer trial of OraL EveROlimus-2 (BOLERO-2), which is a randomized, multicenter phase III trial was conducted to evaluate the benefit of combining steroidal AI, AROMASIN® (Exemestane) and an mTOR inhibitor AFINITOR®, for treatment of postmenopausal patients with HR+ and HER2 negative advanced breast cancer, who had either recurrent or progressive disease after non steroidal AI’s. Seven hundred and twenty four (N=724) patients were randomly assigned in a 2:1 ratio to receive either a combination of a steroidal AI, AROMASIN® at 25 mg PO QD and AFINITOR® at 10 mg PO QD (N=485) or AROMASIN® along with a placebo (N=239). Both treatment groups were well balanced and patients were stratified according to sensitivity to previous hormonal therapy and presence of visceral metastases. The primary endpoint for this study was Progression Free Survival (PFS) and secondary endpoints included overall Response Rate (RR), Clinical Benefit Rate (CBR defined as complete response + partial response + stable disease for at least 6 months), Overall Survival, Quality of Life, changes in bone marker levels and patient safety. In this study, close to 60% of the patients had visceral disease and approximately 80% of the patients had prior therapies for metastatic disease, with only 20% receiving the study drugs as their first therapy for metastatic disease. The combination of AFINITOR® and AROMASIN® significantly prolonged PFS compared to AROMASIN® alone (11 vs 4.1 months, HR=0.38, P<0.0001) and the Clinical Benefit Rate was 51% in the combination group and 26% in the AROMASIN® alone group (P<0.0001). The combination of AFINITOR® and AROMASIN® benefitted all subgroups of patients including those who had disease recurrence during or after neoadjuvant/adjuvant non steroidal AI therapy, those with visceral and bone metastases, as well as those who had prior chemotherapy. The most common adverse events in all age groups were rash, stomatitis, fatigue, diarrhea and nausea and these toxicities were manageable. The authors concluded that AFINITOR® given along with AROMASIN® in the study population can decrease the risk of disease progression by 60% and can double the Clinical Benefit Rate, compared to AROMASIN® alone, even in patients 65 years of age or older, thereby delaying the need for chemotherapy. Beck JT, Hortobagyi GN, Campone M, et al. Breast Cancer Res Treat. 2014; 143: 459–467
Yoga's Impact on Inflammation, Mood, and Fatigue in Breast Cancer Survivors: A Randomized Controlled Trial
SUMMARY: Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%), will develop invasive breast cancer during their lifetime. Approximately, 233,000 new cases of invasive breast cancer will be diagnosed in 2014 and 40,000 women will die of the disease. According to the SEER database, the 5 year survival of women with early stage breast cancer is well over 90%. This is mainly because of early detection and effective therapies. A third of these cancer survivors however are likely to be physically inactive due to fatigue, general deconditioning and effects of cancer treatment. There is a reduction in their cardiorespiratory fitness. Chronic inflammation has been implicated in decreased physical functioning, disability and mortality, even in those who are in remission. Regular exercise has been shown to reduce inflammation and fatigue. The authors in this study evaluated the impact of Yoga, which provides graded exercise, on inflammation, mood and fatigue. Yoga by definition is a physical, mental, and spiritual practice and hatha Yoga which is practiced in the Western countries, consists of physical and mental strength building exercises and postures. In this randomized controlled study, 200 breast cancer survivors between ages 27 and 76 years, were assigned to either the Yoga group (N=100) or control/no Yoga group (N=100). Both groups were well balanced with no significant difference between the groups in measures of activity, fatigue, body mass index or depressive symptoms. Enrolled patients were stratified by cancer stage and whether radiation therapy was given or not. Participants had Stage 0 – Stage IIIa breast cancer and had completed cancer treatment within the past three years and were at least 2 months post surgery, adjuvant chemotherapy or radiation treatment. Patients in the Yoga group participated in a 90 minute Yoga class, twice a week for 12 weeks (3 months). The protocol included the measurement of cytokines associated with inflammation and they included Interleukin-6 (IL-6), Interleukin-1beta (IL-1b) and Tumor Necrosis Factor-alfa (TNFa). In addition, other validated instruments were utilized to measure Fatigue and Vitality. Immediately following 12 weeks of Yoga intervention, there was a significant improvement in the Vitality score in the Yoga group (P=0.01) and at 3 months post intervention, the Vitality score was even higher (P=0.01). Fatigue was significantly lower 3 months post intervention, in the Yoga group (P=0.002). At 3 months post intervention, all inflammatory cytokines were lower as well, in the Yoga group - IL-6 (P=0.027), TNFa (P=0.027) and IL-1b (P=0.037). More frequent Yoga practice resulted in greater benefits, with improved sleep and decreased inflammation. It should be noted that sleep problems are 2-3 times more common in cancer survivors and close to two thirds of the cancer survivors experience insomnia. The authors concluded that this is the first and largest randomized controlled study that has demonstrated that practice of Yoga or similar such activities, can substantially reduce inflammation and Fatigue and improve Vitality in breast cancer survivors. Kiecolt-Glaser JK, Bennett JM, Andridge R, et al. J Clin Oncol 2014;32:1040-1049
Chemotherapy for isolated locoregional recurrence of breast cancer (CALOR): a randomised trial
SUMMARY: It has been well recognized that patients with Breast Cancer with Isolated LocoRegional Recurrences (ILRR), without evidence of distant metastasis, are at a high risk of developing subsequent distant metastasis and may have poor outcomes, in spite of surgical resection of the ILRR. The role of systemic chemotherapy following surgery in this patient population has remained unclear although hormonal intervention in ER positive patients demonstrated disease free survival benefit. To address this question, three cooperative groups, The International Breast Cancer Study Group (IBCSG), Breast International Group (BIG) and NSABP collaborated and conducted this study to find out whether adjuvant chemotherapy improves the outcome of patients with ILRR following surgical resection. In this multicenter trial, 162 patients were randomly assigned to receive chemotherapy (N=85) or no chemotherapy (N=77). Eligible patients had histologically proven first ILRR resected and radiotherapy was recommended for all patients, but was mandated for those with microscopically involved surgical margins. All patients with ER or PR positive recurrent tumors received endocrine therapy and patients randomized to the chemotherapy group received at least two standard cytotoxic drugs (investigators choice) for 3-6 months. Both treatment groups were well balanced, and hormone receptor status was positive in 68% of the patients belonging to each group. The primary endpoint was Disease Free Survival and secondary endpoints included Overall Survival. The five year Disease Free Survival in the adjuvant chemotherapy group was 69% compared with 57% in the no chemotherapy group (HR=0.59, P=0.046). The five year overall survival was also significantly longer in the chemotherapy group compared to the no chemotherapy group (88% vs 76%, HR=0.41, P=0.024). With regards to the chemotherapy benefit based on hormone receptor status, patients with hormone receptor negative ILRR, had a longer Disease Free Survival with adjuvant chemotherapy (P=0.046). The authors concluded that adjuvant chemotherapy significantly improves Disease Free and Overall Survival in Breast Cancer patients with ILRR. This benefit is even more so, in those with ER negative ILRR. Adjuvant chemotherapy should therefore be considered in this patient population. Aebi S, Gelber S, Anderson SJ, et al. The Lancet Oncology 2014;15:156-163
Rapid Increase in Breast Magnetic Resonance Imaging Use Trends From 2000 to 2011
SUMMARY: Breast cancer is the most common cancer among women in the United States and 1 in 8 women will develop invasive breast cancer during their lifetime. Screening mammography complemented by breast self exam and clinical breast exam has resulted in early detection of breast cancer and successful outcomes. Even though mammography is a sensitive screening test, a small percentage of breast cancers may not show up on mammograms but may be palpable on examination by the patient or the clinician. Further, mammograms are less likely to find breast tumors in younger women with dense breast tissue. A breast Magnetic Resonance Imaging (MRI) is more sensitive than mammography although the specificity of a breast MRI is lower, resulting in a higher rate of false-positive findings and potentially unnecessary biopsies. Microcalcifications in the breast can be missed by a breast MRI. Taking these factors into consideration, appropriate utilization of breast MRI becomes relevant.
 The American Cancer Society (ACS) recommends an annual MRI as an adjunct to screening mammogram and clinical breast exam in certain groups with increased risk of breast cancer (See Figures). The authors conducted this study to determine the utilization of breast (MRI) in community settings. In this retrospective study, the authors reviewed data on 10,518 women from a not-for-profit health plan, and these women had at least one breast MRI between January 2000 and December 2011. The appropriateness of a breast MRI was determined using a prediction model obtained from electronic medical records on a subset of patients. Between 2000 and 2009, there was a 20-fold increase in the use of breast MRI from 6.5 per 10,000 women in the year 2000 to 130.7 per 10,000 in 2009. This increase then declined and stabilized to 104.8 per 10,000 by 2011. With regards to indications for a breast MRI, 51.7% had a family history of breast cancer, 30.1% had a personal history of breast cancer and 3.5% of women had a documented genetic mutation. It is interesting to note that of those who received screening or surveillance breast MRI’s, only 21% fulfilled the American Cancer Society (ACS) criteria for a breast MRI. Conversely, fewer participants (48.4%), with documented deleterious genetic mutations, received breast MRI screening. The authors concluded that breast MRI was over utilized in those who did not fit the ACS criteria and was under utilized in those with documented genetic mutations. It is their opinion that routine breast MRI screening is not recommended for a new breast cancer diagnosis or for breast cancer surveillance and should only be considered for the group of individuals who have the most benefit. Breast MRI is performed preferably between days 7-15 of menstrual cycle for premenopausal women, using a dedicated breast coil, with the ability to perform a biopsy under MRI guidance by experienced radiologists, during the same visit. Stout NK, Nekhlyudov L, Li L, et al. JAMA Intern Med. 2014;174:114-121.
The American Cancer Society (ACS) recommends an annual MRI as an adjunct to screening mammogram and clinical breast exam in certain groups with increased risk of breast cancer (See Figures). The authors conducted this study to determine the utilization of breast (MRI) in community settings. In this retrospective study, the authors reviewed data on 10,518 women from a not-for-profit health plan, and these women had at least one breast MRI between January 2000 and December 2011. The appropriateness of a breast MRI was determined using a prediction model obtained from electronic medical records on a subset of patients. Between 2000 and 2009, there was a 20-fold increase in the use of breast MRI from 6.5 per 10,000 women in the year 2000 to 130.7 per 10,000 in 2009. This increase then declined and stabilized to 104.8 per 10,000 by 2011. With regards to indications for a breast MRI, 51.7% had a family history of breast cancer, 30.1% had a personal history of breast cancer and 3.5% of women had a documented genetic mutation. It is interesting to note that of those who received screening or surveillance breast MRI’s, only 21% fulfilled the American Cancer Society (ACS) criteria for a breast MRI. Conversely, fewer participants (48.4%), with documented deleterious genetic mutations, received breast MRI screening. The authors concluded that breast MRI was over utilized in those who did not fit the ACS criteria and was under utilized in those with documented genetic mutations. It is their opinion that routine breast MRI screening is not recommended for a new breast cancer diagnosis or for breast cancer surveillance and should only be considered for the group of individuals who have the most benefit. Breast MRI is performed preferably between days 7-15 of menstrual cycle for premenopausal women, using a dedicated breast coil, with the ability to perform a biopsy under MRI guidance by experienced radiologists, during the same visit. Stout NK, Nekhlyudov L, Li L, et al. JAMA Intern Med. 2014;174:114-121.

Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial.
SUMMARY: The FDA on September 30, 2013 granted accelerated approval to PERJETA® (Pertuzumab) for use in combination with HERCEPTIN® (Trastuzumab) and other chemotherapy for the neoadjuvant (preoperative) treatment of patients with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or with positive lymph nodes), as part of a complete treatment regimen for early breast cancer. Following surgery, patients should continue to receive HERCEPTIN® to complete one year of treatment. The HER family of receptors consist of HER1, HER2, HER3 and HER4.  These proteins are transmembrane tyrosine kinases and are involved in normal cell growth and differentiation. HER1 is also known as Epidermal Growth Factor Receptor or EGFR. These receptors are activated following ligand binding, receptor pairing or dimerization and phosphorylation. This dimerization (receptor pairing) occurs often within the HER family of receptors. This has been no ligand identified for HER2 receptor, although it is able to form homo and heterodimers with other members of the HER family readily. Dimerization of HER2 and HER3 is believed to produce the strongest mitogenic signaling and activates two important pathways that regulate cell survival and growth - Mitogen Activated Protein Kinase (MAPK) pathway and PhosphoInositide 3-Kinase (PI3K) pathway. For this reason inhibiting HER2 dimerization appears to be an important step in the treatment of cancer. Overexpression of HER2 in breast cancer has been associated with higher risk for relapse as well as overall survival. Approximately 20 percent of breast cancers are HER2-positive. HERCEPTIN® is a humanized monoclonal antibody targeting HER2. It binds to the extracellular subdomain IV of the receptor and disrupts ligand independent HER2 downstream cell signaling pathways. PERJETA® is a recombinant, humanized, monoclonal antibody that binds to the HER2 subdomain II and blocks ligand dependent HER2 heterodimerization with other HER receptors, ie. HER3, HER1 and HER4. Thus HERCEPTIN® along with PERJETA® provide a more comprehensive blockade of HER2 driven signaling pathways. The accelerated approval of PERJETA® for the neoadjuvant treatment of breast cancer was based on a randomized, multicenter, open-label, phase II trial, in which 417 patients with HER2-positive, operable, locally advanced or inflammatory breast cancer (T2-4d), were randomly assigned to receive preoperative therapy with either HERCEPTIN® plus TAXOTERE® (Docetaxel), PERJETA® plus HERCEPTIN® and TAXOTERE®, PERJETA® plus HERCEPTIN® or PERJETA® plus TAXOTERE®. Patients in the three drug group received preoperative therapy with PERJETA®, HERCEPTIN® and TAXOTERE® every 3 weeks for a total of 4 cycles and following surgery, all patients received 3 cycles of Fluorouracil, ELLENCE® (Epirubicin), and CYTOXAN® (Cyclophosphamide) - (FEC) IV every 3 weeks and HERCEPTIN® was continued every 3 weeks for a total of one year of therapy. The primary endpoint was pathological Complete Response (pCR) rate defined as the absence of invasive cancer in the breast. The FDA definition of pCR is the absence of invasive cancer in the breast and lymph nodes. All treatment groups were well balanced. Seven percent of patients had inflammatory breast cancer, 32% had locally advanced cancer and 70% had clinically node-positive breast cancer. Forty-seven percent of the patients had hormone receptor-positive disease. The FDA defined pCR rates were 39.3% in the PERJETA® plus HERCEPTIN® and TAXOTERE® group and 21.5% in the HERCEPTIN® plus TAXOTERE® group (P=0.0063). Of Interest, the pCR rates in the three drug group were lower in patients with hormone receptor positive tumors compared to patients with hormone receptor negative tumors. The most common adverse events in the three drug group were alopecia, diarrhea, nausea and neutropenia. Other significant side effects included decreased cardiac function, infusion-related reactions, hypersensitivity reactions and anaphylaxis. Based on clinical studies, for the neoadjuvant treatment of breast cancer, PERJETA® should be administered every 3 weeks for 3 to 6 cycles as part of one of the following treatment regimens for early breast cancer. • Four preoperative cycles of PERJETA® in combination with HERCEPTIN® and TAXOTERE® followed by 3 postoperative cycles of Fluorouracil, ELLENCE® and CYTOXAN® (FEC). • Three preoperative cycles of FEC alone followed by 3 preoperative cycles of PERJETA® in combination with TAXOTERE® and HERCEPTIN®. • Six preoperative cycles of PERJETA® in combination with TAXOTERE®, Carboplatin, and HERCEPTIN® (TCH). Following surgery, patients should continue to receive HERCEPTIN® to complete 1 year of treatment. The accelerated approval by the FDA was based solely on the improved pCR rate with the three drug combination with no demonstrable improvement in event-free survival or overall survival. A confirmatory phase III trial is underway, with results expected in 2016. Gianni L, Pienkowski T, Im YH, et al. Lancet Oncol. 2012;13:25-32
These proteins are transmembrane tyrosine kinases and are involved in normal cell growth and differentiation. HER1 is also known as Epidermal Growth Factor Receptor or EGFR. These receptors are activated following ligand binding, receptor pairing or dimerization and phosphorylation. This dimerization (receptor pairing) occurs often within the HER family of receptors. This has been no ligand identified for HER2 receptor, although it is able to form homo and heterodimers with other members of the HER family readily. Dimerization of HER2 and HER3 is believed to produce the strongest mitogenic signaling and activates two important pathways that regulate cell survival and growth - Mitogen Activated Protein Kinase (MAPK) pathway and PhosphoInositide 3-Kinase (PI3K) pathway. For this reason inhibiting HER2 dimerization appears to be an important step in the treatment of cancer. Overexpression of HER2 in breast cancer has been associated with higher risk for relapse as well as overall survival. Approximately 20 percent of breast cancers are HER2-positive. HERCEPTIN® is a humanized monoclonal antibody targeting HER2. It binds to the extracellular subdomain IV of the receptor and disrupts ligand independent HER2 downstream cell signaling pathways. PERJETA® is a recombinant, humanized, monoclonal antibody that binds to the HER2 subdomain II and blocks ligand dependent HER2 heterodimerization with other HER receptors, ie. HER3, HER1 and HER4. Thus HERCEPTIN® along with PERJETA® provide a more comprehensive blockade of HER2 driven signaling pathways. The accelerated approval of PERJETA® for the neoadjuvant treatment of breast cancer was based on a randomized, multicenter, open-label, phase II trial, in which 417 patients with HER2-positive, operable, locally advanced or inflammatory breast cancer (T2-4d), were randomly assigned to receive preoperative therapy with either HERCEPTIN® plus TAXOTERE® (Docetaxel), PERJETA® plus HERCEPTIN® and TAXOTERE®, PERJETA® plus HERCEPTIN® or PERJETA® plus TAXOTERE®. Patients in the three drug group received preoperative therapy with PERJETA®, HERCEPTIN® and TAXOTERE® every 3 weeks for a total of 4 cycles and following surgery, all patients received 3 cycles of Fluorouracil, ELLENCE® (Epirubicin), and CYTOXAN® (Cyclophosphamide) - (FEC) IV every 3 weeks and HERCEPTIN® was continued every 3 weeks for a total of one year of therapy. The primary endpoint was pathological Complete Response (pCR) rate defined as the absence of invasive cancer in the breast. The FDA definition of pCR is the absence of invasive cancer in the breast and lymph nodes. All treatment groups were well balanced. Seven percent of patients had inflammatory breast cancer, 32% had locally advanced cancer and 70% had clinically node-positive breast cancer. Forty-seven percent of the patients had hormone receptor-positive disease. The FDA defined pCR rates were 39.3% in the PERJETA® plus HERCEPTIN® and TAXOTERE® group and 21.5% in the HERCEPTIN® plus TAXOTERE® group (P=0.0063). Of Interest, the pCR rates in the three drug group were lower in patients with hormone receptor positive tumors compared to patients with hormone receptor negative tumors. The most common adverse events in the three drug group were alopecia, diarrhea, nausea and neutropenia. Other significant side effects included decreased cardiac function, infusion-related reactions, hypersensitivity reactions and anaphylaxis. Based on clinical studies, for the neoadjuvant treatment of breast cancer, PERJETA® should be administered every 3 weeks for 3 to 6 cycles as part of one of the following treatment regimens for early breast cancer. • Four preoperative cycles of PERJETA® in combination with HERCEPTIN® and TAXOTERE® followed by 3 postoperative cycles of Fluorouracil, ELLENCE® and CYTOXAN® (FEC). • Three preoperative cycles of FEC alone followed by 3 preoperative cycles of PERJETA® in combination with TAXOTERE® and HERCEPTIN®. • Six preoperative cycles of PERJETA® in combination with TAXOTERE®, Carboplatin, and HERCEPTIN® (TCH). Following surgery, patients should continue to receive HERCEPTIN® to complete 1 year of treatment. The accelerated approval by the FDA was based solely on the improved pCR rate with the three drug combination with no demonstrable improvement in event-free survival or overall survival. A confirmatory phase III trial is underway, with results expected in 2016. Gianni L, Pienkowski T, Im YH, et al. Lancet Oncol. 2012;13:25-32
Effects of bisphosphonate treatment on recurrence and cause-specific mortality in women with early breast cancer: a meta-analysis of individual patient data from randomised trials.
SUMMARY: Bisphosphonates are presently indicated for the treatment of osteoporosis. The approved bisphosphonates in the U.S. include FOSAMAX® (Alendronate), BONIVA® (Ibandronate), ACTONEL® (Risedronate) and RECLAST® (Zoledronic acid). Several trials conducted over the past 2 decades have suggested that bisphosphonates may have anti proliferative effect in patients with breast cancer. Data from several Women's Health Initiative (WHI) studies involving more than 150,000 healthy postmenopausal women, of whom 2216 used oral bisphosphonates, revealed that women taking bisphosphonates for osteoporosis had a 32% reduction in invasive breast cancer. The AZURE investigators conducted a study to determine whether the addition of RECLAST® (Zoledronic acid) to standard adjuvant therapy would improve disease outcomes in patients with early-stage breast cancer. They noted that in the subset analysis, the addition of RECLAST® significantly improved disease free survival and overall survival in postmenopausal patients, independent of estrogen receptor status, tumor stage, and lymph node involvement (N Engl J Med 2011;365:1396-1405). With this background, the authors belonging to the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), conducted a meta-analysis and reviewed data from 15 years of bisphosphonate trials, which included 36 trials of adjuvant bisphosphonates in breast cancer and involved over 17,000 pre and postmenopausal women. RECLAST® (Zoledronic acid) and Clodronate were the most common bisphosphonates used in these trials. The primary outcomes analyzed were time to distant recurrence, local recurrence, new second primary breast cancer (ipsilateral or contralateral), time to first distant recurrence (ignoring any previous locoregional or contralateral recurrences), and breast cancer mortality. Planned subset analyses included site of recurrence, site of first distant metastasis (bone vs other), menopausal status (pre, peri and post) type of bisphosphonate (aminobisphosphonates such as RECLAST® or Clodronate) and drug schedule of bisphosphonate therapy (for bone protection vs advanced cancer). Adjuvant bisphosphonates resulted in a 34% reduction in the risk of bone recurrence (P = 0.00001) and a 17% reduction in the risk of breast cancer death (P =0.004). This benefit was seen regardless of estrogen receptor status, nodal status or whether chemotherapy was used or not. Bisphosphonates had no significant impact on non-breast cancer related deaths, contralateral breast cancer or loco-regional recurrence. In this meta-analysis, all these benefits were only seen in postmenopausal women and premenopausal women had no benefit on any disease outcomes with bisphosphonates. The authors emphasized that low estrogen environment as is seen in postmenopausal women, or women rendered menopausal by suppression of ovarian function is a prerequisite for adjuvant bisphosphonate activity. Based on this large meta-analysis, the authors recommended the use of RECLAST® once every six months or oral Clodronate, where available. Because of paucity of data, they do not recommend the use of weekly dose of oral bisphosphonates, often used to prevent osteoporosis, to achieve these benefits. Coleman R, Gnant M, Paterson A, et al. San Antonio Breast Cancer Symposium 2013; San Antonio, TX. Abstract S4-07.
The AZURE investigators conducted a study to determine whether the addition of RECLAST® (Zoledronic acid) to standard adjuvant therapy would improve disease outcomes in patients with early-stage breast cancer. They noted that in the subset analysis, the addition of RECLAST® significantly improved disease free survival and overall survival in postmenopausal patients, independent of estrogen receptor status, tumor stage, and lymph node involvement (N Engl J Med 2011;365:1396-1405). With this background, the authors belonging to the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), conducted a meta-analysis and reviewed data from 15 years of bisphosphonate trials, which included 36 trials of adjuvant bisphosphonates in breast cancer and involved over 17,000 pre and postmenopausal women. RECLAST® (Zoledronic acid) and Clodronate were the most common bisphosphonates used in these trials. The primary outcomes analyzed were time to distant recurrence, local recurrence, new second primary breast cancer (ipsilateral or contralateral), time to first distant recurrence (ignoring any previous locoregional or contralateral recurrences), and breast cancer mortality. Planned subset analyses included site of recurrence, site of first distant metastasis (bone vs other), menopausal status (pre, peri and post) type of bisphosphonate (aminobisphosphonates such as RECLAST® or Clodronate) and drug schedule of bisphosphonate therapy (for bone protection vs advanced cancer). Adjuvant bisphosphonates resulted in a 34% reduction in the risk of bone recurrence (P = 0.00001) and a 17% reduction in the risk of breast cancer death (P =0.004). This benefit was seen regardless of estrogen receptor status, nodal status or whether chemotherapy was used or not. Bisphosphonates had no significant impact on non-breast cancer related deaths, contralateral breast cancer or loco-regional recurrence. In this meta-analysis, all these benefits were only seen in postmenopausal women and premenopausal women had no benefit on any disease outcomes with bisphosphonates. The authors emphasized that low estrogen environment as is seen in postmenopausal women, or women rendered menopausal by suppression of ovarian function is a prerequisite for adjuvant bisphosphonate activity. Based on this large meta-analysis, the authors recommended the use of RECLAST® once every six months or oral Clodronate, where available. Because of paucity of data, they do not recommend the use of weekly dose of oral bisphosphonates, often used to prevent osteoporosis, to achieve these benefits. Coleman R, Gnant M, Paterson A, et al. San Antonio Breast Cancer Symposium 2013; San Antonio, TX. Abstract S4-07.
Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial
SUMMARY: NOLVADEX® (Tamoxifen) is a Selective Estrogen Receptor Modulator (SERM), approved by the FDA to also reduce the incidence of breast cancer in women considered to be at high risk (ChemoPrevention). This agent however has been linked to endometrial cancer and thromboembolic phenomenon in some women. ARIMIDEX® is a non-steroidal Aromatase Inhibitor proven to be more efficacious than NOLVADEX® both in metastatic and adjuvant settings. Similar to NOLVADEX®, ARIMIDEX® also prevents the occurrence of new primary tumors in the contralateral breast, in postmenopausal females. With this background, the International Breast Cancer Intervention Study (IBIS) -II trial enrolled 3864 postmenopausal women considered to be at increased risk of breast cancer and randomized them to receive either ARIMIDEX® (Anastrazole) 1 mg QD (N=1920) or Placebo QD (N=1944)for 5 years. Patients were considered to be at high risk if they had a family history of breast cancer, atypical ductal hyperplasia, lobular carcinoma in-situ or dense breast tissue. The median age was 59 years. The primary end point was histologically confirmed Invasive breast cancer or Ductal Carcinoma In- Situ. At a median follow up of 5 years, the incidence of breast cancer in the ARIMIDEX® group was 2% and in the placebo group was 4%. The predicted cumulative incidence of breast cancer after 7 years was 2.8% in the ARIMIDEX® group and 5.6% in the placebo group (HR=0.47; P<0.0001). This represented a 53% reduction in the risk of breast cancer. The adverse events were comparable in both groups with slight increase in the musculoskeletal and vasomotor events noted in the ARIMIDEX® group. The authors concluded that ARIMIDEX® reduced the risk of primary breast cancer by more than 50% in high risk postmenopausal women. Other unrelated studies have shown that acceptance of breast cancer prevention with medications (ChemoPrevention) appeared to be related to education and income, putting emphasis on education and adequate counseling of women, considered to be at high risk. Cuzick J, Sestak I, Forbes JF, et al. The Lancet, Early Online Publication, 12 December 2013
Safety and Efficacy of Everolimus With Exemestane vs. Exemestane Alone in Elderly Patients With HER2-Negative, Hormone Receptor-Positive Breast Cancer in BOLERO-2
SUMMARY: Exemestane (AROMASIN®) is a steroidal, Aromatase Inhibitor (AI) which has been shown to improve Progression Free Survival (PFS) in postmenopausal patients with ER positive metastatic breast cancer. Patients tend to have limited clinical benefit with subsequent hormonal interventions if their disease recurs or progresses on AI’s. Further, cytotoxic chemotherapy may not be feasible in elderly patients. The PI3K/AKT/mTOR is a complex pathway, essential for cell proliferation, survival and apoptosis (programmed cell death). This pathway is of interest because of its increased activity in malignant cells as a result of amplification or mutation of the genes associated with PI3-kinase (phosphatidylinositol-3 kinase) and AKT, as well as loss of function of PTEN. PTEN normally prevents activation of AKT and its downstream pathways. Elevated ER signaling and breast cancer progression has been associated with activation of this mTOR (mammalian Target Of Rapamycin) pathway. Everolimus (AFINITOR®) is a mTOR inhibitor that has been shown to inhibit ER signaling and restore sensitivity to anti-estrogen therapies. In the phase III BOLERO-2 study, patients with ER positive, advanced breast cancer, who had recurred or progressed on prior therapy with nonsteroidal AI’s such as Anastrozole (ARIMIDEX®) or Letrazole (FEMARA®), had doubling of PFS with a combination of AROMASIN® and AFINITOR® compared to AROMASIN® alone. The authors now report the Safety and Efficacy of AROMASIN® and AFINITOR® combination in 316 patients, 65 years of age or older, out of the 724 patients in the BOLERO-2 study. The median follow up was 18 months. Baseline treatment characteristics were similar in all treatment subsets. The treatment combination resulted in 55% reduction in the risk of disease progression regardless of the age compared to AROMASIN® alone (PFS - 7.8 months vs 3.2 months, HR=0.45, P<0.0001). Further the combination treatment resulted in greater clinical benefit compared to single agent AROMASIN®. Approximately two third of the patients regardless of their age required dose modifications and the most common adverse events in all age groups with similar incidences were stomatitis, rash, fatigue , hyperglycemia and pneumonitis. It is recommended that elderly patients be carefully monitored and toxicities promptly addressed during treatment. The authors concluded that a combination of AROMASIN® and AFINITOR® can provide significant clinical benefit with acceptable toxicities, even in patients 65 years of age or older, thereby delaying the need for chemotherapy. Pritchard KI, Burris HA, Ito Y, et al. Clinical Breast Cancer 2013;13:421-432.
Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial.
SUMMARY: PERJETA® (Pertuzumab) is a recombinant, humanized, monoclonal antibody that binds to the HER2 dimerization domain and prevents the dimerization of HER2 with other HER receptors, ie. HER3, HER1 and HER4. HERCEPTIN® (Trastuzumab) is a monoclonal antibody that specifically targets the HER2 receptor and blocks the downstream signalling pathways. The accelerated approval of PERJETA® for the neoadjuvant treatment of breast cancer was based on a randomized, multicenter, open-label, phase II trial, in which 417 patients with HER2-positive, operable, locally advanced or inflammatory breast cancer (T2-4d), were randomly assigned to receive preoperative therapy with either HERCEPTIN® plus TAXOTERE® (Docetaxel), PERJETA® plus HERCEPTIN® and TAXOTERE®, PERJETA® plus HERCEPTIN® or PERJETA® plus TAXOTERE®. Patients in the three drug group received preoperative therapy with PERJETA®, HERCEPTIN® and TAXOTERE® every 3 weeks for a total of 4 cycles and following surgery, all patients received 3 cycles of Fluorouracil, ELLENCE® (Epirubicin), and CYTOXAN® (Cyclophosphamide) - (FEC) IV every 3 weeks and HERCEPTIN® was continued every 3 weeks for a total of one year of therapy. The primary endpoint was pathological Complete Response (pCR) rate defined as the absence of invasive cancer in the breast. The FDA definition of pCR is the absence of invasive cancer in the breast and lymph nodes. All treatment groups were well balanced. Seven percent of patients had inflammatory breast cancer, 32% had locally advanced cancer and 70% had clinically node-positive breast cancer. Forty-seven percent of the patients had hormone receptor-positive disease. The FDA defined pCR rates were 39.3% in the PERJETA® plus HERCEPTIN® and TAXOTERE® group and 21.5% in the HERCEPTIN® plus TAXOTERE® group P=0.0063). Of Interest, the pCR rates in the three drug group were lower in patients with hormone receptor positive tumors compared to patients with hormone receptor negative tumors. The most common adverse events in the three drug group were alopecia, diarrhea, nausea and neutropenia. The accelerated approval by the FDA was based solely on the improved pCR rate with the three drug combination with no demonstrable improvement in event-free survival or overall survival. A confirmatory phase III trial is underway, with results expected in 2016. Gianni L, Pienkowski T, Im YH, et al. Lancet Oncol. 2012;13:25-32
Primary results from EMILIA, a phase III study of trastuzumab emtansine (T-DM1) versus capecitabine (X) and lapatinib (L) in HER2-positive locally advanced or metastatic breast cancer (MBC) previously treated with trastuzumab (T) and a taxane.
SUMMARY: The HER or erbB family of receptors consist of HER1,HER2,HER3 and HER4. Overexpression of HER2 in breast cancer has been associated with higher risk for relapse as well as overall survival. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2. It binds to the extracellular domain of the receptor and blocks the downstream cell signaling pathways. KADCYLA® (Ado-Trastuzumab Emtansine, T-DM1) is an antibody-drug conjugate (ADC) comprised of the antibody Trastuzumab and the chemotherapy agent Emtansine, linked together. It inhibits HER2 signaling and destroys the HER2-positive tumor cells by delivering the chemotherapy agent Emtansine which binds to tubulin, directly inside the tumor cells. The EMILIA trial is a phase III study in which 991 patients with HER2-positive locally advanced or metastatic breast cancer who had previously received treatment with HERCEPTIN® and a Taxane chemotherapy, were enrolled. Patient received either KADCYLA® or XELODA® (Capecitabine) and TYKERB® (Lapatinib) doublet. The primary endpoints were Progression Free Survival (PFS), Overall Survival (OS) and safety. Patients receiving KADCYLA® had an improved PFS compared to XELODA® and TYKERB® (9.6 months vs 6.4 months, HR=0.65, P <0.0001). The median overall survival was 30.9 months in the KADCYLA® group and 25.1 months with the XELODA® and TYKERB® doublet. KADCYLA® is the fourth drug approved by the FDA, that targets the HER2 oncogene. The other FDA-approved drugs used to treat HER2-positive breast cancer include HERCEPTIN® (1998), TYKERB® (2007) and PERJETA® (Pertuzumab) (2012). Blackwell KL, Miles D, Gianni L, et al. J Clin Oncol 30, 2012 (suppl; abstr LBA1)
Results of a randomized phase 2 study of PD 0332991, a cyclin-dependent kinase (CDK) 4/6 inhibitor, in combination with letrozole vs letrozole alone for first-line treatment of ER+/HER2- advanced breast cancer (BC).
SUMMARY: PD 0332991 is an oral, selective inhibitor of CDK4/6 kinases. This agent interrupts cellular DNA synthesis by inhibiting the progression of the cell cycle from G1 to S phase and thus prevents tumor cell growth. The results presented, includes the pooled data from the study of 2 cohorts of patients. Both groups included postmenopausal women with advanced breast cancer and ER positive, HER2 negative tumors. Patients were randomized 1:1 to receive either letrozole (FEMARA®) along with PD 0332991 or FEMARA® alone. Group 1 enrolled 66 patients and Group 2 enrolled 99 patients. Group 2 patient tumors were also evaluated for the biomarkers cyclinD1 amplification and/or loss of p16, by FISH analysis. For both these study groups, the primary endpoint was Progression Free Survival (PFS). Secondary endpoints included response rates, overall survival, safety, and biomarker correlates. Data from the pooled analysis which included 165 women from both the groups demonstrated a median PFS of 26.1 months for the combination compared to 7.5 months with FEMARA® alone. This represented a 63% reduction in risk of progression (hazard ratio =0.37; P < 001). The most common adverse events noted in the combination group included uncomplicated neutropenia, anemia, and fatigue. Biomarkers expression (cyclinD1 amplification and/or loss of p16) had no impact on outcomes suggesting that the biomarker for PD0332991 may be the estrogen receptor itself rather than CDK4/6 kinases. Finn RS, Crown JP, Lang I, et al. CTRC-AACR San Antonio Breast Cancer Symposium 2012; Abstract S1-6.
Prospective study of treatment pattern, effectiveness, and safety of zoledronic acid (ZOL) therapy beyond 24 months: subgroup analysis of patients (pts) with metastatic bone disease (MBD) from breast cancer (BC).
Safety and Efficacy of Zoledronic Acid Beyond 24 Months in Breast Cancer Patients.
SUMMARY: There is not much data on the effectiveness and safety of Zoledronic acid (ZOMETA®) beyond 2 years. Two studies one from Belgium and the other from Japan shed some light on this issue. In the prospective multicenter Belgian trial, 108 women with breast cancer prior to enrollment had received at least 24 months of therapy with ZOMETA® infusions given every 3 to 4 weeks and 21% had received at least 48 months of therapy. They were followed for 18 months and monitored for Skeletal Related Events (SRE’s), Osteo Necrosis of the Jaw bone (ONJ), renal failure and hypocalcemia. During this follow up period, SRE’s were low and 83% of the women were free of SRE’s. ONJ was seen in 7 patients (4.5%). The rate of ONJ however rose to 11% after any invasive dental procedure. The risk of renal failure was low but increased to 12%when the dose of ZOMETA® was not adjusted for renal function. The Japanese study was a retrospective analysis of 83 patients who had been treated with ZOMETA® for at least 24 months (median 33 months). SRE’s were low and the frequency of ONJ was 3.6% compared to 2.4% for those patients who had been ZOMETA® for shorter periods. Both these studies demonstrated that longer duration of therapy with ZOMETA® resulted in increased rate of ONJ. Therefore, particular attention should be paid to prevent this complication by adhering to proper dental hygiene and avoiding dental trauma and extractions. Van den Wyngaert T, Delforge M, Doyen C, et al. and Suzuki Y, Saito Y, Ogiya R, et al. CTRC-AACR San Antonio Breast Cancer Symposium 2012; Poster P3-13-01 and P3-13-02.
Final analysis of overall survival for the phase III CONFIRM trial: fulvestrant 500 mg versus 250 mg.
SUMMARY: Fulvestrant (FASOLODEX®) is an Estrogen Receptor (ER) antagonist and downregulates the cellular levels of ER in a dose-dependent manner. The CONFIRM trial is a phase III study in which postmenopausal women with estrogen receptor (ER) positive advanced breast cancer, who had progressed after prior endocrine therapy, were randomized to be treated with either FASLODEX® 500 mg (n=362) or FASLODEX® 250 mg (n=374) every 28 days. The primary end point for this study was Progression Free Survival (PFS). In the primary analysis, FASLODEX® 500 mg was associated with a statistically significant increase in PFS compared with FASLODEX® 250 mg. Even though there was a trend towards improved overall survival (OS) with the higher dose, this was not statistically significant. In the updated second survival analysis presented at this symposium, the median OS trend prevailed with FASLODEX® 500mg compared with FASLODEX® 250mg, given every 28 days (26.4 months vs 22.3 months, P=0.16). This translated into a 4 month increase in median overall survival and a 19% reduction in the risk of death. The authors concluded that the higher dose of FASLODEX® may indeed confer some survival benefit to this patient subsets. Di Leo A, Jerusalem G, Petruzelka L, et al. CTRC-AACR San Antonio Breast Cancer Symposium; 2012; Abstract S1-4.
Neurocognitive impact in adjuvant chemotherapy for breast cancer linked to fatigue: a prospective functional MRI study.
SUMMARY: Cognitive impairment in patients with breast cancer has been frequently attributed to chemotherapy (Chemo Brain) without any data supporting this hypothesis. Utilizing functional magnetic resonance imaging, brain function was tested while the patients were performing a working memory task in the scanner, before adjuvant treatment and then one month after adjuvant treatment. Sixty six breast cancer patients with Stages 0-IIIA were studied and their cognitive function was compared with 32 healthy controls. Patients on treatment were receiving either an anthracyline-based adjuvant chemotherapy regimen (n = 29) or radiotherapy (n = 37). Patients self-reported on levels of cognitive functioning and fatigue after each imaging study. Pretreatment brain imaging revealed decreased functioning in the frontal lobe of the brain (responsible for memory and cognition), compared to the controls and this cognitive impairment was most severe in patients awaiting chemotherapy, whereas the radiotherapy group fell between the pre-chemotherapy and control group. Of Interest, the decreased functioning in the frontal lobe area before treatment predicted the severity of fatigue. Further, those with greater fatigue experienced greater cognitive impairment over time. The authors concluded that the cognitive problems are probably related to worry and fatigue prior to treatment intervention rather than the treatment itself. They recommend identifying patients at risk and early intervention. Cimprich B, Hayes DF, Askren MK, et al. CTRC-AACR San Antonio Breast Cancer Symposium, 2012; Abstract S6-3.
ATLAS. 10 v 5 years of adjuvant tamoxifen (TAM) in ER+ disease: effects on outcome in the first and in the second decade after diagnosis.
SUMMARY: Historically, adjuvant treatment with tamoxifen beyond five years has not been recommended, as the benefit of tamoxifen beyond five years was unknown but there was an increased risk of endometrial cancer. (Tamoxifen Treatment for Breast Cancer and Risk of Endometrial Cancer: A Case-Control Study, J Natl Cancer Inst 2005; 97: 375-384). The ATLAS trial has now shed some light on the duration of tamoxifen treatment. In this study, 6846 women with ER positive breast cancer were enrolled between 1996 and 2005 and following five years of adjuvant tamoxifen were randomized to five additional years of tamoxifen or observation. Women who continued on tamoxifen had a 25% lower recurrence rate and 29% lower breast cancer mortality rate compared with women who stopped tamoxifen after five years. This significant benefit was seen in the second decade after diagnosis with little benefit seen in the 5-9 year period after diagnosis. There was a higher cumulative risk of death from endometrial cancer for those who continued tamoxifen beyond 5 years compared to those who did not (0.4% vs 0.2%). It appears that the reduction in breast cancer deaths outweigh the risk of endometrial cancer and other adverse events associated with longer duration of tamoxifen use. This new information will help physicians make appropriate treatment recommendations for those patients on adjuvant tamoxifen. Davis C, Hongchao P, Godwin J, et al. CTRC-AACR San Antonio Breast Cancer Symposium, 2012; Abstract S1-2.
Chemotherapy prolongs survival for isolated local or regional recurrence of breast cancer: the CALOR trial (Chemotherapy as Adjuvant for Locally Recurrent breast cancer; IBCSG 27-02, NSABP B-37, BIG 1-02).
SUMMARY: Approximately 5% to 15% of all breast cancer patients present with local and/or regional recurrences. The role of adjuvant chemotherapy after resection of an isolated recurrence of breast cancer is unknown. To address this, in the CALOR trial, 162 patients with recurrent tumors underwent surgery and were then randomized to receive either four cycles of a multidrug chemotherapy regimen of investigator’s choice (n=85) or observation (n=77). The median age was 56 years. The treatment groups were well balanced and 68% and 58% of patients in the observation and treatment arms respectively received prior adjuvant chemotherapy. The median time to recurrence was 5 years in the treatment group and 6 years in the observation group. The primary end point was disease free survival (DFS). The 5-year DFS rate for the chemotherapy group and observation group was 69% vs 57% respectively. This translated into a relative risk reduction of 41% (hazard ratio [HR], 0.59; P = 0.045). The 5-year overall survival rate between the chemotherapy group and observation group was 88% vs 76% with a 59% reduction in the relative risk for death for patients treated with chemotherapy (HR, 0.41; P = 0.02). In patients with ER-negative tumors, the 5-year DFS was 67% in the treatment arm versus 35% in the control group (HR = 0.32; P = 0.007). This data supports the role of chemotherapy for patients with completely resected isolated locoregional recurrences of breast cancer. Aebi S, Gelber S, Láng I, et al. Presented at: CTRC-AACR San Antonio Breast Cancer Symposium, 2012. Abstract S3-2.
Primary results from EMILIA, a phase III study of trastuzumab emtansine (T-DM1) versus capecitabine (X) and lapatinib (L) in HER2-positive locally advanced or metastatic breast cancer (MBC) previously treated with trastuzumab (T) and a taxane.
SUMMARY: The HER or erbB family of receptors consist of HER 1,HER 2,HER 3 and HER 4. Overexpression of HER 2 in breast cancer has been associated with higher risk for relapse as well as overall survival. Trastuzumab is a humanized monoclonal antibody targeting HER 2. It binds to the extracellular domain of the receptor and blocks the downstream cell signaling pathways. Trastuzumab emtansine (T-DM1) is an antibody-drug conjugate (ADC) comprised of the antibody trastuzumab and the chemotherapy agent DM1 linked together and inhibits HER2 signaling and destroys the HER2-positive tumor cells by delivering the chemotherapy agent DM1 directly inside the tumor cells. The EMILIA trial is a phase III study in which 978 patients with HER2-positive locally advanced or metastatic breast cancer who had previously received treatment with HERCEPTIN® (Trastuzumab) and a taxane chemotherapy, were enrolled. Patient received either trastuzumab emtansine (T-DM1) or XELODA® (Capecitabine) and TYKERB® (Lapatinib) doublet. The primary endpoints were Progression Free Survival (PFS), Overall Survival (OS) and safety. Patients receiving T-DM1 had an improved PFS compared to XELODA® and TYKERB® (9.6 months vs 6.4 months, HR=0.65, P <0.0001). A final OS analysis will be available at a later date. Blackwell KL, Miles D, Gianni L, et al. J Clin Oncol 30, 2012 (suppl; abstr LBA1)
Everolimus in Postmenopausal Hormone-Receptor–Positive Advanced Breast Cancer
SUMMARY: The mechanism of resistance to hormonal therapy in breast cancer is thought to be related to the activation of mTOR pathway, independent of ER (estrogen receptor) signalling pathway. To test this hypothesis, the authors in this study (BOLERO-2) decided to combine mTOR inhibition with estrogen deprivation. In a phase III trial, patients with ER positive, advanced breast cancer, who had recurred or progressed on prior therapy with nonsteroidal aromatase inhibitors (anastrozole or letrazole), were randomized in a 2:1 ratio to either receive everolimus (mTOR inhibitor) and exemestane (steroidal aromatase inhibitor) or exemestane and placebo. The primary end point of Progression Free Survival was significant with 10.6 months for mTOR inhibitor plus exemestane vs 4.1 months for exemestane plus placebo, according to central review (hazard ratio, 0.36; P<0.001). This new paradigm of adding everolimus to hormonal therapy should change the way we manage patients with advanced breast cancer, who are resistant to hormonal therapy alone. Baselga J, Campone M, Piccart M, et al. N Engl J Med 2012; 366:520-529
Risk Factors for Breast Cancer for Women Aged 40 to 49 Years
SUMMARY: There is presently no clear consensus regarding breast cancer screening for women in their 40’s. The risk/benefit ratio of screening mammography has not been properly defined in this age group. In this review, Nelson and colleagues identified the risk factors for breast cancer and concluded that there is a 2 fold increase in the risk for breast cancer in women 40-49 years of age, who have dense breast tissue and first degree relatives with breast cancer. This meta analysis has identified a subset of patients in their 40’s who would benefit from screening mammography. Nelson HD, Zakher B, Cantor A, et al. Ann Intern Med. 2012;156:635-648
Pertuzumab plus Trastuzumab plus Docetaxel for Metastatic Breast Cancer
SUMMARY: The CLEOPATRA trial is a phase III trial in which 808 HER positive metastatic breast cancer patients, for their first line treatment, were randomized to either HERCEPTIN® plus Docetaxel or the above two drug combination given along with PERJETA® (Pertuzumab). PERJETA® is a recombinant humanized monoclonal antibody that binds to the HER2 dimerization domain and prevents the dimerization of HER2 with other HER receptors ie. HER3, HER1 and HER4. The addition of PERJETA® to the combination of HERCEPTIN® and Docetaxel significantly prolonged progression-free survival compared to HERCEPTIN® plus Docetaxel alone (18.5 months vs 12.4 months, P<0.001). This benefit was seen without increase in cardiotoxicity. It appears that PERJETA® complements HERCEPTIN® in targeting HER-2 receptor. Baselga J, Cortés J, Kim S, et al. N Engl J Med 2012; 366:109-119
Exemestane for Breast-Cancer Prevention in Postmenopausal Women
SUMMARY: Breast cancer is the second most common cause of death in the United States. Presently two Selective Estrogen Receptor Modulators (SERM’s), tamoxifen and raloxifene are approved by the FDA, for the primary prevention of breast cancer. Tamoxifen has been associated with increased risk of venous thromboembolism and endometrial cancers. Raloxifene can also be associated with venous thromboembolism but has not been associated with endometrial cancers. Exemestane is an irreversible, steroidal aromatase inactivator. This agent was evaluated in a randomized, placebo controlled, double blind study, to reduce the risk of invasive breast cancer in postmenopausal women, considered to be at moderately increased risk of developing breast cancer. Risk factors included age over 60 years, Gail five year risk score greater than 1.66% and prior atypical ductal/ lobular hyperplasia, LCIS, or DCIS with mastectomy. At a median follow up of 3 years, exemestane reduced the relative incidence of invasive breast cancers by 65% compared to placebo and this benefit was accomplished without serious toxicities and with minimal changes in quality of life. This is the first aromatase inhibitor/inactivator to demonstrate proven efficacy in breast cancer prevention, in a randomized clinical trial. N Engl J Med 2011; 364:2381-2391
Iniparib plus Chemotherapy in Metastatic Triple-Negative Breast Cancer
SUMMARY: Patients with triple negative breast cancer have inherent defects in several DNA repair pathways. These cancer cells therefore become increasing dependent on another DNA damage repair pathway called base excision repair (BER) pathway, for survival. It so happens that PARP 1(PolyAdenosine diphosphate Ribose Polymerase) is an important enzyme regulating the BER pathway. By inhibiting PARP1, the BER pathway is inhibited leading to extreme levels of DNA damage and eventual death of cancer cells. In an article published in the January, 2011 issue of the NEJM, the addition of a PARP inhibitor Iniparib to chemotherapy improved clinical benefit and survival of patients with advanced triple negative breast cancer. A phase II open label trial was conducted, in which 123 patients with metastatic triple negative breast cancer were randomly assigned to receive a combination of carboplatin and GEMZAR® (gemcitabine) with or without iniparib. Patients who received chemotherapy in combination with iniparib demonstrated improved rate of clinical benefit (partial or complete response plus stable disease for 6 or more months) from 34% to 56% (P=0.01). This was the primary end point. The addition of iniparib to chemotherapy also prolonged the median progression free survival from 3.6 months to 5.9 months (HR for progression, 0.59; P=0.01) and median overall survival from 7.7 months to 12.3 months (HR for death, 0.57; P=0.01). These gains were achieved without any significant increase in toxicities. This difficult to treat subtype of breast cancer may soon become extinct. N Engl J Med 2011; 364:205-214
A phase III study (EMBRACE) of eribulin mesylate versus treatment of physician's choice in patients with locally recurrent or metastatic breast cancer previously treated with an anthracycline and a taxane.
SUMMARY: HALAVEN ® (Eribulin) is a non taxane inhibitor of microtubule dynamics and is a synthetic analog of halichondrin B, a product derived from a a sea sponge Halichodria okadai. It triggers apoptosis of cancer cells following prolonged mitotic inhibition. The EMBRACE trial is a randomized open label phase III study involving 762 heavily pretreated patients with locally recurrent or metastatic breast cancer. These patients must have had at least two prior chemotherapy regimens including taxanes and an anthracycline. Patients were randomized to either HALAVEN ® (508 patients) or to an approved treatment of their physician's choice and this could be single agent chemotherapy, hormonal therapy, biological therapy or palliative radiation therapy (254 patients). Nineteen percent of the patients had triple negative disease. There was a statistically significant improvement in overall survival in the HALAVEN ® group 13.1 months compared to 10.7 months in the control group. At one year, 54% of the patients were alive in the HALAVEN ® group compared to 44% in the control group. This statistically significant benefit was also seen in the overall response rates. We now have another agent with a distinct survival advantage for heavily pretreated metastatic breast cancer patients. J Clin Oncol 28:18s, 2010 (suppl; abstr CRA1004)
